What Is Heat Simply?
What is Heat?
Heat is a form of energy transfer between objects or systems that are at different temperatures. Heat always flows spontaneously from a hotter object to a colder object until they reach thermal equilibrium. Heat is a kinetic form of energy associated with the random motion of atoms and molecules in matter. The hotter an object is, the faster its atoms and molecules vibrate and move. As the hot object comes in contact with a colder object, the increased molecular motion and energy is transferred from the hotter object to the colder object. This energy transfer manifests as heat.
While heat and temperature are related, they are distinct concepts. Temperature measures the average kinetic energy of molecular motion in a substance while heat is the transfer of energy due to a temperature difference. An object does not contain heat; its molecules contain kinetic energy that can be transferred as heat. Heat is a process, while temperature describes an equilibrium state. Additionally, the amount of heat transferred depends on the temperature difference, the materials involved, the surface area, and the time interval.
Forms of Heat Transfer
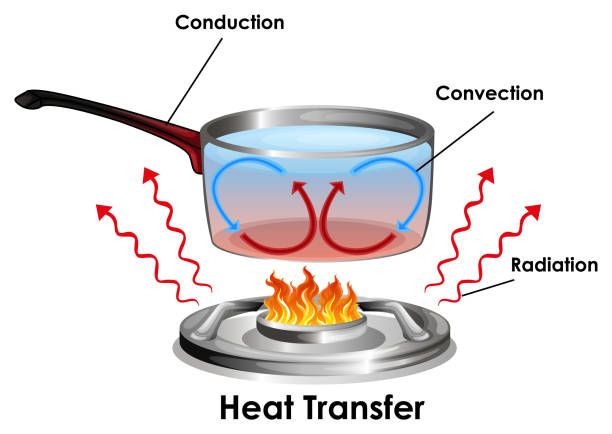
There are three main forms of heat transfer: conduction, convection, and radiation.
Conduction
Conduction is the transfer of heat between substances that are in direct contact with each other. It occurs when electrons from the hottest area transfer kinetic energy to electrons in cooler areas. Metals are good thermal conductors. Conduction can occur in solids, liquids, or gases.
Convection
Convection is the transfer of heat by the movement of heated fluid particles from one place to another. Convection depends on the movement of currents within a fluid. For example, when water is heated on a stove, hot water rises while cooler water sinks. This sets up convection currents that transfer heat through the water.
Radiation
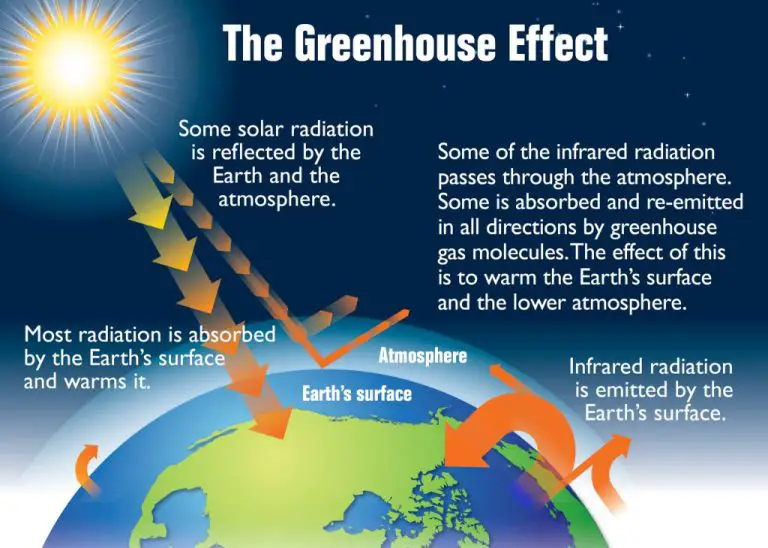
Radiation is the transfer of heat not requiring a medium to travel through. It occurs when electromagnetic waves or photons carry heat directly from the heat source to an object. For example, we feel the warmth of the sun across space by radiation.
Measuring Heat
Heat is measured in units called calories or joules. A calorie is the amount of heat required to raise the temperature of 1 gram of water by 1°C. A joule is the SI unit of heat and defined as the amount of work done when a force of 1 Newton moves an object 1 meter.
The amount of heat required to change an object’s temperature depends on its heat capacity. Heat capacity measures how much heat must be absorbed or lost for a substance to change its temperature by 1°C. Substances with higher heat capacities require more heat to change their temperatures.
An object’s specific heat is the amount of heat required to raise the temperature of 1 gram of a substance by 1°C. Specific heat indicates how easily a material’s temperature changes when heat is added or removed. Substances like water have high specific heats, while metals have low specific heats.
Effects of Heat
Heat can produce several important effects, including:
Thermal Expansion
When an object is heated, the kinetic energy of its molecules increases. The molecules vibrate more rapidly and usually maintain a greater average separation. This causes materials to expand in volume. Thermal expansion generally increases with temperature.
Different materials expand by different amounts. This thermal expansion property is used in many everyday applications. For example, bimetal strips bend when heated due to the different thermal expansion rates of metals. Thermal expansion must also be accounted for when fitting machine parts together and when designing bridges and roads.
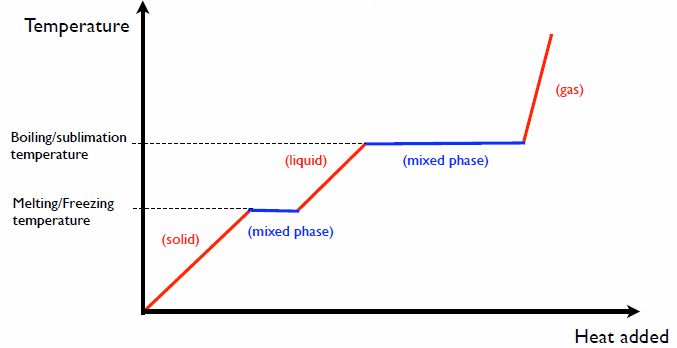
Phase Changes
Adding heat can cause phase transitions in matter, where a material changes from one state to another. For example, water transforms from solid ice to liquid water to gaseous steam with increasing temperature. Phase changes require the input or release of energy called latent heat.
The absorption or release of latent heat allows materials to maintain a constant temperature during a phase transition, even as heat is continually added or removed. Materials can thus store substantial amounts of energy in their bonds before changing phase.
Chemical Reactions
Most chemical reactions speed up at higher temperatures. Added heat boosts the kinetic energy of reactant molecules, allowing them to more easily break bonds and rearrange to form products. Some reactions will essentially not occur below a certain activation temperature.
Exothermic reactions release heat, while endothermic reactions absorb heat. Changing the temperature can help shift the equilibrium point of reversible reactions. Heat also enables thermal decomposition and combustion reactions.
Heat Transfer Mechanisms
There are three main mechanisms of heat transfer: conduction, convection, and radiation.
Conduction
Conduction is the transfer of heat between substances that are in direct contact with each other. The better the conductor, the more rapidly heat will be transferred. Examples of good conducting materials are metals such as copper, aluminum, and steel.
A pot on a stove transfers heat from the hot burner to the cooler water through conduction. The metal pot bottom is in direct contact with the hot metal stove burner so heat energy is transferred between them.
Convection
Convection is the transfer of heat by the movement of heated fluid or gas. As the fluid/gas is heated, it expands, becomes less dense, and rises. Cooler fluid/gas then takes its place. This moving current transfers the heat from one place to another.
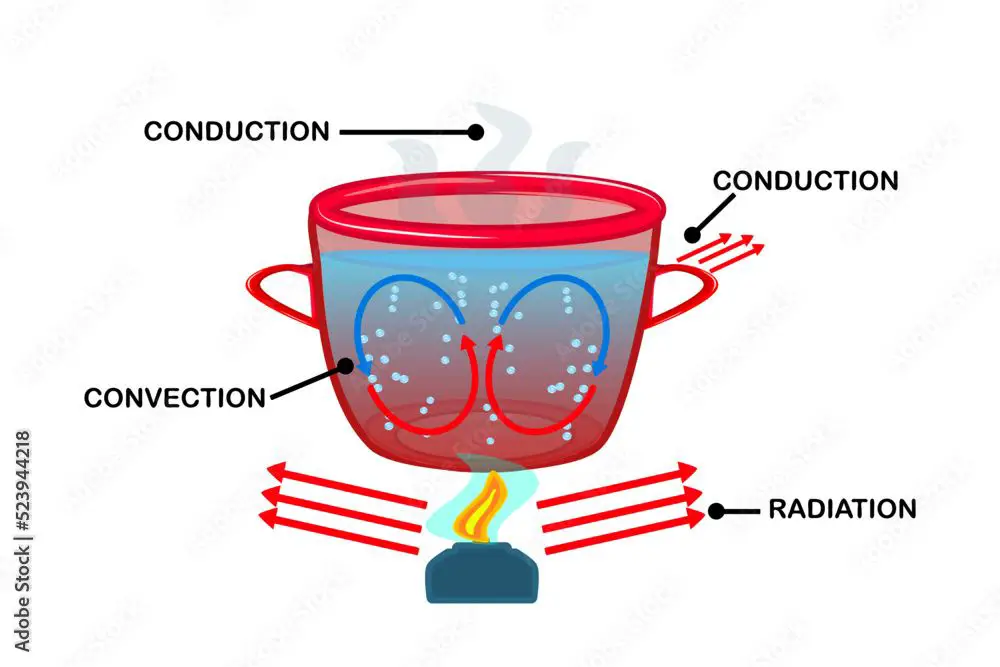
Examples of convection include hot air rising from a radiator, the movement of magma in the Earth’s mantle, and ocean currents circulating heat around the globe.
Radiation
Radiation is the transfer of heat not requiring a medium to be present. Heat in the form of infrared radiation is emitted from a hot surface and absorbed by a cooler surface. No direct contact between the heat source and destination is needed.
Examples of radiative heat transfer include the warming of the Earth by the sun, an incandescent light bulb heating a room, and heat radiating off a fire into the surrounding air.
Heat and Temperature
Heat and temperature are often used interchangeably in everyday language, but they are distinct scientific concepts. The main difference between heat and temperature is that heat is a form of energy transfer while temperature is a measure of the average kinetic energy of molecules and atoms.
Temperature measures how hot or cold an object is. It refers to the average kinetic energy of atoms and molecules in a substance. As the kinetic energy increases, atoms and molecules move faster and spread apart, leading to an increase in temperature. Temperature is measured in scales like Fahrenheit, Celsius and Kelvin.
On the other hand, heat is the transfer of thermal energy between substances due to a temperature difference. It flows spontaneously from a hotter to a colder body. Heat is a form of energy, while temperature just provides a measure of the average energy in a system. Heat transfer changes the internal energy of both systems involved according to the laws of thermodynamics.
While temperature provides a measure of the average kinetic energy of particles, heat refers to the total energy transferred between systems due to differences in temperature. Understanding the distinction between heat and temperature is crucial in the study of thermodynamics and heat transfer.
Heat Transfer Equation
The transfer of heat between two bodies can be described mathematically using heat transfer equations based on the laws of thermodynamics. Two important heat transfer equations are Fourier’s Law of Heat Conduction and Newton’s Law of Cooling.
Fourier’s Law of Heat Conduction
This law describes the process of conductive heat transfer through a solid material. It relates the heat flux through the material to the temperature gradient and the thermal conductivity of the material.
The equation is:
q = -k(dT/dx)
Where q is the heat flux (amount of heat transferred per unit area per unit time), k is the thermal conductivity, dT is the temperature difference across the material, and dx is the material thickness.
Newton’s Law of Cooling
This law describes convective heat transfer between a body and its surrounding fluid. It relates the rate of heat loss from the body to the temperature difference between the body and the fluid.
The equation is:
q = hA(Tb – T¥)
Where q is the heat transfer rate, h is the convective heat transfer coefficient, A is the surface area of the body, Tb is the body temperature, and T¥ is the fluid temperature.
These fundamental equations allow mathematical modeling of heat transfer processes and are widely used in physics, engineering, and industry.
Applications of Heat
Heat has many important applications in our everyday lives and in industry. Some of the main applications of heat include:
Heating and Cooling
Heat is used to provide heating and cooling in buildings and vehicles. Heating systems like furnaces, boilers, and heat pumps rely on heat transfer to provide warmth during cold weather. Air conditioners and refrigerators transfer heat out of indoor spaces to provide cooling.
Industrial Processes
Many industrial processes require the application of heat to refine, cure, or transform materials. Heat is used in smelting metals, annealing, drying materials, chemical reactions, and polymer production. Controlled heating allows materials to be carefully processed on a large scale.
Cooking
The cooking process relies on heat to transform ingredients into food. Methods like baking, frying, searing, and boiling all use various types of heat transfer to cook food. Cooking allows the flavors and textures of ingredients to be altered in desirable ways through the application of heat.
Heat and Thermodynamics
Heat is closely related to the laws of thermodynamics, which describe how thermal energy interacts with matter. The First Law states that energy can neither be created nor destroyed in an isolated system. The Second Law states that heat flows spontaneously from hotter to colder objects until they reach the same temperature.
These thermodynamic principles allow heat engines and heat pumps to operate. Heat engines convert thermal energy into mechanical work. For example, car engines burn fuel to generate heat which pushes the pistons. Heat pumps use work to move heat from a colder location to a hotter one, as in a refrigerator. They operate by compressing and expanding a working fluid like refrigerant.
In both engines and pumps, the goal is to maximize efficiency. This is done by minimizing wasted heat and entropy production. Understanding thermodynamics reveals optimal ways to transfer and utilize heat energy.
Conclusion
In summary, heat is a form of energy that can be transferred between objects or systems due to temperature differences. The three main mechanisms of heat transfer are conduction, convection and radiation. Understanding how heat transfers is crucial for many scientific and engineering applications, from designing heat sinks for electronics, to predicting weather patterns, to developing more efficient engines and energy systems. Properly accounting for heat transfer mechanisms allows us to control temperatures for comfort and safety, utilize heat for energy generation, and gain insights into the nature of thermodynamic processes.
The study of heat and thermodynamics has enabled tremendous technological and industrial progress over the last few centuries. From steam engines to internal combustion engines to rocket engines, our civilization has harnessed heat as a resource to overcome limitations of muscle power. Looking forward, insights into nanoscale heat transfer may enable future advances in computing and electronics. By continuing to deepen our knowledge of this ubiquitous form of energy, we can further improve technologies and engineering practices to build a more sustainable future.