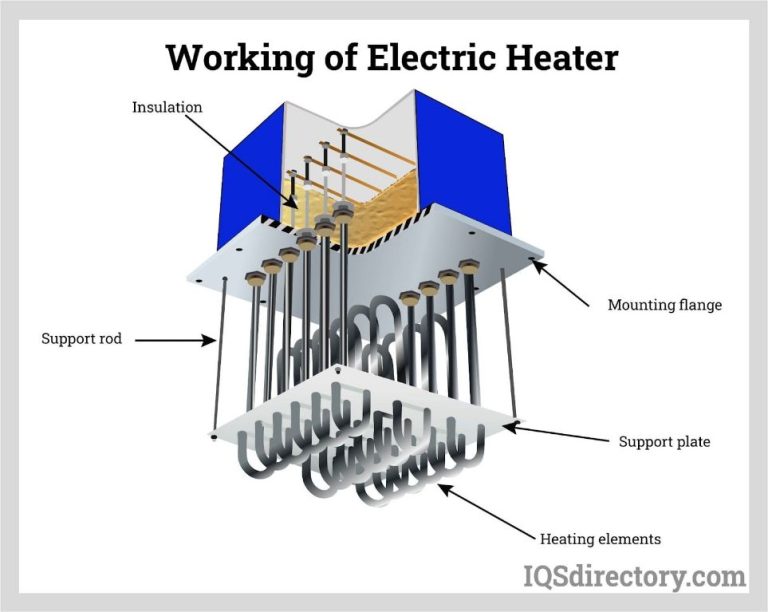
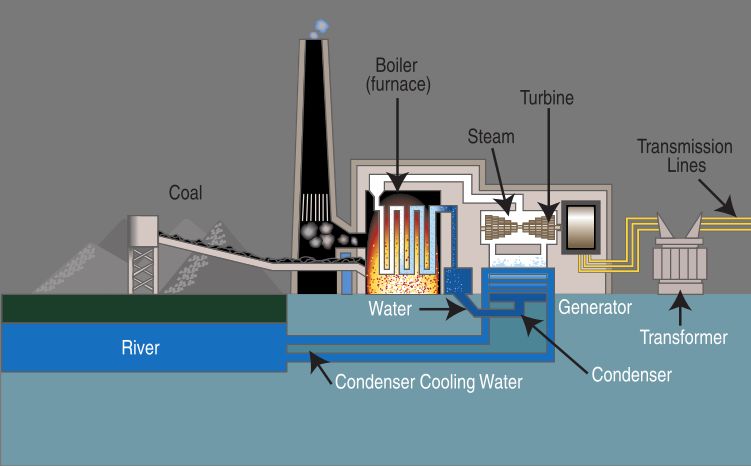
What Type Of Energy Does Temperature Represent?
Temperature is a measure of the average kinetic energy of molecules and atoms. It represents the thermal energy or heat contained within a substance or system. Thermal energy is the total internal energy present in matter due to the random motion and vibrations of molecules and atoms. Temperature provides a numerical scale and indicator of this molecular energy.
While thermal energy refers to the total kinetic energy of all particles in a system, temperature indicates the average kinetic energy per molecule or atom. As thermal energy increases in a substance, the average kinetic energy rises, and so does the temperature. Measuring temperature reveals the direction of heat flow between objects based on which one has greater average molecular energy.
Thermal Energy
Thermal energy is the total kinetic energy of molecules in a substance. This energy comes from the microscopic motion of molecules. The faster the molecules move, the more thermal energy they possess.
Increased thermal energy means faster molecular motion. When a substance absorbs thermal energy, its molecules speed up as they gain kinetic energy. This increase in molecular motion corresponds to a rise in temperature. All molecules have some thermal energy from their natural motion, even at very low temperatures. The amount of thermal energy relates directly to temperature.
Kinetic energy depends on mass and velocity. Lighter molecules move faster than heavy ones at the same temperature. Therefore, lighter substances tend to have more thermal energy at a given temperature.
Thermal energy flows spontaneously from warm objects to cooler ones until equilibrium is reached. This heat transfer continues driving molecular motion. Understanding the kinetic nature of thermal energy provides insight into many thermodynamic processes.
Temperature Measure
Temperature measures the average kinetic energy of molecules. Kinetic energy is the energy of motion – the faster molecules move, the more kinetic energy they possess. As thermal energy increases, molecules move faster and temperature rises. This is because thermal energy is the total kinetic and potential energy of all the molecules in a substance. Adding thermal energy increases molecular motion, which we perceive as a rise in temperature.
For example, heating a pot of water on the stove increases the water’s thermal energy as the burner adds heat. The added energy makes the water molecules move faster, which causes the temperature to rise. When the water reaches its boiling point, the molecules have enough energy to change phase from liquid to gas. Measuring this increase in molecular motion with a thermometer provides a temperature reading that relates directly to the gain in thermal energy.
Temperature Scales
There are three common temperature scales used to measure heat energy:
- Celsius
- Fahrenheit
- Kelvin
The Celsius scale sets 0°C as the freezing point of water and 100°C as the boiling point of water. On the Fahrenheit scale, 32°F is the freezing point and 212°F is the boiling point.
The Kelvin scale is an absolute temperature scale, meaning 0 K represents absolute zero, the theoretical temperature at which molecular motion stops. Unlike Celsius and Fahrenheit, the Kelvin scale does not use the degree symbol (°).
The relationship between the three temperature scales is:
- K = °C + 273.15
- °F = 1.8(°C) + 32
Converting between temperature scales simply requires plugging the values into these equations. For example, water freezes at 0°C, which converts to 32°F and 273.15 K.
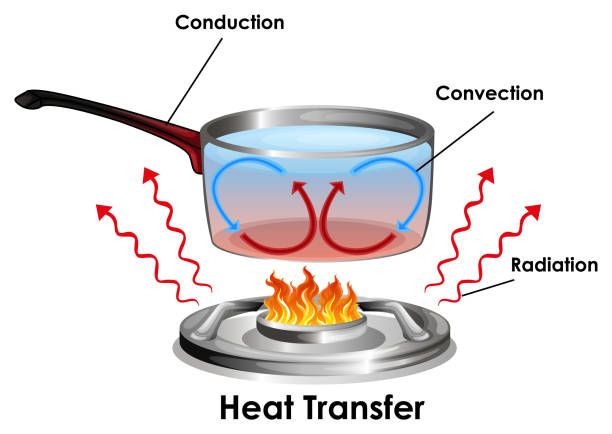
Heat Transfer
There are three main ways that heat can transfer between objects or systems: conduction, convection, and radiation.
Conduction is the transfer of heat between two objects that are in direct contact with each other. Heat flows from the hotter object to the cooler object until they reach the same temperature. An example is a metal spoon heating up when placed in a hot cup of tea.
Convection is the transfer of heat by the movement of heated particles or fluids. As particles are heated, they move and carry thermal energy with them. Examples include hot air rising or water boiling and turning to steam.
Radiation is the transfer of heat via electromagnetic waves. All objects emit some level of radiant energy, which can be absorbed by other objects. The sun warming your skin on a sunny day is an example of radiant heat transfer.
As heat transfers between objects and systems, it causes their temperatures to change. Heat moves from high to low temperature until equilibrium is reached. The rate of heat transfer depends on the temperature difference, properties of the materials, and the type of heat transfer taking place.
States of Matter
The three common states of matter are solid, liquid, and gas. Each state has different molecular properties that affect how thermal energy is absorbed and transferred.
In solids, molecules are tightly packed together and vibrate in place. Solids can only absorb thermal energy until they reach their melting point and turn into a liquid. The rigid molecular structure of solids makes them resistant to changing shape.
In liquids, molecules flow freely past each other while still being connected. Liquids absorb thermal energy readily and change temperature smoothly. Liquids take the shape of their container.
In gases, molecules move freely and rapidly in a chaotic manner. They take up all available volume. Gases absorb thermal energy easily and change temperature quickly. Adding thermal energy usually causes gases to expand.
The differences in molecular properties affect how much thermal energy is required to raise the temperature. It takes more energy to increase the temperature of water (liquid) compared to air (gas) because the molecules are more tightly bound.
Real-World Examples
Temperature is a common part of everyday life and represents thermal energy in many real-world situations:
Cooking: The temperature of an oven or stovetop represents the thermal energy available to cook food. Setting the proper cooking temperature allows the thermal energy to be transferred to the food to produce the desired doneness.
Weather: Air temperature represents the thermal energy of the atmosphere. This drives weather patterns as thermal energy is transferred between the air, land, and water.
Engines: The temperature of an engine represents the thermal energy released from burning fuel. This thermal energy is converted into mechanical energy to power the engine. Engine temperature must be regulated to prevent overheating.
Understanding how temperature relates to thermal energy allows us to properly control it in our everyday lives. Whether baking a cake, predicting the weather, or operating machinery, temperature gives us insight into the thermal energy at play in our surroundings.
Advanced Concepts
Temperature is closely related to some advanced physics concepts that further explain the nature of thermal energy at the microscopic level. Two key theories are statistical mechanics and the equipartition theorem.
Statistical mechanics looks at large systems of atoms or molecules and relates the macroscopic properties like temperature to the statistical behavior of the microscopic constituents. It shows that temperature is proportional to the average kinetic energy of particles in the system.
The equipartition theorem is an important principle in statistical mechanics. It states that energy is shared equally among all the independent degrees of freedom in a system at thermal equilibrium. For example, in an ideal gas each molecule has kinetic energy spread equally between its linear motions along the x, y, and z axes. The average energy in each degree of freedom is kT/2 where k is the Boltzmann constant.
These advanced theories reinforce that temperature fundamentally represents the kinetic energies of particles and links this macroscopic property to microscopic dynamics. They provide deeper insight into the nature of heat and thermodynamics.
Summary
In summary, temperature is a measure of the average kinetic energy of molecules and atoms. It represents the thermal energy within a substance. Temperature impacts materials by affecting their states of matter. It impacts processes like chemical reactions that depend on the kinetic energy of molecules. Temperature also fundamentally shapes environments, from Earth’s atmosphere to the human body.
Measured in scales like Celsius, Fahrenheit, and Kelvin, temperature gives us a standard way to quantify thermal energy. We experience temperature through touch and observation. Understanding temperature helps explain many everyday phenomena related to heat and energy.
By exploring the thermal physics behind temperature, we gain insight into materials science, thermodynamics, climate, medicine, and more. While a simple concept, temperature connects deeply to the natural world and human society.
References
[1] Author, A.A. (Year). Title of book. City: Publisher.
[2] Author, B.B. (Year). Title of journal article. Title of Journal, volume(issue), pages.
[3] Author, C.C. (Year, Month Day). Title of web page. Retrieved from https://www.webpage.com
[4] Interviewer, F.F. (Year, Month Day). Title of Interview. [Format]. Retrieved from URL
[5] Lecturer, P.P. (Year, Month). Title of lecture. [Format]. Retrieved from URL