Does Thermal Energy Mean Hot Or Cold?
Introducing Thermal Energy
Thermal energy refers to the total kinetic and potential energy associated with the random motions of atoms and molecules in a substance. It is often used interchangeably with the term “heat” in everyday language. However, thermal energy and heat are scientifically distinct concepts.
Thermal energy is measured in joules and represents the total internal energy within a system. Heat, on the other hand, refers specifically to the transfer of thermal energy between objects or systems due to temperature differences. Heat flows spontaneously from higher temperature to lower temperature objects and is measured in joules.
While thermal energy describes the total kinetic energy of particles, temperature measures the average kinetic energy of particles in a substance. Temperature determines the direction of heat transfer, but does not quantify the amount of thermal energy present like internal energy does. An object may contain a lot of internal thermal energy, but have a low temperature if the energy is spread among many particles.
In summary, thermal energy refers to the total kinetic and potential energy of particles in a substance, while heat deals with thermal energy transfer between objects due to temperature differences. Both concepts are important in thermodynamics.
Hot Thermal Energy
Thermal energy refers to the total kinetic energy and potential energy of atoms and molecules in a substance. This energy is associated with the motions and relative positions of the particles that make up a material. Hot thermal energy involves higher temperatures, where the atoms and molecules have more kinetic energy and vibrate rapidly.
Some examples of high temperature thermal energy include:
- Fire – The chemical reaction of combustion releases a substantial amount of thermal energy in the form of heat and light.
- Steam engines – Fossil fuels like coal are burned to heat water into pressurized steam that drives mechanical motion.
- Nuclear reactions – Fission and fusion release tremendous amounts of thermal energy from the breaking and forming of atomic bonds.
- Friction heating – Kinetic energy is converted to thermal energy through friction, like rubbing hands together.
- Electric heaters – Electrical current flowing through resistors generates thermal energy that heats up the filaments.
- Gas burners – The combustion of natural gas or propane releases hot gaseous thermal energy that is used for cooking.
In all these high-temperature examples, thermal energy is being actively generated and transferred to other objects. The increased molecular vibrations correspond to more internal energy in the system.
Cold Thermal Energy
Cold thermal energy refers to thermal energy associated with low temperatures. The most common examples of cold thermal energy that we encounter in everyday life are ice and refrigerators/freezers.
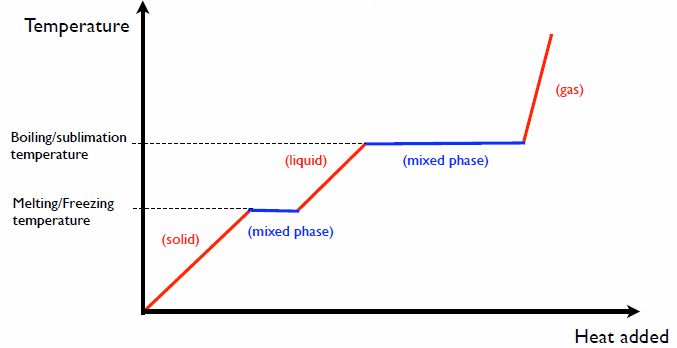
Ice is a classic example of matter that contains cold thermal energy. The molecules in ice vibrate more slowly and have less kinetic energy than liquid water. This makes ice feel cold to the touch. In fact, ice has a temperature of 0°C or 32°F by definition. As heat is added to ice, some of the thermal energy goes into speeding up molecular vibrations, causing the ice to melt into liquid water. This absorption of heat is why ice can be used for cooling purposes.
Refrigerators and freezers are devices designed to produce cold temperatures inside by removing thermal energy. They have a refrigeration system that uses the evaporation of refrigerant liquids to absorb heat from the interior. This keeps the inside at cold temperatures suitable for preserving foods and drinks. The typical refrigerator temperature is around 3-4°C (37-39°F) while freezers are usually around -18°C (0°F). The cold thermal energy inside refrigerators and freezers comes from actively pumping heat out of the confined space.
Other examples of cold thermal energy include cold drinks, ice packs, cryogenics, dry ice, and air conditioning. Anything that feels cold to the touch contains lesser amounts of thermal energy than our bodies do.
Measuring Thermal Energy
Thermal energy can be precisely measured using various units. The most common units are:
- Joules – The joule is the SI unit for energy. It equates to the amount of work done by a force of one newton when its point of application moves one meter in the direction of action of the force.
- Calories – A calorie is the amount of energy required to raise one gram of water by one degree Celsius at standard atmospheric pressure. This is approximately 4.184 joules.
- BTUs – The British Thermal Unit (BTU) originated in the early nineteenth century. It is defined as the amount of heat required to raise the temperature of one pound of liquid water by one degree Fahrenheit at a constant atmospheric pressure.
There are various instruments that can precisely quantify thermal energy in these units, such as calorimeters. Thermometers and other temperature sensors are also useful for indirect measurements of thermal energy based on temperature change.
Thermal Energy Transfer
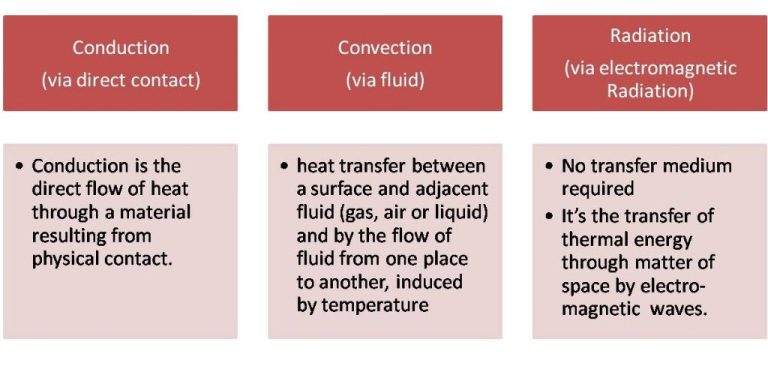
Thermal energy can be transferred between objects or locations through three main mechanisms: conduction, convection, and radiation.
Conduction is the transfer of thermal energy between objects that are in direct contact with each other. It occurs when atoms and molecules with higher kinetic energy collide with and transfer some of their energy to neighboring atoms and molecules with lower kinetic energy. Metals are good conductors of thermal energy.
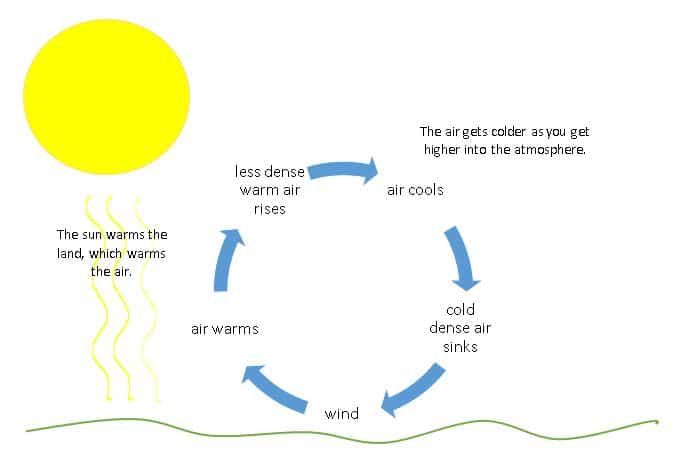
Convection is the transfer of thermal energy by the movement of heated matter. It occurs in liquids and gases, where hot, less dense material rises, and colder, denser material sinks. This movement distributes the thermal energy. Examples include heating water on a stove or the motion of air in the atmosphere.
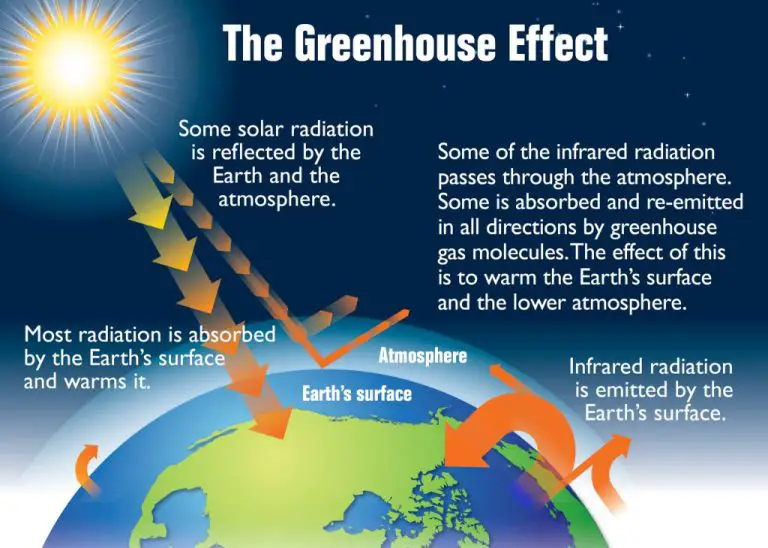
Radiation is the transfer of thermal energy by electromagnetic waves. All objects emit infrared radiation related to their temperature. Thermal energy is transferred when these infrared rays are absorbed by another object. Radiation does not require direct contact between objects and can even occur in a vacuum, such as between the Earth and the Sun.
Understanding thermal energy transfer mechanisms allows us to engineer more efficient heating and cooling systems and devices.
Thermal Energy in Physics
Thermal energy has important applications in the field of physics, especially in relation to thermodynamics and heat transfer laws. The study of thermodynamics looks at relationships between heat and other forms of energy. It describes how thermal energy is transferred between objects or systems due to temperature differences. The laws of thermodynamics set limits on how efficiently heat can be converted into useful work. Thermal physics also deals with the kinetic theory, which explains heat as the kinetic energy of atoms and molecules in matter. The greater the thermal energy, the faster the particles move.
Several key laws relate to thermal energy in physics. The Zeroth Law of Thermodynamics states that if two objects are in thermal equilibrium with a third object, they are also in thermal equilibrium with each other. The First Law states that energy is conserved in a closed system. The Second Law describes how entropy, a measure of randomness or disorder, always increases in an isolated system. The Third Law says that entropy approaches a minimum value as temperature approaches absolute zero.
In addition to the laws of thermodynamics, physics has defined equations that describe the rate of heat transfer between objects or systems. These include Fourier’s Law which relates heat flux to temperature gradient, Newton’s Law of Cooling which describes convective heat transfer, and the Stefan-Boltzmann Law which deals with thermal radiation. The transfer of thermal energy is central to fields like thermodynamics, statistical mechanics, and quantum mechanics.
Thermal Energy in Chemistry
Thermal energy plays an important role in chemistry, particularly in understanding endothermic and exothermic reactions. An endothermic reaction absorbs heat energy from the surroundings, resulting in a decrease in thermal energy. In contrast, an exothermic reaction releases heat energy to the surroundings, resulting in an increase in thermal energy.
The transfer of thermal energy in chemical reactions follows Hess’s law. This law states that the total enthalpy change in a chemical reaction is the same whether the reaction takes place in one step or many steps. Enthalpy, symbolized as H, is a measure of the total energy content of a system including its internal energy and pressure-volume work. The enthalpy change (ΔH) in a reaction is directly related to the amount of thermal energy absorbed or released.
By applying Hess’s law, chemists can determine the enthalpy change for a reaction by breaking it down into component steps of known enthalpy changes and adding them together. For example, the enthalpy change for a reaction A + B → C can be determined by:
ΔHreaction = ΔH1 + ΔH2 + ΔH3
Where ΔH1, ΔH2, and ΔH3 are known enthalpy changes for intermediate steps in the reaction.
Measuring and calculating enthalpy changes for chemical reactions provides insights into how much heat is involved in the breaking and formation of chemical bonds during the reaction. This allows chemists to categorize reactions as endothermic or exothermic and determine how much thermal energy they absorb or release to better control reactions.
Thermal Energy in Biology
Thermal energy plays a critical role in biology and living organisms. At the cellular level, thermal energy drives metabolic processes and chemical reactions that are essential for life.
Thermoregulation, or maintaining homeostasis of body temperature, is another key function related to thermal energy. Endotherms like mammals and birds use various methods to control their internal body temperature against variations in external temperature. For example, mechanisms like shivering, sweating, and panting either produce or dissipate heat to keep the body within a narrow temperature range.
Ectotherms like reptiles, amphibians, and other cold-blooded animals have adapted strategies to regulate their body temperature through behavioral means, like basking in the sun or cooling off in the shade or water. Their metabolic rates are strongly influenced by external thermal conditions.
At the ecosystem level, thermal energy affects habitats and natural selection. Adaptations like insulation, camouflage coloration, and migration patterns allow various organisms to thrive in diverse thermal environments.
Overall, thermal energy is a critical driver of metabolism, homeostasis, and adaptations in living organisms across scales from cells to ecosystems.
Applications of Thermal Energy
Thermal energy has many important real-world applications in power generation, heating and cooling systems, and industrial processes.
In power generation, thermal energy can be used to produce electricity. Power plants often burn fossil fuels such as coal, oil or natural gas to heat water and produce steam. This steam then spins turbines which drive electric generators, converting the thermal energy into electrical energy.
Thermal energy is also essential for heating and cooling systems. Furnaces, boilers, and heaters rely on the combustion of fuels or electricity to provide heat. Air conditioners, refrigerators, and heat pumps use principles like compression and expansion of gases to transfer thermal energy and provide cooling.
In industry, thermal energy plays a vital role in smelting metals, refining petroleum, manufacturing ceramics and glass, and many other processes. These applications require carefully controlled high temperatures provided by the burning of fuels, electricity, or focused sunlight.
Overall, thermal energy powers many critical systems that provide electricity, heating, cooling, and enable industrial production. Understanding thermal physics allows us to harness this form of energy for human needs.
Conclusion
Thermal energy refers to the total kinetic energy of molecules within an object. All objects contain thermal energy, whether hot or cold. Thermal energy is directly related to temperature, which is a measure of the average kinetic energy of molecules. While we may colloquially refer to “heat” as a form of thermal energy, technically both hot and cold objects contain thermal energy in the form of molecular motion.
In summary, thermal energy exists on a spectrum from low to high levels, corresponding to cold and hot temperatures. But the key point is that all objects, regardless of temperature, contain thermal energy in the form of kinetic energy at the molecular level. So next time you touch a cold object, remember that it still contains thermal energy, just less than a hot object. Thermal energy is an essential concept in physics, chemistry, biology and engineering, underlying processes from chemical reactions to biological systems to the operation of heat engines and power plants.