What Happens When You Heat Energy?
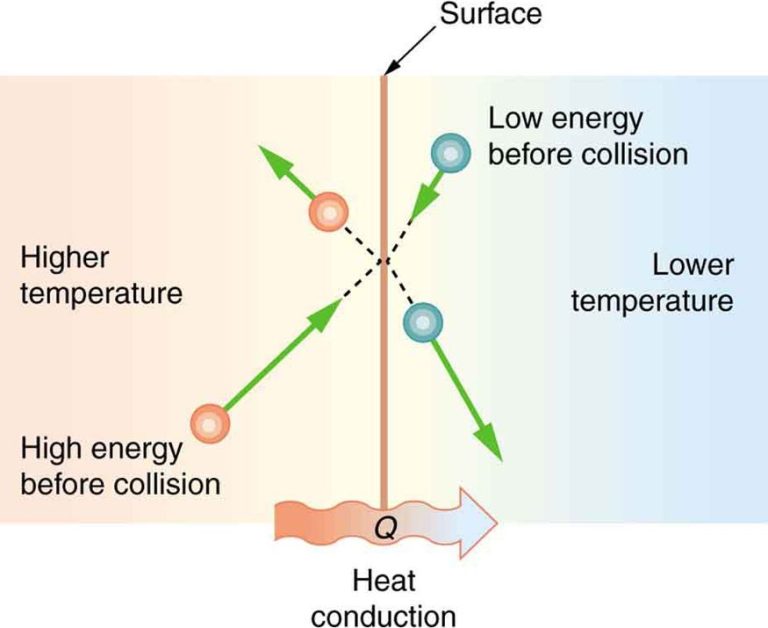
Heat is energy that is transferred from one object or system to another as a result of temperature difference. Heat flows naturally from a hotter object to a cooler one until thermal equilibrium is reached, meaning both objects reach the same temperature.
Energy comes in many forms like electrical, chemical, nuclear, mechanical, and thermal energy. Thermal energy refers specifically to the total kinetic energy of molecules within an object. This kinetic energy is directly associated with the temperature of the object.
When two objects at different temperatures come into contact, heat flows from the hotter object to the cooler one, increasing the temperature of the cooler object while decreasing the temperature of the hotter object. This heat transfer continues until the two objects reach the same temperature.
Heat Transfer Methods
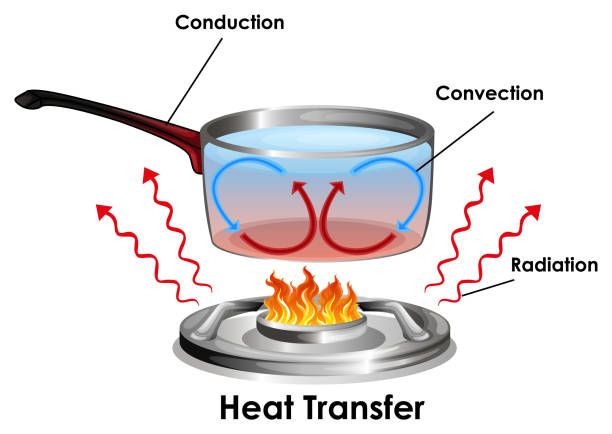
Heating matter fundamentally involves the transfer of energy in the form of heat. There are three main methods by which heat transfers from one object or system to another: conduction, convection, and radiation.
Conduction occurs when two objects come into direct physical contact with each other. Heat energy transfers from the warmer object to the cooler one until they reach thermal equilibrium. An example is a spoon getting hot after being left in a cup of hot coffee. The metal spoon conducts heat from the hot liquid it is touching.
Convection occurs when heat transfers through fluids, either liquids or gases. As the fluid is heated, it expands, becomes less dense, and rises. Cooler, denser fluid then moves in to take its place, creating a continuous circulation. This moving fluid transfers heat energy from one place to another. Examples include heating water on a stove or hot air rising from a radiator.
Radiation does not rely on direct contact or fluid circulation. Instead, infrared electromagnetic waves carry heat energy directly across space. The warmth from a fireplace reaching across a room is an example of radiant heat transfer. The sun also heats the Earth predominantly through radiation.
Effects of Heating Matter
When matter is heated, it undergoes several changes at the molecular level. Here are some of the key effects of heating matter:
Thermal Expansion
As matter is heated, the molecules gain kinetic energy and start moving and vibrating more. This causes the material to expand in size, which is known as thermal expansion. Solids typically expand in all directions when heated. Liquids also expand when heated, but only negligibly compared to solids.
Change of State
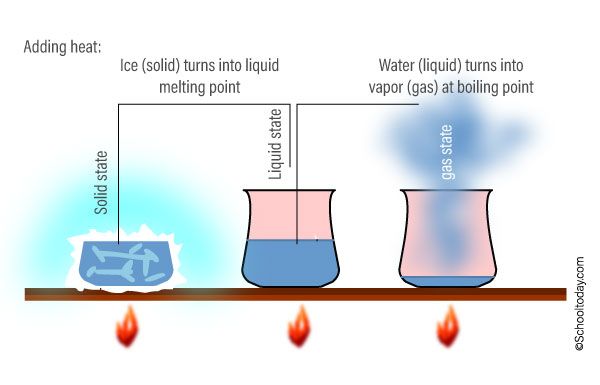
Heating can cause phase changes or changes of state in matter. For example, heating a solid causes the molecules to gain enough kinetic energy to break free from their fixed positions and start moving freely, melting the solid into a liquid. Further heating of the liquid provides enough energy for molecules to transition to a gaseous state, boiling the liquid into a gas. These transitions depend on the material’s melting and boiling points.
Chemical Changes
Extreme heating can break apart molecular bonds completely and cause chemical changes. This can result in chemical decomposition, where a compound breaks down into simpler substances. For example, heating wood causes it to undergo thermal decomposition and break down into charcoal, carbon dioxide, carbon monoxide, and other byproducts. High heat can also cause oxidation or burning in the presence of oxygen. So heating matter provides energy for chemical reactions that may not occur at lower temperatures.
Everyday Examples
Heating is a part of our everyday lives in many ways that we often take for granted. Here are some of the most common examples of how we use heating on a regular basis:
Cooking food – Applying heat is essential for cooking most foods. The different cooking methods we use, such as baking, frying, boiling etc., all rely on heating to transform ingredients into tasty meals.
Heating homes – Most modern heating systems for homes use the combustion of natural gas or heating oil to generate heat. This heat warms up air or water which is then pumped throughout the home via radiators, vents or underfloor heating.
Car engines – The fuel in car engines is ignited through heat generated by compressing the fuel-air mixture. This small explosion pushes the pistons and enables the engine to run.
Heating Water
When water is heated, it begins to vibrate more rapidly. As the temperature increases, the water molecules gain enough energy to overcome the attractive forces binding them together and transform from a liquid to a gas in the process known as vaporization. This conversion of water from a liquid to a vapor is called boiling.
Before reaching its boiling point, water forms convection currents as it is heated. The hot water, which is less dense than the surrounding cooler water, rises to the top while the cooler, denser water sinks. This movement and circulation distributes the heat more evenly throughout the water.
At sea level, water boils at 212°F (100°C). As the water reaches this temperature, bubbles begin to form at nucleation sites on the bottom and sides of the container as the water transforms to steam. When the bubbles reach the surface, they burst and release the steam into the air. The vigorous motion and turbulence during boiling is caused by the high energy state of the water molecules.
Understanding how water behaves when heated is useful for many cooking applications and industrial processes. The convection currents distribute the heat for even cooking, while the steam created from boiling water can be harnessed to power mechanical engines.
Heating Air
When air is heated, it expands and becomes less dense than the surrounding cooler air. This causes the warm air to rise, while the cooler air sinks to take its place. This movement of air due to differences in density is known as convection.
As the warm air rises, it transfers heat to the surrounding air, cooling as it rises. The surrounding cooler air is heated by this transfer of heat, causing it to become less dense and rise as well. This cycle of rising warm air and sinking cool air creates convection currents.
Convection currents play an important role in everyday heating processes and weather patterns. For example, they are responsible for the updrafts that enable birds and gliders to soar, and storms to develop. Convection currents also circulate air in homes, distributing warm air from heaters or fireplaces.
Understanding how heating air leads to convection currents helps explain many natural phenomena related to heat transfer and fluid dynamics.
Heating Solids
When a solid material is heated, the molecules gain kinetic energy and start vibrating faster. As the vibrations increase, the solid expands in size. This effect is called thermal expansion, and most materials expand to some degree when heated. Metals typically expand the most compared to other solid materials.
If enough heat is applied, the molecular vibrations become so intense that the solid’s rigid structure breaks down. At this point, the solid begins to melt and turns into a liquid. Each substance has a specific melting point temperature where this occurs. For example, water ice melts at 0°C (32°F), while iron melts at 1538°C (2800°F).
With even more heating, a solid can reach its ignition temperature and catch fire. This is an exothermic chemical reaction that releases heat, light, and various gases. The solid fuel combines with oxygen and is chemically transformed into new combustion products. For example, wood ignites at 300-400°C (570-750°F) and burns into charcoal ash and gases.
So in summary, heating a solid material progressively causes thermal expansion, melting, and then possibly burning. The intensity of heating determines which effect dominates and the physical state of the resulting material.
Heating Gases
Gases react distinctly when heated due to their molecular composition. While solids and liquids are made of molecules packed tightly together, gases have large spaces between their molecules. When you apply heat to a gas, the added energy causes the gas molecules to speed up and move farther apart from each other. This expansion makes the gas less dense overall, enabling it to rise and become lighter than surrounding cooler air.
For example, when you boil water the added heat energy causes the liquid water molecules to transition to a gaseous state as water vapor or steam. The water vapor expands to occupy more space, becoming less dense than the surrounding air. This causes the steam to rise rapidly from the heated liquid surface as the hot gas is more buoyant in the cooler air.
Heating air can also decrease its density and make it rise. Hot air balloons capitalize on this principle to create lift and float. The propane burner heats the air inside the balloon, causing it to expand. The hot, less dense air inside has greater buoyancy compared to the cooler air outside the balloon. This discrepancy in density produces enough lift for the balloon to rise high off the ground.
Understanding how heating affects gases aids in designing heating and cooling systems, predicting weather patterns, and leveraging the unique properties of gases across many industrial and commercial applications.
Physics Principles
When heat is transferred to matter, several physics principles and laws come into play. The most fundamental are the laws of thermodynamics, which describe the relationships between thermal energy, work, and heat. The first law of thermodynamics states that energy is conserved – the total energy of a closed system remains constant. When heat is added to a system, some of it goes into changing the internal energy of the system while the rest is used to do work.
The second law of thermodynamics deals with entropy, which is a measure of disorder within a system. It states that the entropy of an isolated system always increases in any spontaneous process. When heat is applied to matter, it increases the motion and vibrations of molecules and atoms, increasing entropy.
There are several equations in thermodynamics that describe heat transfer and the relationships between different thermodynamic properties. Some key ones are:
- The heat transfer rate equation calculates conductive heat transfer based on temperature difference, thermal conductivity, and other factors.
- The heat capacity equation relates the amount of heat transfer to the temperature change in matter.
- The heat of fusion and heat of vaporization equations describe the amount of heat needed to change phase from solid to liquid and liquid to gas, respectively.
Understanding these physics principles allows scientists and engineers to predict and calculate what will happen when heat is applied to any type of matter.
Conclusion
In summary, heating energy involves the transfer of heat between objects or systems. This transfer occurs through conduction, convection or radiation. Heating matter causes changes in temperature, physical state, volume, and other properties. Understanding how heat interacts with water, air, solids, and gases has many real-world applications. Cooking food, powering engines, and predicting weather all rely on principles of heating energy. Although a common phenomenon, heating energy exhibits complex physics behavior. This brief overview provided key highlights, but many details remain to fully explain heat transfer across scientific disciplines. Appreciating heat dynamics helps us harness energy safely and efficiently for human needs. Whether heating your home or exploring the farthest reaches of the universe, the principles outlined here advance our comprehension of thermal energy.