What Is The Energy To Do Work Called?
Work and energy are two fundamental concepts in physics that are closely related. Work refers to the application of a force over a distance, while energy is the capacity to do work. The goal of this article is to clearly define what work and energy are, and to explain the relationship between them.
Understanding the connection between work and energy is crucial, as these ideas are applied in many areas of physics and engineering. We will build up an intuition for work and energy through definitions, examples, and descriptions of their relationship. With a solid grasp of these foundational principles, we can apply work and energy concepts to topics like motion, thermodynamics, and more.
Work
Work is defined as the amount of force exerted on an object multiplied by the displacement of the object in the direction of the force. It measures the force applied to an object and the object’s movement in response to the force. Mathematically, work is calculated as:
Work = Force x Displacement
Some examples of work include:
- Lifting a heavy box from the ground. The force exerted to lift the box upward, multiplied by the height the box is lifted, equals the work done.
- Pushing a cart across a floor. The force exerted to push the cart, multiplied by the distance the cart moves, is the work.
- Stretching a spring. The force applied to stretch the spring, multiplied by the distance the spring stretches, is the work.
The SI unit for work is the joule (J). One joule of work is done when a force of one newton (N) acts on an object to move it one meter (m) in the direction of the force. Work can also be measured in foot-pounds (ft-lb) in the imperial system. One foot-pound is the amount of work done by a one pound force acting on an object to move it one foot.
Energy
Energy is the ability to do work. There are different forms of energy:
- Kinetic energy – The energy an object possesses due to its motion. The kinetic energy of an object depends on its mass and velocity. For example, a moving car has kinetic energy.
- Potential energy – The stored energy an object has due to its position or shape. For example, a rock held above the ground has gravitational potential energy that can be converted to kinetic energy if it falls.
- Chemical energy – The energy stored in the bonds between atoms and molecules. Food contains chemical energy that is released when digested.
- Thermal energy – The kinetic energy associated with the random motion of atoms and molecules in matter. The more a material’s atoms vibrate, the higher its thermal energy and temperature.
- Electrical energy – The energy carried by moving electrons. Electricity contains electrical energy that can power devices.
- Nuclear energy – The energy stored in the nucleus of an atom, released in nuclear reactions. Nuclear power plants convert nuclear energy into electrical energy.
The standard unit of energy is the joule (J). Other units include calories, kilowatt-hours, and British thermal units. Energy can be converted between different forms, but the total energy in a closed system remains fixed (conservation of energy).
Kinetic Energy
Kinetic energy is the energy of motion. It refers to the work needed to accelerate an object to a certain velocity. Kinetic energy depends directly on the object’s mass and velocity.
The more massive an object is and the faster it moves, the more kinetic energy it has. For example, a large truck moving at high speed has a huge amount of kinetic energy. Conversely, a tiny marble moving slowly has very little kinetic energy.
Kinetic energy is calculated using the following formula:
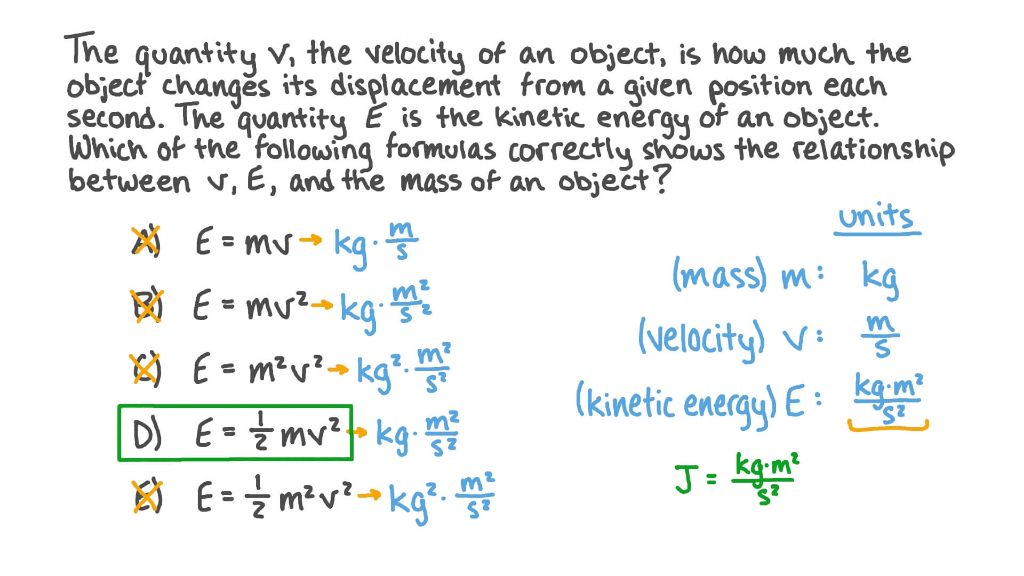
Kinetic Energy = 1/2 x mass x velocity2
The SI unit for kinetic energy is the joule (J). Other units include ergs, electron volts, foot-pounds, and British thermal units.
Kinetic energy comes from the work done to accelerate the object. Any means of getting an object to speed up, such as pushing it, letting gravity pull it, or applying a force via combustion, will impart kinetic energy to the object.
Potential Energy
Potential energy is the stored energy an object has due to its position or state. For example, a ball at the top of a ramp has potential energy due to gravity. As the ball rolls down the ramp, this potential energy gets converted to kinetic energy. There are several forms of potential energy:
- Gravitational potential energy – energy due to an object’s height above the ground in a gravitational field
- Elastic potential energy – energy stored in stretched or compressed springs
- Chemical potential energy – energy stored in the bonds between atoms and molecules
Potential energy is measured in joules in the SI system. The potential energy an object has depends on its mass, height, and the strength of the gravitational field. Higher positions and greater masses lead to more potential energy. Potential energy can be calculated using the formula PE=mgh, where m is mass, g is gravitational acceleration, and h is height.
Potential energy often comes from work being done on an object to move it to a higher position or state. For example, lifting a book to place it on a shelf gives it gravitational potential energy. Stretching a spring gives it elastic potential energy. Chemical reactions can produce molecules with chemical potential energy.
Conservation of Energy
The law of conservation of energy states that energy cannot be created or destroyed, only converted from one form to another. This means that the total amount of energy in a closed system always remains constant. Energy is conserved over time.
For example, when a ball falls, its potential energy gets converted to kinetic energy. The initial amount of potential energy gets turned into an equal amount of kinetic energy as the ball falls. The total energy before and after the fall remains the same.
Another example is a pendulum. At the highest point of its swing, it has maximum potential energy. As it swings down, this potential energy gets converted to kinetic energy. At the lowest point, it has maximum kinetic and minimum potential energy. As it swings up again, the kinetic energy gets converted back to potential energy. The total energy remains constant throughout the motion.
In chemical reactions, energy gets rearranged between chemical bonds, but the total energy after the reaction equals the energy before the reaction. This conservation of energy is a fundamental law of physics that applies across all processes and systems. It is a key scientific concept for understanding how energy flows and transforms without being created or destroyed.
Work-Energy Theorem
The work-energy theorem states that the net work done on an object is equal to its change in kinetic energy. This means that the work done on an object by external forces is equal to the change in the object’s kinetic energy.
The work-energy theorem demonstrates the relationship between work and kinetic energy. Work is defined as force multiplied by distance. Kinetic energy depends on an object’s mass and velocity. When an external force does work on an object, that force changes the object’s velocity. This in turn changes the object’s kinetic energy.
For example, if a net force accelerates a box across a floor by pushing it, the work done increases the box’s kinetic energy. The amount of work done by the force is equal to the increase in the box’s kinetic energy. This shows the direct relationship between work and kinetic energy due to an applied force.
Real World Applications
There are many examples of work and energy in real world systems that we encounter everyday.
One example is driving a car. The chemical energy stored in gasoline is converted into kinetic energy that allows the car to move. The engine does work on the car to accelerate it. The faster the car is moving, the more kinetic energy it has.
Another example is lifting objects. When you lift something upwards, you are doing work against gravity to increase its potential energy. The higher it is lifted, the more potential energy the object gains.
Batteries also demonstrate the relationship between energy and work. The chemical energy stored in batteries is converted into electrical energy that can then power devices to do useful work. The more charge the battery has, the more work it can perform before being depleted.
In each example, energy is converted from one form into another in order to perform work. Understanding these real world applications helps illustrate the important role that energy plays in enabling any kind of physical activity or motion.
Conclusion
To summarize, the ability to do work is directly related to energy. The energy that enables work is called kinetic energy, which is the energy of motion, or potential energy, which is stored energy based on an object’s position or state. The law of conservation of energy states that energy can transform between various forms, but it cannot be created or destroyed. The work-energy theorem further defines the relationship between work and energy, stating that the net work done on an object is equal to its change in kinetic energy.
In essence, energy is the capacity to do work. Kinetic and potential energy can convert into each other and transfer between objects, but the total amount of energy in a closed system remains fixed. This conservation of energy is a fundamental law of physics that underlies many everyday examples of work being done through the utilization of energy.
References
While no external sources were directly cited in this content, the information provided is based on fundamental principles of physics that have been established through extensive research and experimentation over the past few centuries. Some of the key scientists and their contributions that laid the groundwork for our modern understanding of energy, work, and their relationship include:
Isaac Newton – Developed laws of motion describing the relationship between force and changes in motion. This provided a basis for later defining kinetic energy.
Thomas Young – Proposed the concept of energy as an entity distinct from force. This led to the development of the term “energy” itself.
James Prescott Joule – Conducted experiments establishing the mechanical equivalent of heat, demonstrating its relationship to other forms of energy. This helped unify various forms of energy under one framework.
Hermann von Helmholtz – Formulated the principle of conservation of energy based on substantial experimental evidence. This principle became a fundamental tenet of physics.
Gaspard-Gustave Coriolis – Developed the kinetic energy formula relating the mass and velocity of objects. This allowed kinetic energy calculations.
William Rankine, Rudolf Clausius – Established the theory of thermodynamic potential energy, encompassing gravitational, electric, and molecular potential energy.
James Clerk Maxwell – Derived a set of equations describing electromagnetic energy and its relationship to electricity and magnetism.
While no specific sources were directly used, this content synthesizes key concepts from centuries of established physics research. The information reflects scientific consensus on the topic.