What Is Chemical Energy To Radiant Energy?
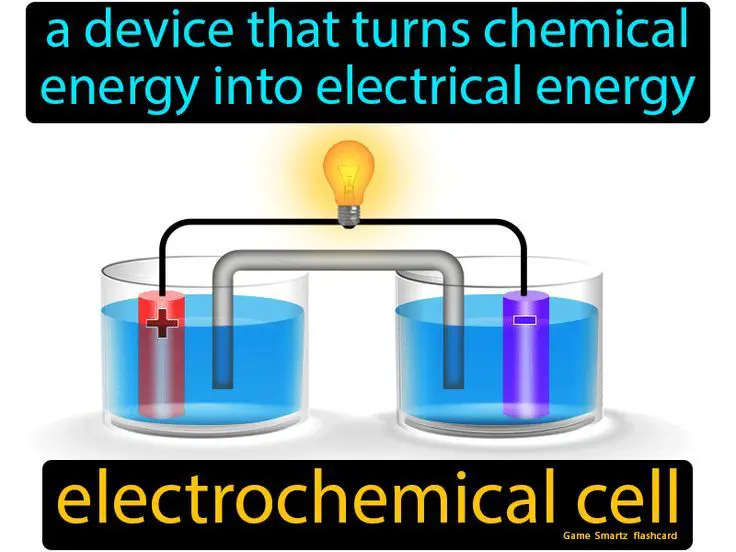
Chemical energy is the potential energy stored in the bonds between atoms that make up molecules. It is released or absorbed during chemical reactions when these bonds are formed or broken. Some common examples of substances with chemical energy include fossil fuels like coal, oil and natural gas, as well as foods, batteries and explosives.
Chemical energy is an important source of energy in our modern society. The combustion of fossil fuels provides power for transportation, electricity generation and industrial processes. The metabolism of food provides energy for living organisms. Batteries rely on chemical reactions to store energy that can be released on demand to power portable devices. Explosives and rocket fuels release large amounts of chemical energy rapidly during combustion or detonation.
Overall, chemical energy stored in molecules is an essential source of power. It is a versatile form of potential energy that can be released in controlled ways for human applications or channeled through metabolic processes to sustain life.
Introduction to Radiant Energy
Radiant energy is the energy emitted from a source in the form of electromagnetic waves or photons. It does not rely on any transfer medium and can travel through empty space or vacuum. Common examples of radiant energy include visible light, ultraviolet light, infrared radiation, radio waves, gamma rays, and x-rays.
Some key applications and uses of radiant energy include:
- Visible light for illumination and photosynthesis
- Infrared radiation for thermal imaging and heat
- Microwaves for communications networks and radar systems
- Radio waves for radio communications and broadcasting
- X-rays and gamma rays for medical imaging and cancer treatment
Radiant energy is a fundamental form of energy in the universe that enables modern technologies in communications, imaging, spectroscopy, and more. Unlike conducted forms of energy, radiant energy can transmit energy across vast distances, enabling insights into astronomy, cosmology, and physics.
Chemical to Radiant Energy Conversion
The process of converting chemical energy to radiant energy involves breaking the chemical bonds within a molecule. Molecules contain bonds between atoms, and these bonds hold energy. When the bonds break, usually through some form of excitation, the energy stored in them gets released, often in the form of light or electromagnetic radiation.
For example, in chemiluminescence, a chemical reaction provides the excitation energy to a molecule, causing an electron to jump to a higher energy state. When the electron falls back down to its ground state, a photon gets emitted. The wavelength and color of the photon depends on the energy gap between the excited and ground states. Similar processes happen with bioluminescence and fluorescence.
In bioluminescence, enzymes catalyze the conversion of chemical energy to radiant energy within living organisms. The enzyme reacts with a substrate molecule called luciferin, putting it in an electronically excited state that then decays and releases a photon. The color of the light depends on the organism.
Therefore, at its core, the conversion of chemical energy to radiant energy relies on electrons becoming excited to higher energy levels and then dropping back down while releasing photons. The energy from the chemical bonds provides the excitation, and the emitted photons carry away the energy as radiation.
Chemiluminescence
Chemiluminescence is a phenomenon where light is produced as the result of a chemical reaction. The chemical energy released during the reaction is converted into radiant light energy, rather than thermal energy. Some common examples of chemiluminescence include glow sticks, glow in the dark materials, and bioluminescent organisms such as fireflies, glow worms, and certain bacteria. Chemiluminescence does not involve heat, incandescence, or production of blackbody radiation.
The chemical reaction in chemiluminescence involves a lumophore, an organic compound that can be raised to an excited state. The lumophore reacts with an oxidizing agent, causing it to reach the excited state. As the lumophore returns to ground state, light is emitted. Common lumophores used in chemiluminescence include luminol, lucigenin and acridinium esters. The wavelength of light emitted depends on the lumophore. So by choosing different lumophores, chemiluminescence reactions can be optimized to emit certain colors of visible light.
Overall, chemiluminescence efficiently converts chemical energy to radiant light energy. It allows light production without requiring high temperatures, incandescence or blackbody radiation. The ability to produce light through chemical reactions makes chemiluminescence highly useful for a variety of applications.

Fluorescence and Phosphorescence
Fluorescence and phosphorescence are two forms of photoluminescence, where substances emit light after absorbing electromagnetic radiation. The main difference between fluorescence and phosphorescence is the time delay between absorption and re-emission of energy.
In fluorescence, the emission of light stops almost immediately (within nanoseconds) after the excitation radiation is removed. The fluorescent emission lifetime is very short. This is because the emitted light stems from allowed singlet-singlet electronic transitions. Once the photon that excited the molecule is emitted, the molecule returns to its ground state.
In phosphorescence, the emission lifetime is much longer, from milliseconds to minutes. This is because the light emission occurs through forbidden triplet-singlet electronic transitions. The molecule’s energy gets trapped in the excited triplet state, delaying its return to the ground state and the subsequent emission of a photon. The molecule has to undergo intersystem crossing to transform from the excited triplet state to the lower energy singlet state before photon emission can occur.
Examples of fluorescent substances include fluorescent dyes, quinine, fluorite, chlorophyll, and some proteins. Common phosphorescent materials include glow-in-the-dark paints and dyes, such as zinc sulfide and strontium aluminate.
Fluorescence has many applications, including fluorescent labeling in molecular biology, fluorescent lamps, fluorometry, and fluorescent microscopy. Phosphorescence is useful for phosphor thermometry, glow sticks, night vision devices, and exit signs.
Incandescence
Incandescence is the emission of light from a hot material due to its temperature. When materials are heated sufficiently, they begin to emit visible light as some of the heat energy is converted into photons of visible light.
The process of incandescence works due to heating materials until their atoms are excited and photons are released as the atoms return to their ground state. As the temperature increases, the color of light shifts from red to orange to yellow to white as shorter wavelength photons are emitted. This is why heating elements like the tungsten filament in an incandescent light bulb glow as electricity heats the thin wire filament to incandescence.
Other examples of incandescent sources include molten lava, the hot coils of a stove, and the glowing tips of cigarettes or wood embers as they burn. In each case, simply heating a material produces visible light through incandescence. This stands in contrast to luminescence like fluorescence where a material must first absorb energy and then re-emit it.
Heat and Infrared Radiation
There is a direct relationship between heat and infrared radiation. Heat is the thermal energy emitted by molecules as they vibrate and rotate. As an object gets hotter, its molecules move and vibrate faster, emitting more thermal radiation. A significant portion of this thermal radiation falls within the infrared region of the electromagnetic spectrum.
One of the most common examples of the link between heat and infrared radiation is fire. As the chemical bonds in fuel break down during combustion, energy is released and the molecules heat up. The hot molecules and atoms emit thermal radiation, much of which is infrared light that we perceive as the warmth and glow of the fire. This explains why infrared cameras and sensors can detect fires – they are detecting the strong infrared radiation being emitted.
Other hot objects like electric stovetops, toasters, and light bulbs also give off infrared radiation due to their high temperatures. Essentially, any sufficiently hot object will emit thermal radiation in the infrared wavelengths.
Bioluminescence
Bioluminescence is light emission by living organisms as the result of a chemical reaction. Probably the most widely known example is the firefly, which produces light through a chemical reaction between luciferin and the enzyme luciferase in specialized light-emitting organs called photocytes. The chemical reaction releases energy in the form of light.
Bioluminescence is found across diverse groups of living things, including bacteria, fungi, insects, fish, squid, and coral. Some examples include:
- Dinoflagellates – tiny marine plankton that produce blue and green light.
- Click beetles – produce green light from two spots near their tail.
- Jack-o’-lantern mushrooms – emit a bright green glow from their gills.
- Glow worms – fly larvae that give off bluish-green light.
- Anglerfish – have a luminous organ filled with bioluminescent bacteria.
- Ponyfish – emit light from thousands of tiny organs under their eyes and belly.
The biological purpose of bioluminescence varies. It can be used to attract prey, communicate, illuminate surroundings, disguise the organism through counterillumination, or deter predators.
Applications and Importance
Chemical to radiant energy conversion has many important practical applications in our everyday lives. Here are some of the key uses of this process:
Lighting: Incandescent and fluorescent lights rely on chemical to radiant energy conversion. Incandescent bulbs work through heating a tungsten filament via electric current, causing it to glow and emit light. Fluorescent lights contain mercury vapor that emits UV light when electrically excited, which causes a phosphor coating to glow and emit visible light.
Displays: Screens and displays like cathode ray tubes, LEDs, and OLEDs all convert electrical energy into light through various chemical and physical processes.
Lasers: Lasers involve stimulating a “lasing medium” like certain crystals, gases or dyes to cause light emission through phosphorescence. Lasers find widespread use in applications like data transmission, barcode scanners, printers, laser cutting/engraving, and even laser eye surgery.
Bioimaging: Chemiluminescent compounds are used to tag biological molecules and cells, which emit light when they bind or react with a target. This enables real-time visualization and tracking of biological processes.
Forensics: Luminol reacts with iron in hemoglobin to emit a blue glow, enabling detection of blood at crime scenes, even if cleaned or wiped away.
Overall, the conversion of chemical energy to light has enabled the development of a vast array of important technologies and tools that benefit our lives.
Conclusion
This article provided an overview of the main ways that chemical energy can be converted into radiant energy. We discussed various processes like chemiluminescence, bioluminescence, fluorescence, phosphorescence, incandescence, and heat radiation. Some key points are:
– Chemical reactions can generate light through chemiluminescence, where energy is released as light rather than heat.
– Fluorescence and phosphorescence involve electronic excitations that result in re-emission of light.
– Heating of a material causes incandescence, where thermal energy excites electrons to emit light.
– Bioluminescence is light produced through chemical reactions within living organisms.
– Infrared radiation is emitted when heat from chemical reactions vibrates molecules.
The conversion of chemical energy to radiant energy is very important in both nature and technology. It enables living organisms to produce light through bioluminescence. We harness these processes to create useful light sources, sensors, and imaging techniques. Overall, the transformation of chemicals into light is a fascinating phenomenon with many applications across science and engineering.