What Is The Description Of Heat?
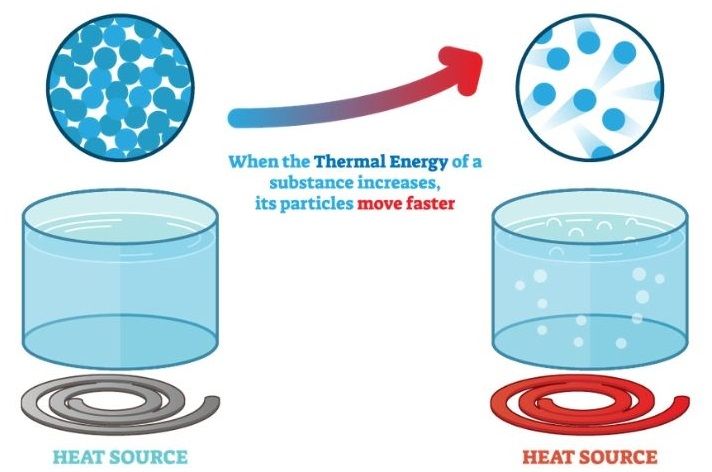
Heat refers to thermal energy or the movement of energy between two objects with different temperatures. Thermal energy is the total energy of microscopic motions of particles in matter and cannot be seen or felt directly. Heat is an important concept in physics, chemistry, engineering, and daily life.
Heat drives many essential processes that allow life to exist. Examples include chemical reactions within cells, digestion and circulation in animals, weather patterns that provide rain and crops, and industrial processes that create modern goods. Heat transfer, or the movement of thermal energy from one object or system to another, enables heating and cooling of homes, cooking of food, generation of electricity, and more.
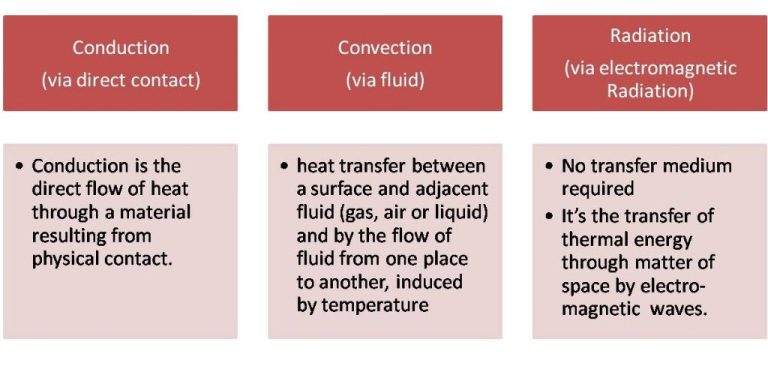
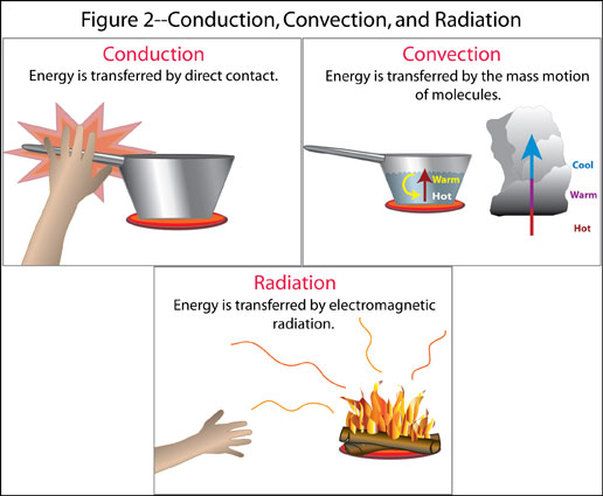
There are three main mechanisms of heat transfer: conduction, convection, and radiation. Conduction transfers heat through direct contact between objects, convection transfers heat via the movement of fluids or gases, and radiation transfers heat through electromagnetic waves. Understanding the principles of heat transfer allows scientists and engineers to design efficient heating, cooling, and power generation systems.
Thermal Energy and Temperature
Although often used interchangeably, thermal energy and temperature are distinct scientific concepts. Thermal energy refers to the total internal energy of a system due to the motion of its atoms and molecules. It is measured in joules in the SI system. Temperature, on the other hand, is a measure of the average kinetic energy of particles in a system. It indicates how hot or cold an object is.
Temperature is expressed on a scale, with common units being degrees Fahrenheit, Celsius, and Kelvin. On the Fahrenheit scale, the freezing point of water is 32°F and the boiling point 212°F. The Celsius scale sets the freezing point and boiling point of water at 0°C and 100°C respectively. The Kelvin temperature scale is an absolute scale, with 0 K (-273.15°C) representing absolute zero. Unlike Celsius and Fahrenheit scales, the Kelvin scale does not use the term degrees.
While temperature depends on the average kinetic energy of molecular motion, thermal energy depends on the total kinetic energy of all particles in a system. Adding heat increases thermal energy, but two objects at the same temperature may have different thermal energies depending on their composition and mass.
Heat Transfer
Heat transfer is the exchange of thermal energy between physical systems. There are three main mechanisms of heat transfer:
Conduction
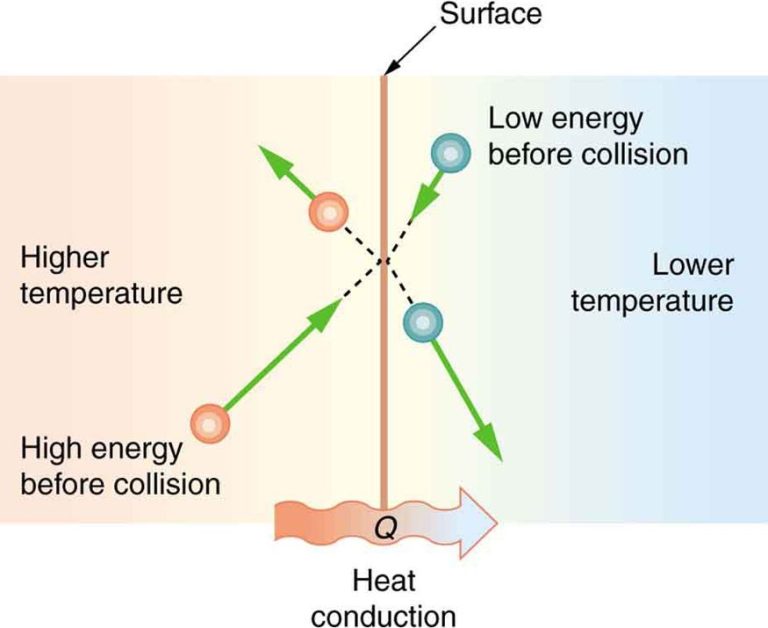
Conduction is the transfer of heat between substances that are in direct contact with each other. It occurs when electrons from high-energy atoms interact with lower energy neighboring atoms, transferring kinetic energy. Metals are good conductors of heat because they contain many free electrons. Examples of conduction include touching a hot stove, heat traveling through a copper wire, or heat moving through the iron plate of an electric skillet into the food.
Convection
Convection is the mode of heat transfer between a surface and a moving fluid such as air or water. It occurs due to the circulation and buoyancy of the fluid. As the fluid is heated, it expands, becomes less dense, and rises. Cooler fluid then displaces it. This cycle continuously circulates heat from the bottom to the top. Examples of convection include steam rising from a pot of boiling water and warm air rising from a radiator.
Radiation
Thermal radiation is the emission of electromagnetic waves from all matter above absolute zero. It propagates heat without relying on particle collisions. Radiation does not require a medium to transfer heat. It can travel through vacuums, like heat from the sun warming the Earth. Other examples include heat glowing from hot stovetop burners and heat escaping through the glass of a greenhouse.
Specific Heat Capacity
Specific heat capacity, also known simply as specific heat, is a material property that describes the amount of heat energy required to raise the temperature of 1 gram of a substance by 1 degree Celsius. It is an intensive property, which means it does not depend on the size or amount of the material.
Substances with a high specific heat capacity require more heat energy to change their temperature compared to substances with a low specific heat capacity. For example, water has a very high specific heat capacity of 4.18 J/g°C. This means it takes 4.18 Joules of heat energy to raise the temperature of 1 gram of water by 1°C. In contrast, iron has a specific heat capacity of only 0.45 J/g°C. So iron heats up and cools down much more quickly than water when the same amount of heat energy is added or removed.
The specific heat capacity of a substance is what determines how quickly its temperature rises or falls when heat is transferred to or from the material. Materials like water that have a high heat capacity will change temperature slowly, while low heat capacity materials like metal will change rapidly. This property has important implications for processes like heating/cooling systems, cooking, and weather.
Latent Heat
Latent heat is the thermal energy absorbed or released by a substance during a change in its physical state that occurs without changing its temperature. Some common phase changes that involve latent heat are the melting of ice into water and the boiling of water into steam.
There are two specific types of latent heat:
Latent Heat of Fusion: The latent heat absorbed or released when a substance changes from solid to liquid or vice versa is called the latent heat of fusion. For example, when ice melts into water, its temperature remains constant at 0°C until all the ice has completely melted. The energy required for this melting process without a temperature change is the latent heat of fusion.
Latent Heat of Vaporization: The latent heat absorbed or released when a substance changes from liquid to gas or vice versa is called the latent heat of vaporization. For instance, when water boils and turns into steam, its temperature remains constant at 100°C until all liquid water has completely vaporized. The energy required for this vaporization process without a temperature change is the latent heat of vaporization.
Latent heat is an important concept in thermodynamics and is useful in understanding heat transfer processes that occur during phase changes. The high values of latent heats explain why significant amounts of energy are required to change the phase of a substance without altering its temperature.
Laws of Thermodynamics
The laws of thermodynamics govern the transfer of energy in thermal systems. There are three primary laws of thermodynamics that provide a mathematical description of the relationships between heat, work, temperature, and energy.
The zeroth law of thermodynamics states that if two systems are in thermal equilibrium with a third system, then they are also in thermal equilibrium with each other. This law establishes the concept of temperature and allows the creation of thermometers.
The first law of thermodynamics, also known as the law of conservation of energy, states that energy cannot be created or destroyed in an isolated system. The total energy of a system remains constant. This means that heat and work are interchangeable. The first law provides a definition for internal energy and establishes its relationship with heat and work.
The second law of thermodynamics introduces the concept of entropy. It states that the total entropy of an isolated system always increases over time. Entropy is a measure of molecular disorder or randomness. The second law implies that it is impossible to have a thermodynamic process that converts heat completely into work. It also imposes a limit on the efficiency of heat engines.
The laws of thermodynamics have wide-ranging implications and applications. They limit what is possible in science and engineering. Heat engines such as internal combustion engines are designed around the limits imposed by the laws of thermodynamics. The laws also form the backbone of statistical mechanics and provide insights into the nature of time and the direction in which processes occur. However, the laws break down when applied to extremely small scales or General Relativity.
Heat Transfer Equipment
Heat transfer equipment utilizes the laws of thermodynamics to transfer heat between two or more process fluids. This equipment is essential for heating, cooling, condensing, evaporating, and controlling process temperatures in a wide range of industrial applications.
Some common types of heat transfer equipment include:
- Heat Exchangers – Used to transfer heat between two fluids without mixing between the fluids. Examples include shell and tube heat exchangers, plate heat exchangers, air cooled heat exchangers, and more.
- Boilers – Transfers heat to water or other process fluids to produce hot water or steam.
- Condensers – Cools hot vapor into a liquid condensate. Used in HVAC systems, power plants, and industrial processes.
- Reboilers – Boils liquid into vapor by transferring heat into the liquid. Used in distillation columns in chemical plants.
- Evaporators – Evaporates a liquid into a vapor by boiling. Used to concentrate solutions.
- Cooling Towers – Transfers waste heat to the atmosphere though the cooling of a water stream to a lower temperature.
Heat exchangers like shell and tube exchangers rely on the laws of thermodynamics to efficiently transfer heat between the hot and cold fluids. The rate of heat transfer depends on having sufficient temperature driving force (temperature difference between fluids), heat transfer surface area, and fluid flow rates, as dictated by the laws of thermodynamics.
Boilers also depend on thermodynamic principles to produce steam or hot water. Combustion or electric heating generates heat according to the laws of thermodynamics. This heat is transferred to the working fluid, with the rate of heat transfer depending on the same factors that govern heat exchangers.
Overall, heat transfer equipment applies the laws of thermodynamics to practically transfer heat in a controlled manner for heating, cooling, condensing, boiling, and evaporation processes in a wide range of industrial and engineering applications.
Heat Transfer in Everyday Life
Heat transfer is a crucial process that enables many everyday technologies and activities. Here are some examples of how heat transfer impacts our daily lives:
Cooking relies heavily on heat transfer. When cooking on a stovetop, conduction transfers heat from the burner to the pot. Convection currents in the water or air then distribute the heat within the pot. Radiation from the heated pot can also warm you. Different cooking methods take advantage of various heat transfer mechanisms.
Heating, ventilation and air conditioning (HVAC) systems use heat transfer to control indoor environments. Furnaces and heaters use conduction and convection to transmit thermal energy. Vents leverage convection to circulate hot and cold air. Radiators and baseboard heaters use radiation to warm living spaces.
Engine operation depends on heat transfer. In combustion engines, heat transfers from burning fuel into the metal engine block. This thermal energy then converts into kinetic energy to move the pistons. Engines also rely on conduction, convection and radiation to cool critical components.
These examples demonstrate that heat transfer principles power many essential appliances and machines. Understanding the heat transfer at work allows engineers to optimize these everyday technologies.
Careers in Thermal Science
Thermal science plays an important role in many industries and careers. Here are some of the key jobs where knowledge of thermodynamics and heat transfer is critical:
Mechanical Engineer: Mechanical engineers design power-generating machines like combustion engines, turbines, refrigeration and air conditioning systems. Understanding thermodynamics allows them to properly size and select components like heat exchangers, pumps and compressors.
Chemical Engineer: Chemical engineers rely on heat transfer principles for designing and operating chemical plants and petroleum refineries. This includes heat integration, reactor design, distillation columns and more.
Physicist: Physicists studying thermodynamics add to our theoretical knowledge of heat transfer and push the boundaries of what’s possible. Their discoveries lead to new technologies and capabilities.
HVAC Technician: HVAC (heating, ventilation and air conditioning) technicians use their knowledge of thermodynamics for sizing, selecting, installing and maintaining building systems. Proper heat transfer principles are key for efficiency and comfort.
Whether developing new engines, improving manufacturing processes or designing comfortable indoor spaces, thermal scientists and engineers play a vital role. Their expertise in thermodynamics and heat transfer leads to significant advances across many fields.
Future of Thermal Science
Thermal science is an active area of research with many exciting developments on the horizon. Here are some of the key areas researchers are exploring:
Nanoscale thermal management
Controlling heat flow at the nanoscale has major implications for improving electronics, energy conversion devices and even medical treatments. New nanomaterials like graphene and carbon nanotubes could revolutionize thermal management in nanoelectronics and thermoelectrics. Microscale thermal transport and interactions also need further study.
Renewable heating and cooling
Renewable energy sources like geothermal, solar thermal and waste heat harvesting can provide sustainable heating and cooling for homes and industry. Improving efficiency and reducing costs will be key areas of innovation.
Thermal energy storage
Storing thermal energy from intermittent sources like the sun and recovering waste heat for later use provides reliability for renewable energy systems. Phase change materials and thermochemical storage are cutting-edge solutions under development.
Biomedical applications
Precise temperature control is crucial for medical therapies like hyperthermia cancer treatment and cryosurgery. Non-invasive temperature monitoring techniques and optimized thermal delivery methods are active research areas that can improve patient outcomes.
High-temperature materials
Materials that can withstand extremely high temperatures are needed for hypersonic vehicles, nuclear fusion reactors and deep space exploration. Ceramic composites, ultra-high temperature ceramics and refractory metals are being studied to push material limits.
Thermal scientists have an essential role to play in solving global energy challenges and developing transformative new technologies. With many fundamental questions still unanswered, the future is wide open for the next generation of thermal science pioneers.