What Is Heat And Heat Formula?
What is Heat?
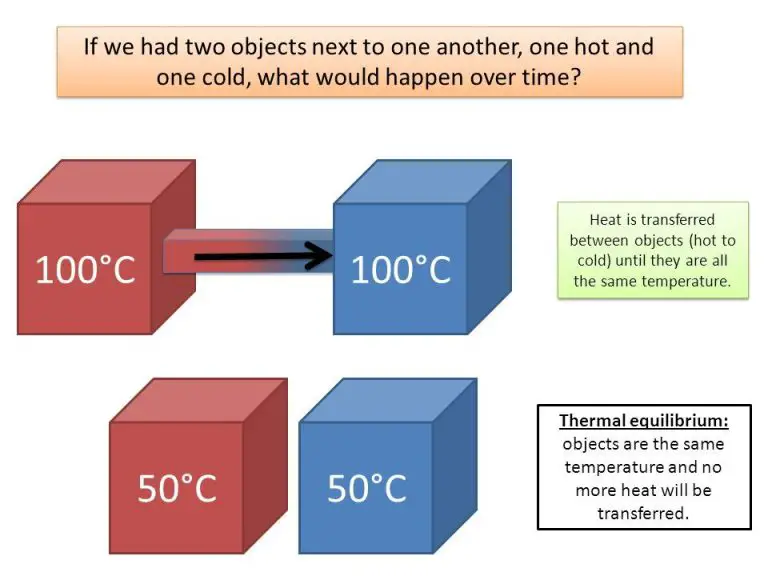
Heat is a form of energy that is transferred between objects or systems due to temperature difference. When two objects at different temperatures come in contact, heat flows spontaneously from the hotter object to the colder one until they reach thermal equilibrium. Unlike temperature, which is a measure of the average kinetic energy of particles in a substance, heat is the total amount of kinetic energy due to molecular motion that is transferred between objects.
For example, when you place your hand on a hot stove, heat energy transfers from the stove, which is at a higher temperature, to your hand, which is at a lower temperature. This flow of heat is what causes the sensation of warmth in your hand. As more heat transfers, the temperature of your hand increases. Meanwhile, the stove loses some of its heat energy, causing it to cool down slightly. Heat will continue to flow between the two objects until their temperatures equalize.
So while temperature refers to the degree of hotness or coldness of an object, heat refers specifically to the amount of energy being transferred due to a temperature difference. Heat is measured in units of energy such as joules or calories, while temperature is measured in degrees like Celsius, Fahrenheit, or Kelvin.
Measuring Heat
Heat is measured in various units depending on the system being used. Some common units for heat are:
- Calories (cal) – This is the amount of heat required to raise 1 gram of water 1°C at standard atmospheric pressure. A calorie is sometimes written with a capital C (Calorie) when referring to the amount of heat required to raise 1 kilogram of water 1°C.
- British thermal units (BTU) – This is the amount of heat required to raise 1 pound of water 1°F. BTUs are commonly used to measure heat in U.S. customary units.
- Joules (J) – The joule is the SI unit for energy and work. One joule is defined as the amount of work required to produce one watt of power for one second. Joules are commonly used to measure heat in the SI system.
The specific heat capacity of a substance is the amount of heat required to raise one unit of mass of that substance by 1°C. Substances with higher specific heat capacities require more heat to change their temperature. The specific heat capacity depends on the types of atoms/molecules in the substance and their arrangements.
Heat Transfer
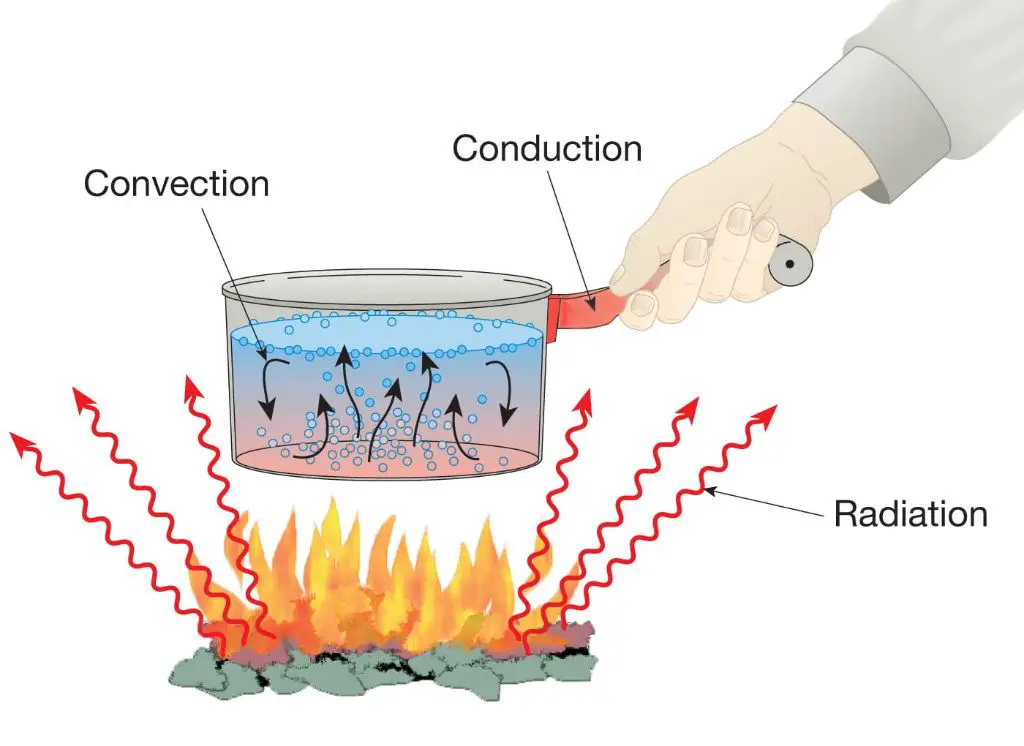
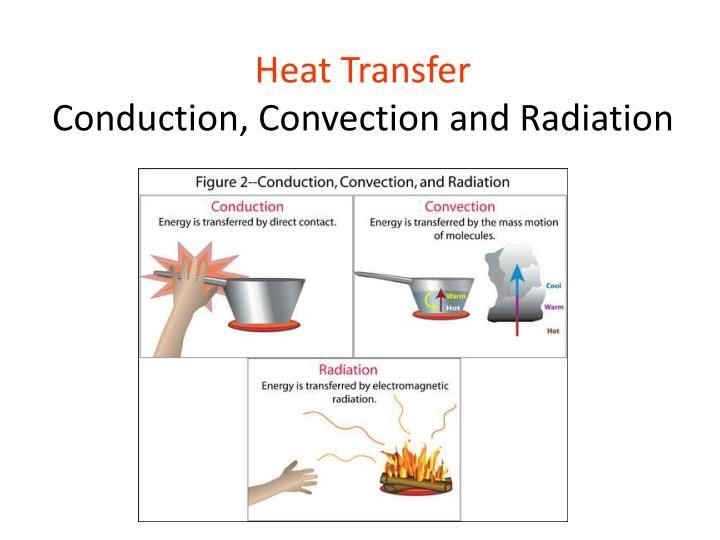
Heat transfer refers to the process of transferring thermal energy from one location, object, or system to another. There are three main modes of heat transfer: conduction, convection, and radiation.
Conduction
Conduction is the direct transfer of thermal energy between neighboring molecules. It occurs in all forms of matter – gases, liquids, solids – although it is most effective in solids. Metals are good conductors as their free electrons can transfer thermal energy rapidly through the structure. Insulators have strong molecular bonds that inhibit conduction.
Convection
Convection is the mode of heat transfer between a surface and a moving fluid such as a gas or liquid. It relies on the circulation and motion of the fluid to transfer heat. Convection can be natural, driven by density differences in the fluid, or forced, driven by external means like fans or pumps. Convection transfers thermal energy through mass transfer of a heated fluid.
Radiation
Thermal radiation refers to electromagnetic waves emitted by a heated surface. It does not require a medium for transmission. All objects emit thermal radiation based on their temperature. Radiation is responsible for heat transfer in a vacuum. It can travel long distances and gets absorbed when it strikes another object. The amount of radiation emitted depends on the object’s temperature and surface properties.
Effects of Heat
Heat has several important effects that are useful to understand.
Thermal Expansion
One major effect of heat is thermal expansion. When an object is heated, the kinetic energy of its molecules increases. The increased motion of the molecules causes them to bump into each other and spread apart slightly. This causes the material to expand in size.
Thermal expansion applies to solids, liquids, and gases. Solids typically expand the least with heat, while gases expand the most. The degree of expansion depends on the material’s coefficient of thermal expansion.
Thermal expansion has many practical implications. It is the reason bridges have small gaps between parts and railroad tracks have gaps between the rails. These gaps allow the materials to expand on hot days without buckling. Thermal expansion also explains why the tops of plastic lids on jars are larger than the bottoms.
Phase Changes
Another major effect of heat is enabling phase changes. When matter gains enough heat energy, the molecules can transition to a higher energy state, changing the phase. For example, when solid ice absorbs enough heat, it transitions to liquid water. With more heat, liquid water turns into gaseous water vapor.
These phase changes — melting, vaporization, boiling, condensation, etc. — require large amounts of latent heat to be added or removed. The latent heat represents the total energy required to change the structure of the molecules and overcome intermolecular forces during a phase change.
Understanding phase changes is crucial for many chemical and physical processes. The addition or removal of heat drives phase transitions that are essential in industry, cooking, weather patterns, and more.
Heat and Temperature
Heat and temperature are closely related concepts, but they are not the same thing. Heat refers to the total kinetic energy of molecules while temperature is a measure of the average kinetic energy of molecules.
As heat is added to a substance, the molecules gain kinetic energy and start moving faster. This increased molecular motion corresponds to an increase in temperature. So heating a substance increases its temperature.
However, temperature does not give information about the total quantity of heat present. For example, a bathtub of hot water has more total heat than a cup of hot water even if both are at the same temperature.
To summarize, heat is the total kinetic energy of particles in a substance while temperature is a measure of the average kinetic energy and speed of particles. Adding heat raises temperature, but equal temperatures do not necessarily mean equal heat content.
Heat Capacity and Specific Heat
Heat capacity is defined as the amount of heat energy required to raise the temperature of a substance by one degree. It depends on the mass of a substance and its specific heat capacity. Heat capacity is measured in joules per kelvin (J/K).
Specific heat capacity, also called specific heat, is defined as the amount of heat energy required to raise the temperature of one gram of a substance by one degree Celsius. It is an intensive property, meaning it is independent of the mass of a substance. Specific heat is measured in joules per gram per degree Celsius (J/g°C).
The specific heat of some common substances is:
| Substance | Specific Heat (J/g°C) |
| Water | 4.18 |
| Ice | 2.05 |
| Iron | 0.449 |
| Gold | 0.129 |
| Aluminum | 0.897 |
The specific heat of a substance can be used along with mass and temperature change to calculate the amount of heat energy absorbed or released in various processes.
Latent Heat
Latent heat is the energy absorbed or released by a substance during a phase change between a solid, liquid or gas. Some common examples of phase changes involving latent heat are:
- Melting – when a solid turns into a liquid, such as ice melting into water. The absorbed latent heat is known as the heat of fusion.
- Freezing – when a liquid turns into a solid, such as water freezing into ice. The released latent heat is the heat of fusion.
- Vaporization – when a liquid turns into a gas, such as water boiling into steam. The absorbed latent heat is the heat of vaporization.
- Condensation – when a gas turns into a liquid, such as steam condensing into water. The released latent heat is the heat of vaporization.
Unlike sensible heat which results in a temperature change, latent heat does not change the temperature. Instead, it allows for a change in the physical state of matter while absorbing or releasing energy. Understanding latent heat is crucial in many scientific and engineering processes and applications.
Heat Transfer Rate
The rate of heat transfer depends on the temperature difference between two objects and the thermal conductivity of the material through which the heat is flowing. This relationship is described mathematically by Fourier’s Law of Heat Conduction:
Where is the heat transfer rate, is the thermal conductivity, A is the cross-sectional area, is the temperature difference, and is the distance over which the temperature difference occurs.
This shows that the greater the temperature difference and the higher the thermal conductivity, the faster heat will be transferred. Materials like metals have high thermal conductivity, allowing heat to flow readily through them. Insulators have low thermal conductivity, severely restricting heat transfer.
Heat Engines
A heat engine is a device that converts heat energy into mechanical energy. The most common examples of heat engines are internal combustion engines and steam turbines.
Heat engines follow a thermodynamic cycle where a working fluid is heated, expands to produce mechanical work, then is cooled and compressed before repeating the cycle. The efficiency of a heat engine is determined by how much of the heat input is converted into useful work, versus lost as excess heat. This is described by the thermal efficiency equation:
Thermal efficiency = (Work output) / (Heat input)
The theoretical maximum efficiency of a heat engine operating between two temperatures is defined by the Carnot efficiency. However, real engines have lower efficiencies than this ideal due to irreversible losses such as friction and heat loss. Gasoline and diesel engines typically have thermal efficiencies around 20-30%. Large power plant steam turbines can reach over 40% efficiency.
Improving the efficiency of heat engines is an important area of thermodynamic research and development. Even small gains in efficiency can produce major energy savings and reductions in waste heat for power generation systems.
Real-World Applications of Heat
Heat plays an important role in many real-world applications and technologies that we use every day:
Cooking
Cooking relies heavily on the transfer and application of heat to food. Different cooking methods like baking, frying, grilling etc. all use heat to transform ingredients into tasty meals. The way heat interacts with different foods affects texture, flavor, aroma and more.
HVAC Systems
HVAC (heating, ventilation and air conditioning) systems use heat pumps and furnaces to control temperatures in homes and buildings. Heat is transferred to or from a space via convection, conduction or radiation to maintain comfortable conditions.
Internal Combustion Engines
Engine heat is essential for powering vehicles with internal combustion engines. Controlled explosions and burning of fuel results in heat release, which gets converted into mechanical motion that propels the vehicle.
Other examples where heat plays a critical role include chemical processing, space heating, electronics cooling, power generation, and more. The principles of heat are applied across many industries to enable modern technologies.