What Determines The Potential Of Energy Examples?
Potential energy is the stored energy an object has due to its position or chemical configuration. Understanding potential energy is important because it allows us to quantify the energy available to do work in a system. Potential energy can take on different forms based on the forces at play in the system, and the configuration of objects within it. The potential energy present determines the maximum amount of kinetic energy that can be produced when that potential energy is converted.
In this article, we will provide an overview of the main types of potential energy and what factors determine the potential energy in each case. This will provide insight into where potential energy comes from, how we can calculate it, and how it can be harnessed to do useful work.
Gravitational Potential Energy
Gravitational potential energy depends on an object’s mass, the strength of gravity, and the height of the object. For example, when you lift an object, you increase its gravitational potential energy. The higher you lift it, the more potential energy it gains. Another example is water held behind a dam. The water has gravitational potential energy that can be converted to electricity as it flows through turbines. Similarly, objects placed on shelves have gravitational potential energy relative to the floor. The heavier the object and the higher the shelf, the greater the stored energy. Gravitational potential energy can be calculated by multiplying an object’s mass, gravitational acceleration, and height. This fundamental source of potential energy plays a role anytime objects are raised or lowered.
Elastic Potential Energy
Elastic potential energy depends on the elasticity of a material and how far it’s stretched or compressed. Some common examples that utilize elastic potential energy are rubber bands, springs, and trampolines. When these objects are stretched or compressed, the elastic material stores energy. When released, this stored energy is converted into kinetic energy as the object returns to its original shape. The amount of elastic potential energy stored depends on the spring constant of the material, as well as how much deformation occurs from the equilibrium state. The spring constant measures how stiff or resistant to deformation a material is. A stiffer material can store more elastic potential energy when deformed by the same distance, compared to a more flexible material.
For example, when you pull back a rubber band, you are doing work to stretch the band. This transfers energy into the rubber band and stores it as elastic potential energy. When you release the stretched rubber band, the stored energy is immediately converted into kinetic energy as the band snaps back to its original shape and size. The farther you stretch the band back, the more potential energy builds up and the faster it will snap back. Springs work through this same principle, building up greater elastic potential energy when compressed tighter.
Chemical Potential Energy
Chemical potential energy depends on the types of molecules involved and the molecular bonds between them. This type of potential energy can be found in batteries, food, fuel, and other substances with chemical reactions.
For example, the chemical bonds within the molecules of gasoline contain potential energy that can be released through combustion in a car engine. The hydrocarbon molecules have high energy bonds between carbon and hydrogen atoms. When these bonds are broken during burning, the energy is released as heat and kinetic energy to power the engine.
Foods like bread, fruits, and vegetables also contain chemical potential energy within the molecular bonds that can be converted to kinetic energy for biological processes. The cells in our bodies break down nutrients through digestion and respiration to harness this potential energy for movement, growth, and other functions.
Batteries rely on reversible chemical reactions to store and release energy on demand. The difference in potential energy between the chemicals involved in the redox reactions allows batteries to provide electricity.
In all cases, the amount of potential energy available depends on the types of molecules present and the strength of their chemical bonds. High energy bonds have greater potential to release energy when broken or altered. Understanding chemical potential energy is key for fields like nutrition, metabolism, combustion, and battery technology.
Nuclear Potential Energy
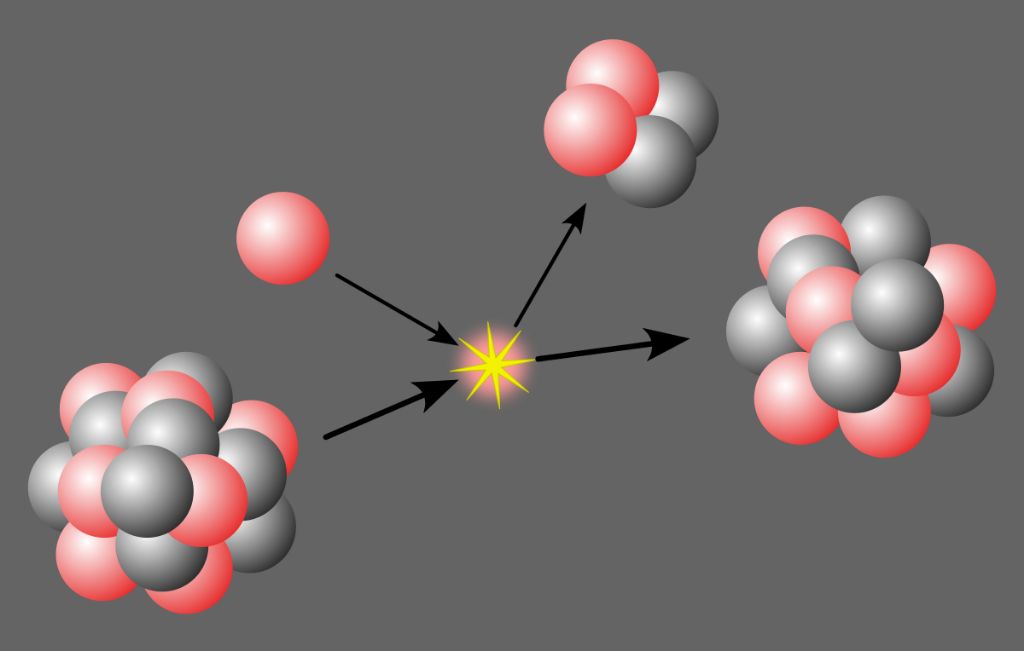
Nuclear potential energy depends on the binding energy holding nuclei together. The strong nuclear force between nucleons (protons and neutrons) in the atomic nucleus counteracts the electrostatic repulsion between protons. When nuclei are split (nuclear fission) or fused (nuclear fusion), this releases energy from the net reduction in binding energy. Nuclear potential energy is the basis for nuclear power generation in nuclear reactors, as well as nuclear weapons such as atomic bombs.
In nuclear fission, a heavy unstable nucleus like uranium-235 breaks apart into lighter nuclei, releasing energy in the process. Nuclear power plants use the heat from fission reactions to produce steam and generate electricity. The opposite process, nuclear fusion, occurs when light nuclei are fused together into heavier and more stable nuclei. Fusion is the process that powers stars like our sun, and could potentially be harnessed for future fusion power plants.
Both fission and fusion convert a fraction of the mass of nuclei into energy, described by Einstein’s famous equation E=mc2. Even a small amount of mass change results in tremendous amounts of energy, millions of times greater than chemical reactions. This makes nuclear potential energy extremely dense and powerful, but also dangerous if not properly controlled and contained.
Electrostatic Potential Energy
Electrostatic potential energy depends on the electric charge and distance between charged objects. It is the potential energy stored in the electric field created by stationary charges. Greater electrostatic potential energy results from larger amounts of charge and smaller distances between them. Some common examples that involve electrostatic potential energy include capacitors, lightning, and static electricity.
Capacitors are devices that store electric charge on conductive plates separated by an insulator. As charge builds up on the plates, electrostatic potential energy increases. This energy can be released to power electronic devices when the circuit is closed.
Lightning is caused by electrostatic discharge between electrically charged regions in clouds or between a cloud and the ground. The large difference in electric potential between the cloud and ground builds up high electrostatic potential energy. When the strength of the electric field exceeds the insulating properties of air, a lightning bolt discharges the energy.
Static electricity is a buildup of electric charge on surfaces through contact and separation. For example, an object can gain excess negative charge and another object excess positive charge. As the distance increases, so does their electrostatic potential energy. An electric discharge occurs when the repulsive electrostatic force exceeds the resistive force and the objects rapidly move toward each other, releasing energy. This is experienced as a small electric shock.
Magnetic Potential Energy
Magnetic potential energy depends on the strength of the magnetic field and the alignment of magnetic dipoles. Magnets have north and south poles. When opposite poles are close to each other, the magnets are attracted to each other. When like poles are close to each other, the magnets repel each other. The strength of the magnetic interaction depends on the strength of the magnetic field. The stronger the magnetic field, the greater the magnetic force.
A common example of magnetic potential energy is magnets sticking to refrigerators. Refrigerators are made of ferromagnetic metals like iron. This allows magnets to stick to them. The magnet has potential energy that can exert an attractive magnetic force onto the refrigerator. When the magnet moves and sticks to the refrigerator, the potential energy is converted into kinetic energy as the magnet accelerates towards the refrigerator.
Thermal Potential Energy
Thermal potential energy depends on the temperature of a system. The higher the temperature, the greater the thermal potential energy. Examples of harnessing thermal potential energy include:
- Geothermal energy – Tapping into the Earth’s internal heat from deep underground reservoirs of hot water and steam. This thermal energy can be used to generate electricity.
- Heat sinks – Objects designed to absorb and dissipate heat from another object. Heat sinks allow the rapid transfer of thermal energy to keep components from overheating.
Thermal potential energy can also refer to the energy stored in chemical bonds that can be released as heat during reactions. Burning fossil fuels like coal, oil, and natural gas converts their chemical potential energy into thermal energy.
Sound Potential Energy
Sound potential energy depends on the loudness or amplitude of the sound waves. The greater the amplitude, the more potential energy is contained within the waves. When sound waves vibrate through a medium like air, they carry energy that can be converted into kinetic energy.
Speakers and musical instruments demonstrate sound potential energy. Playing a loud guitar note involves plucking the string with enough force to create high amplitude sound waves. Those energetic waves emanate from the guitar and travel through the air until they reach your ear. The membrane of your eardrum vibrates sympathetically with the sound waves, converting the potential energy back into kinetic energy.
Other everyday examples include shouting, clapping your hands, tapping your feet to music. The louder the sound, the greater the energy within the sound waves produced. This potential energy is what allows sound to propagate and be perceived.
Conclusion
In conclusion, the potential energy of a system is determined by its configuration and the forces acting on it. We explored several types of potential energy that demonstrate this.
Gravitational potential energy depends on the height of an object in a gravitational field. Objects at higher elevations have more gravitational potential energy.
Elastic potential energy is stored in deformed elastic materials like springs. More deformation leads to more elastic potential energy.
Chemical potential energy depends on the types of bonds and molecules in a substance. Some chemical configurations have higher energy than others.
Nuclear potential energy arises from the binding forces within an atomic nucleus. Heavy, unstable nuclei have more potential energy that can be released in nuclear reactions.
Electrostatic potential energy comes from electric charge separation. Bringing opposite charges closer together releases this energy.
Magnetic potential energy results from aligning magnetic dipoles against an external magnetic field. Flipping their alignment releases the stored energy.
Thermal energy depends on the kinetic energy of particles in a substance. Heating increases this random motion, raising the thermal potential.
Sound energy is stored in high-pressure compression waves. This potential propagates through mediums as the energy is released.
In all cases, the potential energy results from the configuration of the system and the forces acting on it. Understanding these relationships allows us to harness potential energy to do useful work.