What Is Also Known As Potential Energy?
What is Potential Energy?
Potential energy is the stored energy an object has due to its position or state. It refers to the capacity for doing work that an object possesses because of its position relative to other objects, stresses within itself, its electric charge, or other factors.
Potential energy can be thought of as energy that is waiting to be released or used. In contrast, kinetic energy is the energy of motion that objects possess because of their velocity. Potential energy is energy that an object could potentially exhibit due to its structure or position.
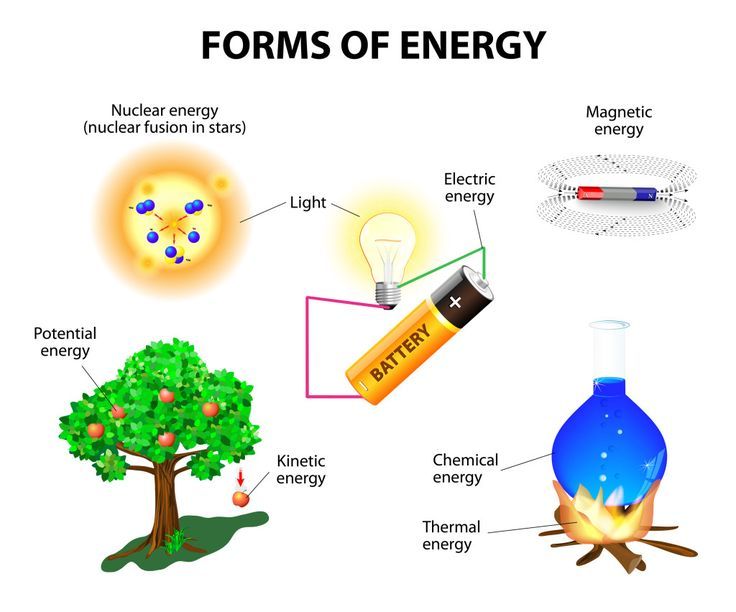
Some common forms of potential energy include:
- Gravitational potential energy – energy stored in an object due to gravity and height.
- Elastic potential energy – energy stored in a compressed or stretched elastic material.
- Chemical potential energy – energy stored in chemical bonds.
- Nuclear potential energy – energy stored in the nucleus of an atom.
Potential energy is converted into kinetic energy when the object starts moving and exerting force. A rock at the edge of a cliff has potential energy due to gravity. When it falls, this potential energy gets converted into kinetic energy from its motion and acceleration due to gravity.
Forms of Potential Energy
There are several main forms of potential energy, based on the different forces or systems involved:
Gravitational Potential Energy
Gravitational potential energy depends on the height of an object above the ground. The higher an object is raised, the more potential energy it has. This energy gets converted to kinetic energy as the object falls.
Elastic Potential Energy
Elastic potential energy refers to energy stored in elastic materials or objects that are deformed. For example, a stretched rubber band or compressed spring has elastic potential energy.
Chemical Potential Energy
Chemical potential energy is energy stored in the chemical bonds within molecules and compounds. This energy can be released in chemical reactions when the bonds are broken.
Electrical Potential Energy
Electrical potential energy results from the separation of positive and negative charges, such as in batteries. The buildup of charge separation contains energy that can be put to use.
Nuclear Potential Energy
Nuclear potential energy is the energy stored within the nucleus of an atom, related to the binding energy of the nuclear particles. It is released in nuclear fission and fusion reactions.
Gravitational Potential Energy
Gravitational potential energy depends on the height of an object above the ground. The higher the object, the more potential energy it has since there is more energy available to do work when it falls. The gravitational potential energy of an object can be calculated using the formula:
PEgrav = mgh
Where m is the mass of the object, g is the gravitational acceleration (9.8 m/s2 on Earth), and h is the height of the object above the ground. Gravitational potential energy is commonly seen with objects on top of cliffs, buildings, or hills. The higher it is initially, the more energy is released as it falls downward and converts to kinetic energy.
Elastic Potential Energy
Elastic potential energy is stored in springs and elastic materials that are deformed or stretched from their natural state. For example, when you stretch a rubber band, you are storing elastic potential energy in it. The more the rubber band is stretched, the more elastic potential energy is stored.
The formula for elastic potential energy is:
Elastic Potential Energy = 1/2 k x2
Where k is the spring constant, and x is the displacement of the spring from its rest position. The spring constant measures how stiff and resistant to stretching the spring is.
Some common examples of elastic potential energy include:
- Stretching a rubber band
- Pulling back the string of a bow
- Compressing a spring
- Stretching a bungee cord
- Bending flexible materials like bamboo
In all these examples, the elastic material is deformed in some way, storing energy that can later be released when the material returns to its original shape.
Chemical Potential Energy
Chemical potential energy is the energy stored in the bonds between atoms and molecules. It is the energy required to break these chemical bonds. When chemical bonds form between atoms and molecules, energy is released, and this energy can be stored and later released when the bonds break again.
Chemical potential energy is converted into kinetic energy when a chemical reaction occurs. Chemical reactions involve breaking and reforming chemical bonds between atoms and molecules. For example, when wood burns, the organic molecules that make up wood break apart into simpler molecules like carbon dioxide and water vapor. This releases energy in the form of heat and light.
Common examples of stored chemical potential energy include:
- Fossil fuels like coal, oil, and natural gas
- Batteries
- Food and organic compounds
- Explosives and fireworks
The chemical potential energy stored in substances like gasoline and batteries can be released on demand to do useful work. For example, the controlled burning of gasoline in car engines converts the chemical energy into kinetic energy that propels the vehicle. Batteries use electrochemical reactions to produce an electric current that can power devices.
Electrical Potential Energy
Electrical potential energy refers to the energy stored in electric fields. This energy results from the presence of charged particles and their interaction with electric fields. When charges build up in capacitors or other components of electrical circuits and devices, they gain the potential to do work. The amount of electrical potential energy that can be stored depends on the voltage applied to the system.
Capacitors are a key example of storing electrical potential energy. Capacitors contain two conductors or plates separated by an insulator. When connected to a battery or other power source, electrons accumulate on one plate, resulting in a negative charge, while the other plate loses electrons and develops a positive charge. This separation of charge establishes an electric field between the plates. The higher the voltage applied, the greater the charge buildup and electric field strength. The capacitance of the capacitor, which depends on its physical properties and geometry, also affects how much charge can be stored at a given voltage.
The energy stored in a capacitor is proportional to the square of the voltage across the device. This relationship is described by the equation E = (1/2)CV2, where E is energy, C is capacitance, and V is voltage. Even after a capacitor is disconnected from a voltage source, it will retain its charge and the accompanying electrical potential energy.
Other devices and components that separate electric charges can also store electrical potential energy. This includes Van de Graaff generators, which use moving belts to accumulate large amounts of static electricity. Lightning rods provide a path for charges to move between clouds and the ground, preventing the further buildup of high voltages.
In summary, electrical potential energy exists whenever there is separation of electric charge. This energy can be stored in capacitors, generators, and other electrical systems and tapped to do useful work. The amount of energy available depends on the voltage applied and the system’s capacitance.
Nuclear Potential Energy
Nuclear potential energy refers to energy stored in the nuclei of atoms. It depends on the binding energy that holds the protons and neutrons together in the atomic nucleus.
Nuclear potential energy can be released when heavy nuclei undergo nuclear fission (splitting) or light nuclei undergo nuclear fusion (joining). In both nuclear fission and fusion, the products have less total binding energy than the reactants, and this mass difference is converted into energy according to Einstein’s equation E=mc^2.
The amount of potential energy released depends on the specific nuclei involved. Fission of uranium-235, for example, releases over 200 MeV per atom. Fusion of hydrogen to form helium releases about 17 MeV per reaction. This large amount of energy released makes nuclear potential energy highly useful for power generation in nuclear reactors and weapons.
However, the precise control and containment required to harness nuclear potential energy also makes it potentially dangerous. Safety precautions are necessary when handling radioactive materials capable of such potent energy release.
Calculating Potential Energy
The calculation of potential energy depends on the specific type and situation. However, there are some general principles for calculating potential energy:
Use the appropriate formula for the situation. For example, gravitational potential energy can be calculated using mgh, where m is mass, g is the gravitational acceleration constant, and h is height. The formula PE=1/2kx^2 can be used to calculate the elastic potential energy of a spring, where k is the spring constant and x is the displacement distance.
Input the values into the formula based on the specific system. This may include the mass of an object, its height above a reference point, the amount of compression or stretching of a spring, the electric charge, or other relevant values. Make sure to use the correct units.
Convert units appropriately to be consistent. For gravitational potential energy using mgh, the mass would need to be in kg, acceleration in m/s^2, and height in m. Make conversions to maintain logical units.
Calculating potential energy requires identifying the correct formula for the situation, plugging in the appropriate values based on the system, and handling units properly. With the right inputs, potential energy can be calculated for a variety of scenarios.
Potential vs Kinetic Energy
Potential energy is energy that is stored within an object due to its position or configuration. For example, a book sitting on a table has gravitational potential energy because gravity is exerting a force on it, keeping it in place. The book has the potential to gain kinetic energy if it falls off the table.
Kinetic energy is energy that an object possesses due to its motion. For example, a moving car has kinetic energy. The faster the car moves, the more kinetic energy it has. Kinetic energy and potential energy can convert back and forth between different forms. When the book falls off the table, its potential energy is converted into kinetic energy as it accelerates towards the ground due to gravity. The kinetic energy can then be converted back into potential energy if the book lands on another surface and comes to rest.
The interconversion between potential and kinetic energy is a key principle behind simple machines and many other mechanical systems. An understanding of these two forms of energy and how they relate is fundamental to physics and engineering.
Applications of Potential Energy
Potential energy has many important real-world applications including:
Hydroelectric power which converts the potential energy of elevated water into electricity. Dams store water at a height, and the potential energy gets converted to kinetic energy as the water falls and turns turbines to generate power.
Launching rockets and enabling space exploration. Rockets are able to leave Earth’s atmosphere by converting chemical potential energy into kinetic energy.
Many renewable energy sources like wind and solar rely on potential energy being converted into electricity. The wind turns turbine blades and the sun’s rays excite electrons in solar panels – in both cases transforming potential energy into kinetic energy and then electricity.