Why Is Energy Never Created Or Destroyed?
The Law of Conservation of Energy
The law of conservation of energy is one of the fundamental laws of physics. It states that the total energy of an isolated system remains constant and is said to be conserved over time. This law means that energy can neither be created nor destroyed – only converted from one form of energy to another.
The law of conservation of energy was first proposed in the 19th century by several scientists including Julius Robert von Mayer, James Prescott Joule, and Hermann von Helmholtz. It is based on the observation that in isolated systems, the sum of all forms of energy, including potential energy, kinetic energy, thermal energy, and others, remains constant. This principle has proven to be universally applicable and is considered to be one of the most fundamental laws governing the universe.
The law of conservation of energy is important because it allows us to quantitatively calculate changes in energy during physical and chemical interactions. We know that the energy before and after will always remain the same, even as it transforms from one type to another. This helps physicists, chemists, engineers, and other scientists to analyze systems and processes where energy is being transferred or transformed.
The conservation of energy has several important implications. For one, it tells us that perpetual motion machines are impossible, since they would have to continually produce energy from nothing. It also implies that there is a finite amount of energy in the universe, and new energy cannot be created out of nothing.
Forms of Energy
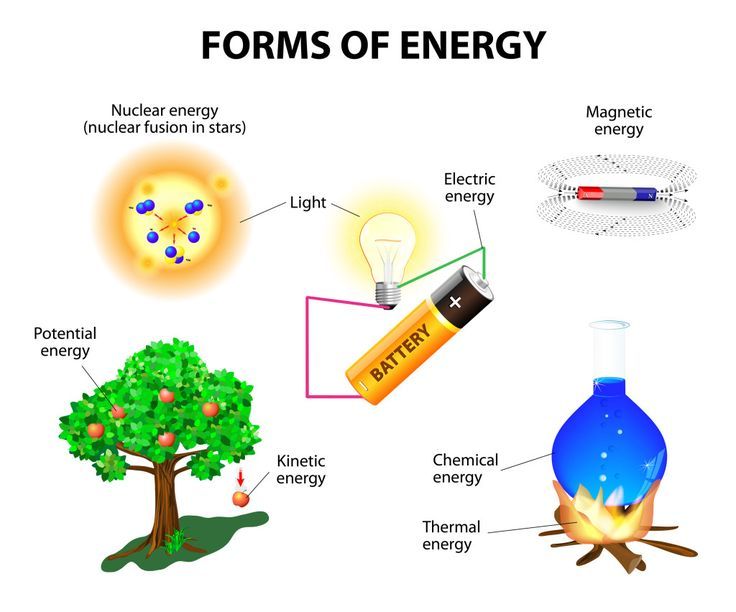
Energy comes in many different forms that can be grouped into two main categories: potential energy and kinetic energy. Here are some of the most common forms that energy takes:
Potential Energy
Potential energy is stored energy that has the potential to do work. Types of potential energy include:
- Chemical Energy – The energy stored in the bonds between atoms and molecules. Examples include batteries, food, fuel, and dynamite.
- Nuclear Energy – The energy stored in the nucleus of an atom. Examples include nuclear power plants that split uranium atoms.
- Gravitational Potential Energy – The energy of an object due to its height above the ground. A ball held in the air has more potential energy than a ball sitting on the ground.
- Elastic Potential Energy – The energy stored in stretched or compressed materials. Examples include stretched springs and rubber bands.
Kinetic Energy
Kinetic energy is energy of motion. Types of kinetic energy include:
- Radiant Energy – The energy carried by electromagnetic radiation such as light, radio waves, gamma rays, and more. Sunshine is a common example.
- Thermal Energy – The total kinetic energy of all the molecules in an object. The higher the temperature, the greater the thermal energy.
- Motion Energy – The energy possessed by a moving object. The faster it moves, the more motion energy it has.
- Sound Energy – Caused by the vibration of molecules, transmitting in waves.
These are the main types of energy that are constantly being converted from one form to another in the universe.
Energy Transformations
Energy can change from one form into another. Some common examples of energy transformations include:
-
Chemical energy in food transforming into kinetic energy as an animal moves
-
Radiant energy from the sun transforming into chemical energy in plants through photosynthesis
-
Mechanical energy from wind transformed into electrical energy in a wind turbine
-
Nuclear energy in an atom transformed into thermal energy and electromagnetic radiation energy during nuclear fission
-
Chemical energy in gasoline transforming into thermal energy and kinetic energy as an engine runs
In all of these examples, when energy transforms from one form into another, the total amount of energy remains constant. Energy is never created or destroyed, just changed between different forms.
Measuring Energy
Energy is quantified and measured using a standard unit called the joule. The joule allows us to numerically define how much energy is contained within a system or transferred between systems. For example, when an apple falls from a tree branch to the ground, it loses potential energy but gains kinetic energy. The total quantity of energy remains constant, but the type of energy changes. We can measure the potential energy of the apple using its mass, gravity, and height off the ground. We can measure its kinetic energy using its mass and velocity right before hitting the ground. The total joules will be equal before and after the fall, demonstrating the law of conservation of energy.
There are other derived units that measure energy as well, like kilojoules, megajoules, and kilowatt-hours. But the joule is the base SI unit. No matter what form energy takes – chemical, electrical, thermal, mechanical, etc. – it can be quantified in joules. This allows us to mathematically balance energy within a defined system and verify that the total amount does not increase or decrease, only convert between different types.
Energy in a Closed System
The law of conservation of energy states that within a closed system, the total amount of energy remains constant. Energy is never created or destroyed, but only converted from one form to another. For example, chemical energy in gasoline can be converted to kinetic energy in a car engine, while friction converts some of that kinetic energy into thermal energy. The total amount of energy before and after these transformations is the same. This principle applies to all closed systems, where no energy enters or leaves.
In an isolated system with no external influence, the initial amount of energy will always equal the final amount of energy, regardless of any changes that occur internally. If 100 units of energy go into a closed system, 100 units must still exist after any conversions. Energy is conserved at all scales, from chemical reactions to biological processes to the mechanics of the entire universe.
The conservation of energy is a fundamental law of nature that has been repeatedly validated through experimentation and calculations. It allows us to track how energy flows and changes form within a system. This principle guides scientific work across many fields, informs how we harness energy sources on Earth, and sheds light on our universe’s origins and workings.
Real World Examples
The law of conservation of energy can be seen in action across many real world systems and processes. Here are a few examples:
Vehicles: The chemical potential energy stored in gasoline is converted into kinetic energy that propels the vehicle forward. Some energy is lost as heat and sound, but the total energy of the system remains constant.
Food metabolism: When food is digested, the chemical potential energy stored in molecules is released and converted into kinetic energy that powers our bodies. The total energy remains constant even as energy changes form.
Hydropower: The potential energy of water held behind a dam is converted into electricity as the water flows through turbines. Friction and heat dissipate some energy, but the total energy balance is maintained.
Solar cells: Photons from sunlight hit solar panels and their energy is converted into electrical current. Though the efficiency isn’t 100%, the net energy is conserved.
Photosynthesis: Radiant light energy is converted and stored as chemical potential energy in plant matter. The total energy remains the same despite the transformation.
Implications and Impact
The law of conservation of energy has profound implications for sustainability and the future of our planet. Since energy cannot be created or destroyed, only transformed, the energy we use today originates from resources that took considerable time to form. Fossil fuels like oil, coal, and natural gas were created from plants and animals that lived hundreds of millions of years ago. Renewable energy sources like sunlight, wind, and hydropower also take time to be replenished.
This means the energy resources we rely on are finite and limited in supply. As the global population increases and countries develop further, our energy demand continues growing rapidly. But the finite reserves of fossil fuels are being depleted much faster than new ones are being created. Renewables can help meet our energy needs sustainably, but only if we use them at a rate matching their replenishment.
The conservation of energy highlights the need to transition to renewable sources and be far more efficient in our energy use. Wasteful consumption of limited resources threatens energy security for future generations and leads to problems like pollution, habitat destruction, and climate change. But by harnessing energy more thoughtfully and sustainably, we can ensure adequate energy to support development while protecting the environment.
Exceptions and Clarifications
While the law of conservation of energy states that energy cannot be created or destroyed in an isolated system, there are some notable exceptions and clarifications.
The most significant exception is when matter is converted into energy, as described by Albert Einstein’s famous equation E=mc2. Here, mass (m) can be converted into energy (E),
with the conversion factor equal to the speed of light (c) squared. This occurs during nuclear reactions, where a small amount of mass is converted into a tremendous amount of energy.
Additionally, the law strictly applies to isolated systems with no external interactions. In reality, most systems interact with their surroundings to some degree, so energy can still be lost or gained. However, the total energy of the system plus surroundings remains constant.
Lastly, the law may not hold true at the quantum level or within extreme gravitational fields like black holes. Here the laws of physics get murky, though the total energy of the universe still appears to remain constant.
So while the law has some exceptions, it remains a fundamental principle for describing energy transformations in our everyday world.
Practical Applications
The law of conservation of energy has many practical applications in fields like engineering and technology. Engineers apply the law when designing things like engines and batteries. Knowing that energy can only change forms but not be created or destroyed helps them maximize efficiency. For example, a combustion engine converts the chemical energy in fuel into kinetic energy that moves the vehicle. Engineers try to reduce wasted heat and friction to get as much of the fuel’s energy into the kinetic energy of motion.
Renewable energy sources like solar, wind and hydropower also rely on the law of conservation. Solar cells convert photon energy from the sun into electric current. Wind turbines change kinetic wind energy into rotational energy to turn a generator. Hydroelectric dams use the gravitational potential energy of elevated water to drive turbine generators. The law of conservation governs how much useful energy can be extracted from these sources.
The law applies to everyday devices too. Batteries, for example, store chemical energy and convert it into electrical energy to power devices. The energy output can never exceed the energy stored initially as chemical energy. Knowing this, battery manufacturers optimize materials and design to maximize efficiency within this limit. Appliances like light bulbs also obey the law—the energy an incandescent bulb uses and gives off as heat and light is equal to the electrical energy it draws. The bulb filament acts to transform the electrical energy into light and heat.
Understanding that energy only changes form but is never destroyed helps inform the design of technology to utilize energy sources effectively and efficiently.
Summary
The law of conservation of energy states that energy can neither be created nor destroyed, only transformed from one form to another. This is one of the most fundamental laws of physics and it applies to closed systems where no energy enters or leaves.
There are many forms of energy such as kinetic, potential, thermal, chemical, nuclear and more. The total energy in a closed system remains constant, even as energy transforms between different types. For example, when a ball falls, its potential energy transforms into kinetic energy. Energy is never “used up” but simply changes form.
This law allows us to quantify energy transfers and transformations. Measuring the energy before and after a process reveals that the amounts are equal, even though the form of the energy has changed. This principle governs all interactions and processes, from chemical reactions to biological processes.
The law of conservation of energy has profound implications. It tells us that energy resources can be transformed for human needs, but never replenished. It also enables technologies like generators and batteries. This fundamental law has shaped our understanding of physics, chemistry and biology.