What Are The Differences Between Heat And Energy?
Defining Heat
Heat is a form of energy that is transferred between objects or systems due to a temperature difference. When two objects at different temperatures come into contact, heat flows from the warmer object to the cooler one until they reach thermal equilibrium.
On a microscopic level, heat is the kinetic energy of atoms and molecules. As the atoms and molecules vibrate and move, they collide with one another and transfer some of their kinetic energy. The faster the atoms move, the higher their kinetic energy and temperature. Heat flows spontaneously from higher temperature to lower temperature objects as the higher energy atoms collide with the lower energy atoms, increasing their kinetic energy.
While related, heat and temperature are distinct concepts. Temperature measures the average kinetic energy of particles in a substance while heat is the total kinetic energy of the particles that can be transferred. An object may have a high temperature but little heat energy to transfer if it has few particles.
Heat is measured in joules (J) or calories (cal). One calorie is the amount of heat needed to raise one gram of water by 1°C. One joule is the amount of work needed to produce one watt of power for one second. The standard unit of heat in the International System of Units (SI) is the joule.
Defining Energy
Energy is the ability to do work or produce heat. More specifically, energy is defined as the capacity to move or change matter. Energy comes in many different forms such as kinetic, potential, thermal, chemical, nuclear and more. Some common examples are the energy in moving objects, stored energy in batteries or gasoline, heat from the sun or a fire, energy locked in the bonds of molecules, and energy released in nuclear reactions.
On a fundamental level, energy allows things to happen or change in our universe. All matter contains energy, and energy flows between objects through radiation, heat transfer or work. This exchange of energy drives essentially all physical, chemical and biological processes. Conservation of energy is a fundamental law of physics which states that energy can change form, but it cannot be created or destroyed.
In summary, energy refers to the capacity to do work and bring about change. It exists in many forms across all matter and drives processes in our universe through transfer and transformation.
Forms of Energy
Energy comes in many different forms that can be categorized into two main types – potential energy and kinetic energy. Here are some of the most common forms that energy takes:
- Potential Energy
- Chemical – Energy stored in the bonds between atoms and molecules. Examples are batteries, food, fuel.
- Nuclear – Energy stored in the nucleus of an atom. Nuclear power plants split atoms to release this energy.
- Gravitational – Energy of an object above ground that can be released when it falls. Hydroelectric dams use the gravitational energy of falling water.
- Elastic – Energy stored in compressed or stretched objects. Springs and stretched rubber bands have elastic potential energy.
- Kinetic Energy
- Radiant – Electromagnetic energy moving through space. Light is a form of radiant energy.
- Thermal – The kinetic energy of atoms and molecules in matter. The higher the temperature, the greater the thermal kinetic energy.
- Motion – The energy of a moving object. The faster it moves, the more kinetic energy it has.
- Electrical – The movement of electrons. Lightning is an example of electrical kinetic energy.
Relationship Between Heat and Energy
While heat and energy are related concepts, they have distinct differences. Heat refers specifically to the transfer of thermal energy between objects or systems. Thermal energy is the internal energy within a system due to the kinetic motion and potential of particles. Heat is a mechanism or process that transfers thermal energy from one object or system to another.
Heat always flows spontaneously from an object at a higher temperature to an object at a lower temperature. The transfer of energy continues until both objects reach the same temperature and are in “thermal equilibrium.” This heat transfer happens through different processes like conduction, convection, and radiation. So while heat itself is not a form of energy, it describes the energy transfer between systems.
The main distinction is that energy is an extensive property of a system tied to the motion and configuration of particles. Heat is the transfer of that energy driven by temperature differences. Energy is contained within a system, while heat flows between systems. Recognizing the nuanced relationship between heat and energy is key to understanding thermodynamics.
Measuring Heat vs Energy
While heat and energy are related concepts, they are measured using different units. Heat is measured in calories or joules, which indicate the amount of thermal energy present. Common units used to measure heat include:
- Calorie (cal) – The amount of heat required to raise 1 gram of water by 1°C.
- Kilocalorie (kcal) – Equal to 1,000 calories.
- BTU (British Thermal Unit) – The amount of heat required to raise 1 pound of water by 1°F.
- Joule (J) – The SI unit for all forms of energy, including heat. 1 cal = 4.184 J.
Energy is measured in units that quantify the ability to do work, such as kinetic energy, potential energy, chemical energy, etc. Common units used to measure energy include:
- Joule (J) – The standard SI unit of energy.
- Kilojoule (kJ) – Equal to 1,000 joules.
- Kilowatt-hour (kWh) – A unit of electrical energy equal to the work done by 1 kilowatt acting for 1 hour.
- Calorie (cal) – While sometimes used to measure energy, the calorie specifically measures heat energy.
So while related, heat and energy use different base units for measurement that quantify different characteristics.
Heat Transfer
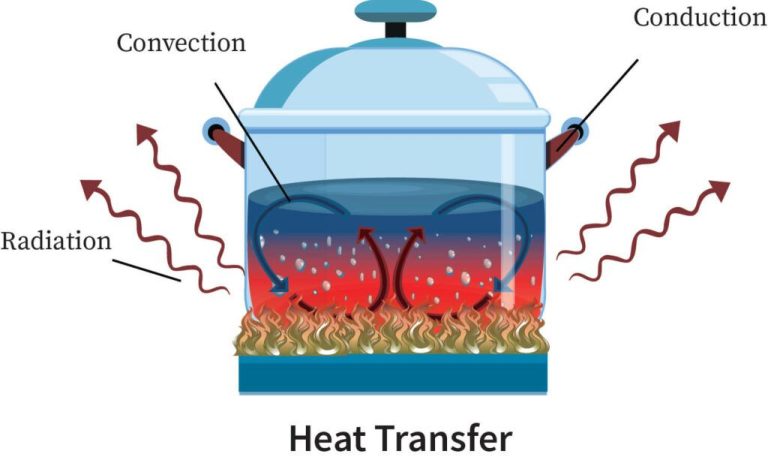
There are three main ways that heat energy is transferred from one place or material to another: conduction, convection, and radiation. These are:
Conduction
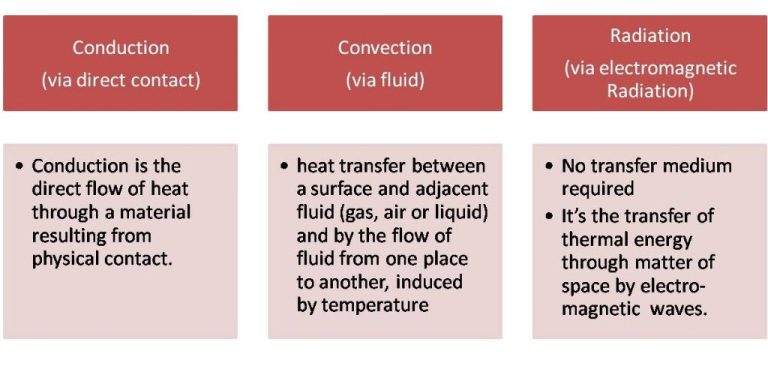
Conduction is the transfer of heat between two objects in direct contact with each other. The heat energy is transferred between molecules when they collide. Metals are good conductors because their atomic structure allows heat to easily flow. Insulators with more complex molecular structures are poor at conducting heat.
Convection
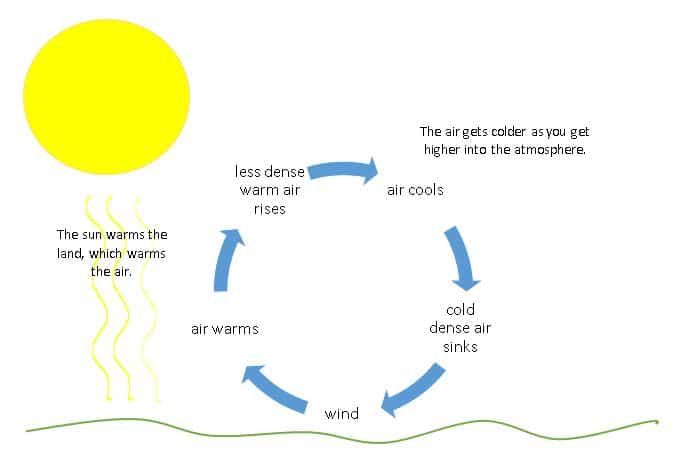
Convection is the transfer of heat by the movement of heated fluid particles (liquid or gas). For example, as the fluid is heated, it expands and becomes less dense than the fluid around it, causing it to rise. As the heated fluid rises, cooler fluid takes its place, creating a continuous circulation of heat transfer through convection currents.
Radiation
Radiation is the transfer of heat via electromagnetic waves or photons. An example is the heat from the sun warming the Earth. The heat we feel from a fire is also radiant heat. Unlike conduction and convection, radiation can transfer heat across a vacuum without relying on molecular contact or fluid movement. All objects constantly emit thermal radiation related to their temperature.
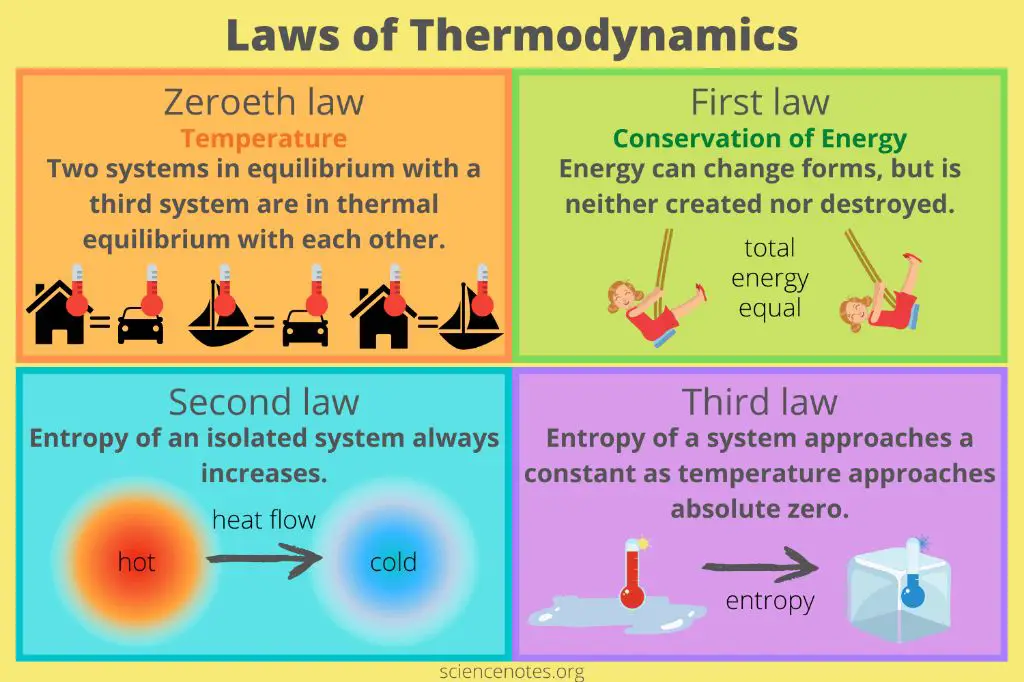
Laws of Thermodynamics
The laws of thermodynamics define the relationship between heat and energy. There are three major laws:
First Law of Thermodynamics: This law states that energy can neither be created nor destroyed, it can only be transformed from one form to another. For example, when you burn a piece of wood, the chemical energy stored in the wood transforms into heat and light. The total energy in the system remains constant.
Second Law of Thermodynamics: This law states that heat will always flow spontaneously from a hotter to a colder body. Thermal equilibrium is reached when two bodies are at the same temperature and no more heat transfer can occur. The second law also introduces the concept of entropy – disorder tends to increase in an isolated system over time.
Third Law of Thermodynamics: This law states that it is impossible to reduce the temperature of a system to absolute zero in a finite number of steps. While you can get infinitesimally close to absolute zero, you can never actually reach it.
The laws of thermodynamics describe fundamental physical principles that govern the relationship between heat and energy. These laws are critical to understanding thermodynamics and how energy flows and transforms.
Examples
Practical examples that demonstrate the difference between heat and energy include:
A stove burner emits heat which is transferred to a pot of water, adding thermal energy to the water and increasing its temperature. The flame on the stove and the heating element contain chemical potential energy that is converted into heat.
The sun emits radiant heat and light which warms your skin on a summer day. The radiant energy from the sun originated as nuclear energy generated by fusion reactions within the sun.
When you rub your hands together briskly, the friction converts your body’s chemical energy into heat, warming your hands. The faster you rub them, the more heat is produced.
A light bulb converts electrical energy into radiant energy in the form of visible light and heat. The light bulb gets hot from the thermal energy it emits.
Metabolic processes within the human body convert chemical energy from food into heat that maintains body temperature. Exercising generates more heat as metabolism increases.
Fossil fuels like coal and oil contain stored chemical energy that can be converted into heat through combustion, such as burning coal to boil water into steam to run a steam turbine.
Common Misconceptions
There are several common misconceptions that people have about the differences between heat and energy:
Heat is a form of energy – This is not correct. While heat transfer does involve the transfer of energy, heat itself is not a form of energy. Heat is the transfer of thermal energy between objects with different temperatures.
Heat and temperature are the same thing – Heat and temperature are related, but not the same. Temperature measures the average kinetic energy of molecules and atoms in a substance. Heat relates to the total thermal energy of the system.
Objects contain heat – Objects do not contain heat, they contain thermal energy. Heat is energy in transit due to temperature difference. It flows between objects, but is not contained within an object.
“Coldness” is the opposite of heat – There is no such thing as “coldness.” Cold is simply the absence of heat. So coldness cannot be transferred between objects like heat can.
Heat rises – While it’s true that hot air rises, heat itself does not necessarily rise. Heat can be transferred in any direction, depending on the temperatures of the objects involved.
Heat and temperature are proportional – The amount of heat transferred does not depend only on temperature change. It also depends on the mass and material properties of an object.
Summary
To recap some of the key differences between heat and energy:
– Heat is a form of energy transfer between objects or systems due to temperature difference. Energy is the capacity to do work and can take many forms (kinetic, potential, chemical, etc).
– While heat is measured in calories or joules, energy is measured in joules. Heat is a process whereas energy is more of a property.
– The laws of thermodynamics describe the relationship between heat and other forms of energy. The first law states that energy can be transferred as heat or work. The second law states that heat flows spontaneously from higher to lower temperatures.
– Common misconceptions are that heat and thermal energy are the same thing (heat is a transfer process), or that measuring the total energy accounts for all heat (some energy may become unavailable to do work).
In summary, heat and energy are related but distinct concepts in thermodynamics. Understanding the key differences allows us to accurately describe thermodynamic processes.