What Are Energy Terms In Chemistry?
Energy is a fundamental concept in chemistry that describes the capacity to do work and drive chemical reactions. Energy comes in different forms that can be categorized into two main types: kinetic energy and potential energy.
Kinetic energy is the energy associated with motion. At the molecular level, kinetic energy refers to the movement of atoms and molecules. The faster a molecule moves, the more kinetic energy it possesses.
Potential energy is stored energy that results from the position or structure of a molecule. Atoms that are pulled apart or pushed together have potential energy due to their separation distance and electrostatic interactions. The structure of a molecule, such as the bonds between atoms, also gives rise to potential energy.
The interconversion between kinetic and potential energy drives chemical reactions. Exothermic reactions involve the release of energy, resulting when the total energy of the products is lower than the reactants. This liberated energy is usually in the form of heat. Endothermic reactions absorb energy, occurring when the products have higher total energy than the reactants. This required energy input often comes in the form of heat.
Understanding the energy changes involved in chemical reactions provides chemists with valuable insights into reactivity and thermodynamics.
Kinetic Energy
Kinetic energy is the energy possessed by an object in motion. It is directly proportional to the mass of the object and the square of its velocity. Kinetic energy is one of the most fundamental concepts in chemistry, as it is closely tied to the motion and collisions of atoms and molecules.
The kinetic energy (Ek) of an object can be calculated using the following equation:
Ek = 1/2 mv2
Where m is the mass of the object and v is its velocity. This shows that an object with more mass moving at higher velocity will have greater kinetic energy.
Examples of kinetic energy on the molecular level include:
- Atoms and molecules moving and vibrating
- Collisions between reactant molecules
- Electrons moving around the nucleus
Kinetic energy is directly related to temperature. Higher temperature indicates greater molecular motion and kinetic energy. Heat is the transfer of kinetic energy between objects, flowing from higher to lower kinetic energy. Therefore, kinetic energy is a key factor governing chemical kinetics and thermodynamics.
Potential Energy
Potential energy is the energy that is stored within a system. It refers to the potential of an object or system to do work because of its position or state. Potential energy exists when there is a force between two objects that depends on the position or shape of the objects. When the position of the objects change, the force can do work.
Some examples of potential energy include:
- Chemical potential energy stored in the bonds between atoms and molecules. When chemical bonds are broken or formed, energy is released or absorbed.
- Gravitational potential energy from an object’s height relative to Earth. As an object falls, gravitational potential energy is converted into kinetic energy.
- Elastic potential energy stored in compressed or stretched springs, rubber bands, etc. This energy can be released when the tension is released.
- Nuclear potential energy from the forces between protons and neutrons in the nucleus of an atom. This energy can be released in nuclear fission or fusion.
Potential energy is a fundamental energy term in chemistry because the structure and reactivity of molecules depend heavily on the potential energy stored in chemical bonds. The making and breaking of chemical bonds during chemical reactions involves conversions between potential and kinetic energy.
Exothermic Reactions
In an exothermic chemical reaction, more energy is released during the reaction than it takes to initiate it. Exothermic reactions are marked by the release of energy in the form of heat and/or light. These types of reactions are very common and play an important role in both nature and industry.
The key feature of an exothermic reaction is that the products formed have less energy than the reactants. This releases energy, usually in the form of heat, but sometimes also as light. A classic example of an exothermic reaction is the burning of fuels like wood, coal, or natural gas. In these reactions, energy stored in the chemical bonds of the fuel is released as heat and light as the fuel combines with oxygen gas.
Other examples of exothermic reactions include:
- Metal oxidation reactions like the rusting of iron or burning of magnesium
- Acid-base neutralization reactions between an acid and a base
- Decomposition reactions like the electrolysis of water into hydrogen and oxygen
- Combustion reactions of hydrocarbons or sugars with oxygen
The energy released by exothermic reactions can be harnessed for helpful purposes. For example, the heat released when fuels undergo combustion reactions powers internal combustion engines and provides warmth through home heating systems. Exothermic reactions also play important roles in chemical processes like the production of cement, iron, steel, and many other materials.
Endothermic Reactions
An endothermic reaction is a chemical reaction that absorbs heat from the surroundings. The energy absorbed is stored as chemical potential energy in the bonds of the products formed. Endothermic reactions feel cold to the touch because heat is removed from the surroundings to provide the energy needed to break bonds in the reactants.
Some examples of endothermic reactions include:
- Thermite reaction between aluminum and iron oxide
- Baking soda and vinegar reaction
- Decomposition of ammonium nitrate into water and nitrogen gas
- Dissolving ammonium chloride in water
In an endothermic reaction, the energy required to break bonds in the reactants is greater than the energy released when new bonds form in the products. This net absorption of energy during the reaction results in the surroundings getting colder. The amount of energy absorbed in an endothermic reaction is called the “heat of reaction.” Endothermic reactions require an input of heat or thermal energy from the surroundings for the reaction to proceed.
Activation Energy
Activation energy is the minimum amount of energy that reactant molecules must possess in order to undergo a chemical reaction. It is often represented by the symbol Ea. Activation energy is required to destabilize bonds within the reactants so that new bonds can form to create the products.
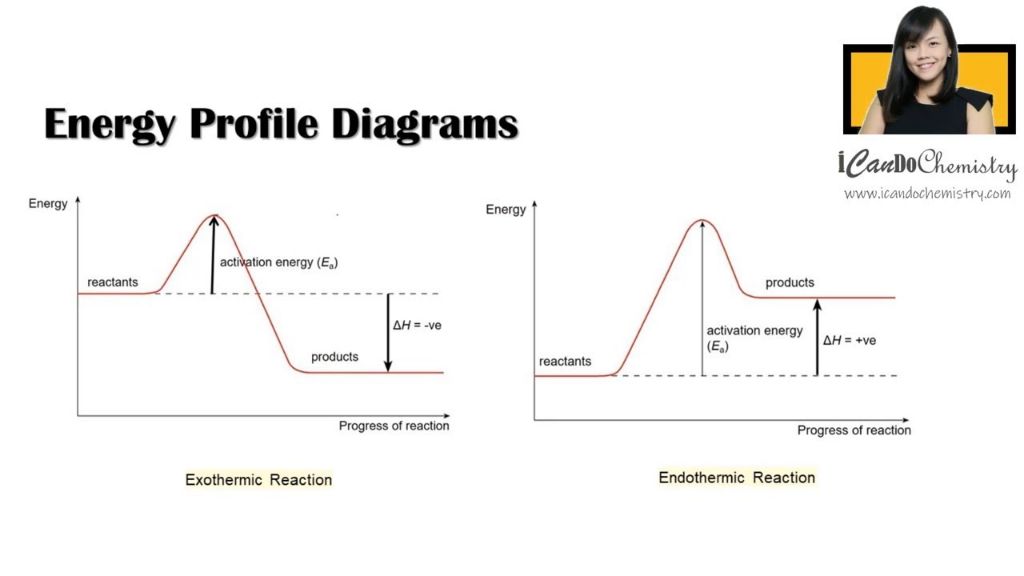
Activation energy plays a key role in reaction kinetics, which is the study of reaction rates. The higher the activation energy, the slower the reaction rate, as more energy must be provided to get the reaction going. Reactions with lower activation energies tend to proceed faster. The Arrhenius equation shows the mathematical relationship between activation energy and reaction rate constants.
Catalysts work by reducing the activation energy required for a reaction. They provide an alternate reaction pathway such that less energy input is needed for the reactants to reach the transition state. Catalysts stabilize transition states and intermediate compounds. By lowering the activation energy barrier, catalysts dramatically increase reaction rates without being consumed in the reaction itself.
Enthalpy
Enthalpy is a thermodynamic property that represents the total heat content of a system. It is defined as the sum of a system’s internal energy plus the product of its pressure and volume. Enthalpy is useful for describing energy transfers in chemical reactions and phase changes.
The change in enthalpy (ΔH) indicates whether a chemical reaction is endothermic or exothermic. An endothermic reaction absorbs heat from the surroundings and has a positive ΔH. This means the enthalpy of the products is higher than the reactants. An exothermic reaction releases heat to the surroundings and has a negative ΔH, meaning the enthalpy of the products is lower than the reactants.
The enthalpy change of a reaction depends on the energies required to break and form chemical bonds. Bond breaking requires energy input (endothermic) while bond making releases energy (exothermic). The magnitude of ΔH depends on the types of bonds being broken and formed.
Entropy
Entropy is a measure of the randomness or disorder of a system. It quantifies the number of specific ways the components of a system can be arranged. The higher the entropy, the greater the disorder and randomness.
For chemical reactions and processes, entropy change is an important thermodynamic property. It provides a quantitative measure of the dispersal of energy that occurs during a reaction. Reactions that proceed with an increase in entropy are favored, as they move the system towards greater randomness and disorder. An increase in entropy provides the driving force that enables many chemical reactions to proceed spontaneously.
The entropy change (ΔS) of a reaction depends on the rearrangement of molecules from reactants to products. If a reaction results in a more disordered system with greater randomness, the entropy increases and ΔS is positive. On the other hand, if a reaction leads to a more ordered system, the entropy decreases and ΔS is negative.
An example is the dissociation of a salt crystal in water. The highly ordered crystal breaks apart into disordered solvated ions, greatly increasing the entropy of the system. The positive ΔS drives the dissociation reaction to proceed spontaneously.
Entropy change can be used along with enthalpy change (ΔH) to determine if a chemical reaction will be spontaneous according to the Second Law of Thermodynamics. Reactions with a positive entropy change tend to be favored, while those with a negative entropy change are not. Considering both ΔH and ΔS provides deeper insights into a reaction’s feasibility.
Gibbs Free Energy
Gibbs free energy is an important thermodynamic function that allows us to determine the spontaneity of a chemical reaction. It is represented by the symbol G and is defined by the following equation:
G = H – TS
Where H is the enthalpy, T is the temperature in Kelvin, and S is the entropy. As we can see from the equation, Gibbs free energy is dependent on both the enthalpy and entropy of the system.
For any chemical reaction, a negative Gibbs free energy change (∆G) indicates that the reaction will proceed spontaneously. This means that the reactants will convert to products without any external energy input needed. On the other hand, a positive ∆G means the reaction is non-spontaneous and will not proceed unless energy is put into the system.
At constant temperature and pressure, the spontaneity of a reaction depends solely on the change in Gibbs free energy. Reactions with a negative ∆G will be spontaneous, while those with a positive ∆G will not occur without an external energy input. This makes Gibbs free energy extremely useful in determining whether a chemical reaction will occur on its own under specified conditions.
Conclusion
To summarize, energy is a fundamental concept in chemistry that helps explain how and why chemical reactions occur. The key energy terms in chemistry include:
Kinetic Energy – The energy associated with the motion of molecules. Higher temperatures correspond to higher average kinetic energy of molecules.
Potential Energy – The stored energy within a molecule, related to its structure and bonds.
Activation Energy – The minimum energy required for a chemical reaction to proceed. Reactions only occur when molecules collide with enough energy to overcome the activation barrier.
Exothermic Reactions – Reactions that release energy, corresponding to lower energy products. The lost energy is transferred as heat to the surroundings.
Endothermic Reactions – Reactions that absorb energy, corresponding to higher energy products. The required energy is absorbed as heat from the surroundings.
Enthalpy – A measure of the total energy content of a system based on its internal energy and pressure-volume work. Enthalpy changes show whether a reaction is exo- or endothermic.
Entropy – A measure of molecular randomness and disorder. Reactions favor an increase in entropy.
Gibbs Free Energy – Used to predict whether a reaction will be spontaneous based on enthalpy and entropy changes. Reactions proceed spontaneously when the Gibbs free energy decreases.
By understanding these key energetic concepts, chemists gain valuable insights into the driving forces behind chemical reactions and the direction in which processes will proceed. Energy is a vitally important foundational topic in the field of chemistry.