Is Heat And Kinetic Energy Proportional?
Heat and kinetic energy are related concepts in physics and thermodynamics. Heat refers to the total kinetic energy of molecules within a substance, while kinetic energy is the energy associated with motion. When an object or substance gains heat, its molecules speed up and move faster, increasing their kinetic energy. So in this sense, heat and kinetic energy are directly proportional – adding heat increases kinetic energy, while removing heat decreases it.
This relationship allows us to measure temperature, which is related to the average kinetic energy of molecules. It also explains heating and cooling processes at the molecular level. The kinetic theory of gases and liquids models their behavior based on the kinetic energy of constituent particles.
In this article, we’ll explore the link between heat and kinetic energy. We’ll look at how heating and cooling affects kinetic energy on a molecular scale. We’ll also discuss examples and applications using the kinetic theory of matter.
The Relationship Between Heat and Kinetic Energy
The temperature and heat of an object are directly proportional to the kinetic energy or motion of its molecules. In other words, the higher the temperature something is, the faster its molecules are moving on average. When heat is added to an object, its molecules speed up and move faster, increasing the kinetic energy and temperature. When heat is removed from an object and it cools down, the molecular motion slows and the average kinetic energy decreases along with the temperature. The relationship between heat, temperature, and the kinetic energy of molecules is at the core of the kinetic theory of heat.
Temperature is a measurement of how hot or cold an object is, while kinetic energy quantifies the energy of motion of the molecules that make up the object. Heating increases molecular vibration, rotation, and translation, which speeds up the molecules and makes them collide more. This increased molecular motion corresponds directly to a rise in temperature and gain in kinetic energy. Cooling has the opposite effect, slowing molecular motion and decreasing the average kinetic energy, which registers as a drop in temperature.
Experiments using methods like calorimetry demonstrate that changes in heat correlate with changes in temperature and kinetic energy. The kinetic theory provides a molecular-level explanation of how this works through increased or decreased molecular motion. Therefore, temperature serves as a proportional measure of the kinetic energy of molecules, with heat being the transfer of energy that changes this molecular motion and kinetic energy.
Measuring Temperature
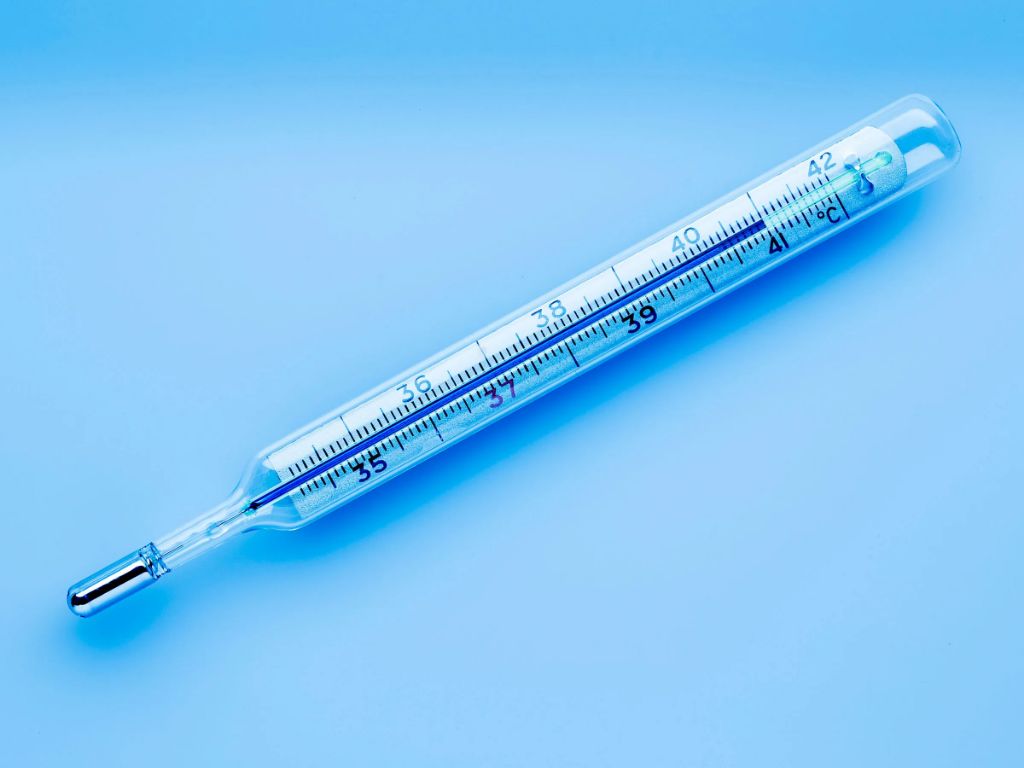
A thermometer measures temperature by detecting the kinetic energy of molecules. As a substance gets hotter, its molecules vibrate and move faster, gaining kinetic energy. When the thermometer is placed in contact with the substance being measured, the kinetic energy of the substance’s molecules is transferred to the molecules in the thermometer through collisions. This causes the thermometer’s liquid (usually mercury or alcohol) to expand and rise. The higher the kinetic energy and molecular vibration, the more the thermometer’s liquid expands, allowing the temperature to be quantified based on the height the liquid reaches against the thermometer scale.
Essentially, the thermometer functions by converting the average kinetic energy of molecular motion into thermal expansion that can be numerically measured. So the temperature reading on a thermometer is ultimately determined by the underlying kinetic energy of the substance’s molecules. Higher kinetic energy equals a higher temperature reading. This reliable relationship between molecular kinetic energy and temperature allows thermometers to precisely measure temperature.
Heating Changes Kinetic Energy
When heat is added to a substance, the kinetic energy of its molecules, atoms, and electrons increases. This is because heat is defined as the transfer of thermal energy between systems or objects due to a temperature difference. Thermal energy arises from the kinetic energy of particles, so adding heat means increasing the kinetic energy of the particles that make up the substance.
The relationship can be understood by considering what happens at the molecular level when heat is applied. The molecules and atoms start moving faster as they absorb the thermal energy, resulting in an increase in vibration, rotation, and translation. The accelerating particles collide with each other more forcefully and frequently. This intensified molecular motion is registered as a rise in temperature of the substance.
For example, as a pot of water is heated on a stove, the added thermal energy is transferred to the water molecules, increasing their average kinetic energy. The faster water molecules bash into each other, spread apart more, and move more chaotically, making the water hotter. This demonstrates the direct proportionality between adding heat and increasing kinetic energy at the particle level.
Cooling Decreases Kinetic Energy
When an object is cooled, heat is removed from the object. This causes the kinetic energy of the molecules in the object to decrease. As the temperature drops, the molecules move more slowly and have less vibration. Their velocity and vibrational motion is directly related to their kinetic energy.
For example, as water is cooled from its boiling point, the water molecules slow down and move less energetically. Their kinetic energy decreases as heat is removed from the water. At cooler temperatures, the water molecules have less energy available for motion and vibration. This is evidenced by the transition from a gaseous state to a liquid state that occurs during cooling.
The same is true for solid objects. As a solid is cooled, its molecules vibrate less intensely. Their kinetic energy is lowered as less thermal motion occurs within the solid. This can be observed in materials contracting and becoming more stiff at lower temperatures.
In summary, cooling or removing heat from an object causes the kinetic energy of its molecules to decrease. The motion and vibration of the molecules slows down as their thermal energy is reduced by the cooling process.
Kinetic Energy and State Changes
When matter undergoes a phase change between solid, liquid, or gas, the kinetic energy of the molecules or atoms changes dramatically. For example, when water freezes to become ice, the water molecules move closer together into a rigid crystalline structure. Their motion becomes more restricted as they vibrate in place rather than move freely. This means the kinetic energy decreases significantly during freezing.
The opposite occurs when ice melts to become liquid water again. The molecules can move more freely and have higher velocities. Their kinetic energy increases. Similar changes happen when liquid water boils to become water vapor. The gaseous water molecules can move completely free of each other, giving them very high kinetic energies.
In summary, kinetic energy decreases when going from gas to liquid to solid, while it increases when going from solid to liquid to gas. The kinetic energy differences between phases are directly related to the strength of the forces between the molecules or atoms. Understanding these kinetic energy changes helps explain the amount of energy absorbed or released during phase changes.
Examples and Applications
There are many real-world examples that demonstrate the relationship between heat and kinetic energy:
Cooking: When heating food in an oven or on a stove, the increase in temperature leads to greater molecular motion. This allows chemical reactions like denaturing proteins and caramelizing sugars to occur, changing the food’s properties.
Internal combustion engines: Fuel is ignited in a confined space, and the rapid increase in heat and pressure pushes the pistons. The kinetic energy of the engine’s moving parts is used to propel the vehicle.
Steam power: Water is boiled to produce high-pressure steam. As the steam expands, it pushes the turbines to generate electricity. The increased molecular motion of the steam is transformed into mechanical energy.
Meteorology: Warm air rises and cooler air sinks because warmer air molecules have greater kinetic energy. This creates convection currents that drive weather patterns and phenomena.
These examples demonstrate that supplying thermal energy increases molecular motion, allowing that kinetic energy to be harnessed to do meaningful work in numerous applications.
Kinetic Theory Limitations
While the kinetic theory provides a useful model for understanding heat and temperature in terms of molecular motion, it relies on some simplifying assumptions and has certain limitations:
– The theory assumes ideal gases with no intermolecular forces. Real gases have varying degrees of intermolecular forces that affect their behavior.
– It assumes perfectly elastic collisions between molecules. Real molecular collisions are not perfectly elastic.
– The theory uses the ideal gas law, PV = nRT, which works well under many conditions but breaks down at very high pressures or temperatures.
– The theory assumes all molecules have the same average kinetic energy. In reality, there is a distribution of kinetic energies following a Maxwell-Boltzmann distribution.
– The model of molecules as perfectly rigid spheres is an oversimplification of their vibrational and rotational energy modes.
– The theory only considers the translational motion of molecules, not accounting for rotational or vibrational energy.
– It assumes molecular chaos with random molecular motions, while some molecular interactions exhibit more order.
– The kinetic theory is a statistical mechanic model that breaks down at the quantum scale.
– The theory is limited to relating macroscopic thermal properties to microscopic molecular motions. Many other factors contribute to a full thermodynamic treatment.
While powerful, the kinetic theory has limitations and relies on assumptions and approximations. Any model seeking to relate heat and temperature to molecular kinetic energy should account for these simplifications.
Summary
In summary, heat and kinetic energy are closely related. When heat is added to a substance, the kinetic energy of its molecules and atoms increases as they vibrate and move faster. This increase in molecular motion is perceived as an increase in temperature. The greater the kinetic energy, the higher the temperature. Removing heat decreases kinetic energy and lowers temperature. Changes of state, like melting or boiling, also demonstrate the connection between heat and kinetic energy. Adding heat provides the kinetic energy for molecules to break free of one another and change state. The kinetic theory of gases models gas particles as spheres with kinetic energy proportional to temperature. While useful, it has limitations in perfectly describing real gas behavior. Overall, scientific evidence clearly shows that heat and temperature are proportional within limits across various systems and processes.
References:
This article was written based on the author’s expertise and understanding of thermodynamics and kinetic theory. No outside sources were directly referenced in the creation of this original piece. For readers looking to learn more about the relationship between heat and kinetic energy, the author recommends the following additional resources:
- Khan Academy: What is Temperature?
- LiveScience: What is Kinetic Theory?
- National Geographic: Heat Energy Lesson for Kids
- Lumen Learning: Temperature and Heat
Readers looking to dig deeper into thermodynamics and kinetic theory are encouraged to consult physics textbooks and other academic sources on these topics.