How Is Heat Made Into Energy?
Heat and energy are two fundamental concepts in physics and engineering. Heat refers to the transfer of thermal energy between substances due to a temperature difference. Energy is the ability to do work or supply heat. The laws of thermodynamics establish the relationship between heat and energy and make it possible to convert heat into other useful forms of energy.
The first law of thermodynamics states that energy can neither be created nor destroyed in an isolated system. Energy can only be transferred from one form to another. The second law of thermodynamics says that heat will always flow spontaneously from a hotter object to a colder object. Using this principle, heat energy can be used to perform mechanical work.
Thermodynamics provides the basis for heat engines and power generation systems that convert heat into electrical or mechanical energy. By harnessing temperature differences, heat energy can be captured and transformed into electricity, motion, and other useful work.
Heat Sources
Heat energy comes from a variety of natural sources that humans have learned to harness and utilize. Some of the primary sources of heat energy are:
Solar Energy
The sun provides a massive and constant supply of thermal energy that reaches the Earth. Solar thermal collectors like solar water heaters and concentrated solar power plants can capture heat from sunlight and convert it into usable energy.
Geothermal Energy
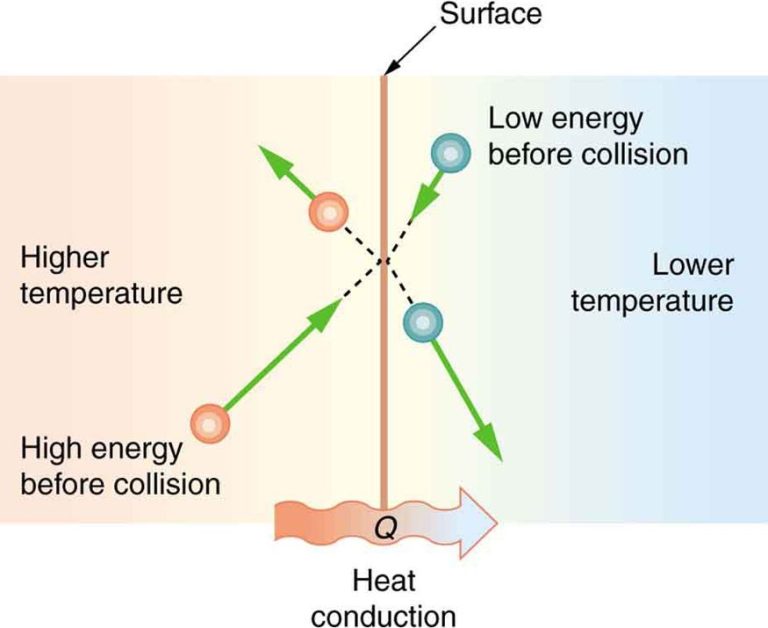
The inner core of the Earth produces a huge amount of thermal energy that gets transferred to the surface through conduction and convection. Geothermal power plants use the naturally heated fluids from deep underground reservoirs to drive turbines and generate electricity.
Fossil Fuels
Fuels like coal, oil and natural gas contain stored chemical energy that gets released as heat when burned. Heat from the combustion of fossil fuels is used to boil water and produce steam that spins turbines connected to generators.
Other heat sources include biofuels derived from plants and waste, nuclear fission reactions in nuclear power plants, and friction heat produced during industrial processes.
Heat Engines
Heat engines are devices that convert heat into mechanical work. They operate on the thermodynamic principle that heat can produce pressure differences that can be used to generate motion.
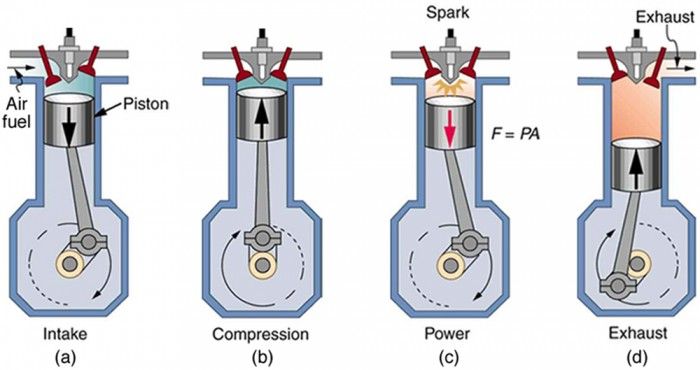
Some common examples of heat engines are internal combustion engines and turbines. In an internal combustion engine, fuel is burned inside a confined space called a combustion chamber. This generates high temperatures and pressures that push a piston to produce rotary motion. The motion is then converted into useful work, like turning the wheels of a car.
In a turbine engine, the high pressure and velocity gas from the combustion is used to spin turbine blades mounted on a shaft. This rotational motion can also be converted into usable work. For example, steam turbines use high pressure steam to spin their blades and generate electricity in power plants.
The key point is that heat engines rely on temperature gradients and pressure differences caused by heating a gas or liquid to harness energy from heat. The mechanical work produced by the engine components like pistons or turbine blades ultimately comes from the heat input into the system.
Thermoelectric Generators
Thermoelectric generators produce electricity directly from heat by converting temperature differences into voltage. This phenomenon is known as the Seebeck effect. Thermoelectric generators work through the interactions between charge carriers in the atomic structure of thermoelectric materials.
Thermoelectric materials are made up of positively charged atoms (holes) and negatively charged electrons that can move through the material. When one end of the thermoelectric material is heated up while the other end remains cool, the electrons and holes start moving. The hot end has more energetic charge carriers, so the electrons and holes spread out and move toward the colder end. This flow of charge carriers creates a voltage difference between the hot and cold ends.
The greater the temperature difference, the larger the voltage generated. Thermoelectric generators consist of multiple thermocouples made from n-type and p-type semiconductor materials that are connected thermally in parallel but electrically in series. This allows the voltage from each thermocouple to add together, producing power that can be used as electricity.
Thermoelectric generators have no moving parts, require no maintenance, and can operate for decades, making them a highly reliable technology for converting heat into electricity using solid-state physics.
Steam Turbines
Steam turbines convert heat energy into rotational kinetic energy through a thermodynamic process. They use high-pressure steam, which carries a large amount of thermal energy, to rotate a turbine and generate mechanical power. This mechanical power can then be used to generate electricity.
Inside a steam turbine, there are stationary and moving blades. The stationary blades are attached to the casing while the moving blades are attached to the rotor shaft. As high-pressure steam passes through the stationary blades, it expands and speeds up, causing the moving blades to rotate. This rotation causes the shaft to spin at high speeds.
The shaft is connected to an electricity generator, so as it spins, it rotates the magnets inside the generator. This motion of the magnets inside wire coils induces a flow of electricity that can then be channeled and used as an energy source.
The speed, pressure and temperature of the steam determines the amount of rotational kinetic energy that can be generated by the steam turbine. Modern steam turbines can operate at tremendous speeds, some up to 3,600 revolutions per minute, helping maximize power output. By optimizing the steam conditions and turbine design, steam turbines are able to convert heat energy into electricity very efficiently.
Heat Exchangers
Heat exchangers are devices designed to efficiently transfer heat between two or more fluids that are at different temperatures. They play a crucial role in many engineering applications by regulating heat transfer in order to achieve desired temperatures and energy efficiency.
The way heat exchangers work is by providing a conductive path between two fluids, allowing the heat from the hot fluid to pass through the conductive walls of the heat exchanger to the cold fluid. This heat transfer occurs without the two fluids having to mix or come into direct contact. The core component of the heat exchanger is often a metal, like steel or aluminum, which has good thermal conductivity to facilitate heat transfer.
There are several types of heat exchangers used for different applications, but they all rely on the basic principle of conductive heat transfer between fluids. Common types include shell and tube heat exchangers, plate heat exchangers, regenerators, condensers and evaporators. The optimal design depends on the types of fluids used, pressures, temperatures, and desired heat transfer rate.
In power generation systems, heat exchangers play a vital role. For example, in nuclear and fossil fuel plants, steam turbines rely on heat exchangers to boil water into steam using the heat from combustion or nuclear fission. The high-pressure steam is then used to turn the turbines and generate electricity. Heat exchangers are also used to condense the steam back into water after passing through the turbine, so it can be reused in the cycle.
In HVAC systems, heat exchangers facilitate heat transfer between indoors and outdoors to heat or cool a space. They are also used in chemical plants, refrigeration, waste heat recovery, and automotive applications like radiators, intercoolers and oil coolers. Advanced heat exchangers continue to be developed to maximize efficiency and heat transfer capabilities. Their role in transferring heat between fluids makes heat exchangers an essential component in many thermal engineering systems.
Power Plants
Power plants utilize various heat sources to boil water into steam, which then spins a turbine to generate electricity. The three main types of power plants are:
Coal Power Plants
Coal power plants burn coal to heat water in a boiler. The steam from the boiler spins a turbine connected to an electrical generator. Coal plants provide about 30% of the world’s electricity. However, burning coal produces air pollution and carbon emissions.
Natural Gas Power Plants
Natural gas power plants burn natural gas in a combustion turbine that is connected to an electrical generator. The leftover heat from the turbine can also produce steam to power a steam turbine for extra electricity generation. Natural gas emits less pollution than coal.
Nuclear Power Plants
Nuclear power plants use the heat from nuclear fission reactions to produce steam. This steam then drives turbine generators to produce electricity. Nuclear provides about 10% of the world’s electricity. While nuclear plants don’t emit air pollution or carbon during operation, concerns remain over radioactive waste disposal and safety.
In summary, power plants allow us to generate massive amounts of electricity from heat produced by burning fossil fuels or through nuclear reactions. However, their environmental impacts must be carefully managed.
Heat Pumps
Heat pumps are devices that move heat from one location to another. They work by taking advantage of the physical properties of refrigerants that allow them to absorb and release heat as they undergo phase changes between liquid and gas. Here’s how heat pumps work:
Heat pumps contain a refrigerant that circulates through indoor and outdoor coils. On the indoor coil side, the refrigerant absorbs heat from inside the home. This causes the refrigerant to evaporate into a gas, which then passes through a compressor. The compressor pressurizes the refrigerant gas, raising its temperature.
The hot refrigerant gas then circulates through the outdoor coil, where it condenses back into a liquid state while transferring the heat outside. This releases, or “pumps”, the absorbed heat from inside to the outdoor environment.
The pressure on the refrigerant is then reduced as it passes through an expansion valve, lowering its temperature again so it can evaporate and absorb more heat when it reaches the indoor coil. This cycle is then repeated continuously while the heat pump is operating.
During summer, the indoor coil absorbs heat from inside the home and the outdoor coil releases heat outside, providing cooling. During winter, the cycle is reversed – the outdoor coil absorbs heat from the external environment while the indoor coil releases heat inside, providing heating. The direction of heat transfer is reversed by a valve that allows the refrigerant to flow in either direction through the system.
Overall, heat pumps use electricity to absorb heat at one place and release it in another, effectively “pumping” heat from a cool space to a warm space or vice versa. This allows them to efficiently heat and cool homes by moving existing heat rather than converting electricity into heat.
Limitations of Converting Heat into Energy
All processes that convert heat into usable energy face efficiency limits and energy losses. This is due to the thermodynamics of heat engines and the limits described by the Carnot Cycle.
The maximum theoretical efficiency of any heat engine operating between two temperatures is defined by the Carnot efficiency. This means that no heat engine converting heat into mechanical work can exceed this theoretical maximum efficiency. For example, between the temperatures of 500°C and 50°C, the Carnot efficiency limit is around 60%. In reality, practical heat engines achieve only 10-50% of this theoretical maximum due to friction, heat losses, and other limitations.
There are also always some energy losses during the conversion process from heat to electricity. These can include:
- Heat losses to the environment from hot components or working fluids
- Frictional losses in mechanical components like turbines
- Electrical resistance losses when generating electricity
- Wasted heat that is not fully converted into useful energy
Additionally, many heat sources have qualities that limit efficiency, such as variable or low temperatures. There are also costs and environmental impacts from heat energy sources like fossil fuels. These limitations prevent the full conversion of heat into usable energy from being achieved.
Ongoing research seeks to push the boundaries of conversion efficiency through improved materials, system designs, and integration techniques. But the inherent limits imposed by thermodynamics mean heat into energy conversion will always involve compromises and tradeoffs.
Conclusion
In conclusion, there are several important methods for converting heat into useful energy forms that rely on principles of thermodynamics. Heat engines like internal combustion engines and steam turbines use heat differences to generate mechanical work. Thermoelectric generators and thermionic converters can turn heat directly into electricity through the Seebeck and thermionic emission effects. Devices like heat pumps and heat exchangers utilize heat transfer to provide heating, cooling, and refrigeration. Power plants generate electricity on a large scale by burning fuel, fission, or using concentrated solar power. Understanding how to harness heat energy is crucial, as heat is abundantly available from sources like combustion, geothermal reservoirs, and waste industrial processes. Advances in thermoelectric materials, heat transfer fluids, and thermal insulation can improve the efficiency of these heat energy conversion systems. Overall, thermodynamics provides the fundamental basis for converting heat into other useful forms of power and energy.