Which Of The Following Shows That Radiant Energy Is Change To Chemical Energy?
Radiant energy is the energy of electromagnetic waves. Examples of radiant energy include visible light, ultraviolet light, infrared radiation, radio waves, and X-rays. Chemical energy is the potential energy stored in the bonds between atoms that make up molecules. The conversion of radiant energy into chemical energy occurs when radiant energy is absorbed by matter and causes chemical reactions or physical changes to take place. An example of radiant energy changing into chemical energy is photosynthesis in plants.
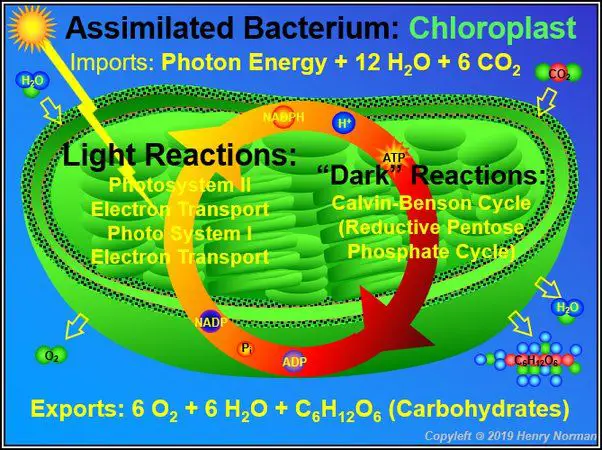
In photosynthesis, radiant energy from sunlight is absorbed by chlorophyll molecules in plant leaves. This radiant light energy causes a chemical reaction that converts carbon dioxide and water into glucose sugar molecules and oxygen. The radiant solar energy has now been converted into the chemical energy stored in the bonds of the glucose molecules. The glucose can then be used by plants as an energy source for growth and reproduction.
The transformation of radiant energy into stored chemical energy is vital to many natural processes and human technologies. This brief overview introduces the concept of radiant energy changing into chemical energy through interactions with matter.
Photosynthesis
Photosynthesis is the process plants use to convert radiant energy from the sun into chemical energy. This process takes place in the chloroplasts of plant cells, which contain the green pigment chlorophyll. When sunlight is absorbed by chlorophyll, the radiant energy is converted into chemical potential energy in the form of ATP and NADPH.
The overall chemical reaction of photosynthesis is:
6CO2 + 6H2O + Light Energy -> C6H12O6 + 6O2
Carbon dioxide and water, using energy from sunlight, are converted into glucose and oxygen. The glucose molecule contains stored chemical energy that the plant can use for growth and other life processes. So in summary, radiant light energy from the sun is absorbed by chlorophyll and through photosynthesis converted into chemical energy that is stored in glucose molecules.
Vision
One example of radiant energy being converted to chemical energy is through vision. The process begins when light enters the eye and is focused onto the retina, which contains light-sensitive receptor cells called rods and cones. When light hits these cells, it causes chemical changes that convert the light energy into electrical signals. These signals are then transmitted through neurons in the retina and optic nerve to the visual processing centers in the brain, where the electrical signals are further processed into visual information.
More specifically, rods contain a photopigment called rhodopsin that contains a derivative of vitamin A called retinal. When light hits rhodopsin, it changes shape in a process called photoisomerization. This alters the structure of the retinal molecule, converting it from the 11-cis-retinal isomer to the all-trans-retinal isomer. This molecular change activates the rhodopsin and triggers a cascade of biochemical reactions that produce an electrical signal, changing the cell’s membrane potential. The electrical signal is passed through interconnected neurons until it reaches the optic nerve.
Cones also contain photopigments that undergo similar chemical changes when struck by light. However, cones enable color vision by containing three different types of photopigments that are sensitive to different wavelengths of light corresponding to blue, green, and red. When light hits the photopigments, the induced chemical changes start a cascade leading to changes in membrane potential and electrical signals. In this way, both rods and cones convert the radiant energy in light to chemical and then electrical energy through photoisomerization and membrane potential shifts.
Vitamin D Synthesis
One example of radiant energy being converted into chemical energy is the synthesis of vitamin D in the human skin. When ultraviolet (UV) radiation from sunlight is absorbed by the skin, it triggers chemical reactions that convert cholesterol into vitamin D, an essential nutrient for bone health.
Specifically, the UVB wavelengths (290-320 nm) penetrate the epidermis and photolyze 7-dehydrocholesterol. This creates previtamin D3, which is thermally isomerized into vitamin D3. Vitamin D3 then enters the bloodstream and is hydroxylated in the liver and kidney to create the active form of vitamin D that promotes intestinal absorption and metabolism of calcium and phosphorus.
Without adequate sun exposure, vitamin D deficiency can develop. Since vitamin D is crucial for bone mineralization and calcium homeostasis, deficiency causes soft, thin, and brittle bones in children (rickets) and adults (osteomalacia). Vitamin D from the diet is usually insufficient to meet bodily requirements.
Therefore, the conversion of UVB radiation into previtamin D3 represents a key example of radiant energy being transformed into stored chemical energy essential for human health.
Solar Cells
Solar cells, also known as photovoltaic cells, are devices that convert light energy from the sun directly into electricity. This conversion process works through the photoelectric effect. When photons from sunlight strike the solar cell, they are absorbed by the semiconductor material in the cell, such as silicon. This photon absorption generates free electrons that can move through the material. The flow of these electrons produces an electric current that can then be captured and delivered for external use.
The electric current generated by solar cells is often stored in batteries, which store the energy chemically for later use. So in solar cells, radiant light energy from the sun is first converted into electric current through absorption by the cell’s semiconductor material. This electrical energy can then be stored as chemical energy in batteries. This demonstrates one way that radiant energy from the sun is transformed into stored chemical energy through photovoltaic processes.
Solar Thermal Systems
One way radiant energy from the sun is converted into chemical energy is through the use of solar thermal systems. These systems use solar collectors, often mounted on rooftops, to absorb thermal radiation from the sun and convert it into heat.
The most common type of solar thermal collector is the flat plate collector which is a thin, flat, rectangular box with a transparent cover on top. Inside the box is a dark absorber plate made of metal with attached copper tubes running through it. The sun’s thermal radiation passes through the transparent cover and hits the absorber plate, heating it up. A heat transfer fluid then circulates through the copper tubes, absorbing heat from the hot plate. This heated fluid is then piped to an insulated storage tank where the thermal energy is stored.
The stored hot water can then be used directly for applications like washing or space heating. Sometimes the thermal energy is transferred to a working fluid to produce steam to spin a turbine and generate electricity. In this way, the radiant energy from the sun is converted into heat and stored for later use as chemical energy.
Food Preparation
One of the most common ways that radiant energy is converted into chemical energy is through cooking food. When you use an oven, stove, grill or any cooking appliance that generates heat, it gives off radiant thermal energy that warms up and cooks the food.
This transfer of radiant heat energy causes physical and chemical changes to occur in the food through various processes like Maillard reactions, caramelization, starch gelatinization and more. The proteins, fats and carbohydrates undergo chemical changes that alter their structure and create new flavor compounds. This is why cooked food generally tastes much different than uncooked food.
The amount of radiant heat energy that gets absorbed by the food and the internal temperature achieved determines how thoroughly it gets cooked and the extent of chemical transformations. Higher heat induces more complex chemical reactions that result in greater changes to the color, texture, aroma and taste of food. Proper cooking makes food safer, more digestible and often more palatable.
So radiant heat energy from various cooking appliances clearly converts into chemical energy within food through the physical and chemical changes that occur during preparation. This exemplifies how radiant energy can turn into stored chemical energy.
Tanning
When skin is exposed to ultraviolet (UV) radiation from sunlight, chemical reactions occur in the skin that convert the radiant energy into increased production of melanin. Melanin is a pigment that gives skin and hair their color. It acts as a natural sunscreen by absorbing UV radiation and blocking it from reaching deeper layers of skin.
There are two types of melanin: eumelanin which gives skin a brown color, and pheomelanin which leads to freckles and red hair. UV exposure stimulates specialized cells called melanocytes to increase their production of melanin through a series of chemical reactions.
As melanin levels rise in the skin, visible darkening occurs which we recognize as a tan. This “suntan” helps protect deeper layers of skin from future UV damage. However, a suntan indicates that DNA damage has already occurred. While melanin has benefits, excessive UV exposure should always be avoided to lower skin cancer risk.
Photographic Film
Photographic film provides an excellent example of radiant energy being transformed into chemical energy. Traditional photographic film contains silver halide crystals that are sensitive to light. When the film is exposed to light during the photographic process, the radiant energy interacts with the silver halide crystals and causes a chemical change.
Specifically, the photons in the radiant light initiate a chemical reaction within the silver halide crystals. The photons are absorbed by the crystals, causing the silver ions to be reduced to metallic silver atoms. This forms a latent image on the film that looks like a hidden or invisible picture. The film is then processed with chemical solutions, which amplify this latent image and make it visible. The areas that were exposed to more light undergo a greater chemical change and appear darker on the processed film.
Therefore, the exposure of the photographic film to radiant light energy causes a chemical change in the silver halide crystals. This converts the radiant energy into a chemical form that captures the photographic image. Without this chemical change caused by the light exposure, the film would not be able to record a photograph.
Conclusion
Radiant energy from the sun converting to chemical energy is an essential process that sustains life on Earth. Through examples like photosynthesis, vitamin D synthesis, and food preparation, we’ve seen how plants, animals, and humans all rely on this energy conversion for growth, health, and survival. The sun’s radiant energy is captured and stored as chemical energy that living organisms depend on as fuel for biological processes. Even human technologies like solar cells and solar cooking take advantage of this natural process to produce electricity and cook food. Overall, the transformation of light to chemical energy powers life and civilization, and helps maintain the Earth’s delicate energy balance.
As we’ve reviewed in this article, radiant energy converting to chemical energy happens through many important chemical reactions and processes in nature. Appreciating and understanding this fundamental mechanism helps us value the role the sun plays in sustaining all life on our planet. Through continued research and innovation, we may find new ways to utilize radiant energy for human needs while working in harmony with natural systems.