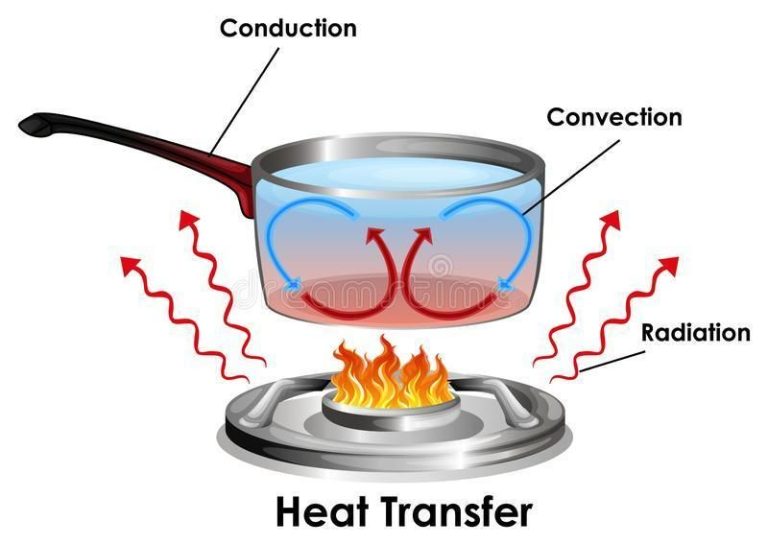
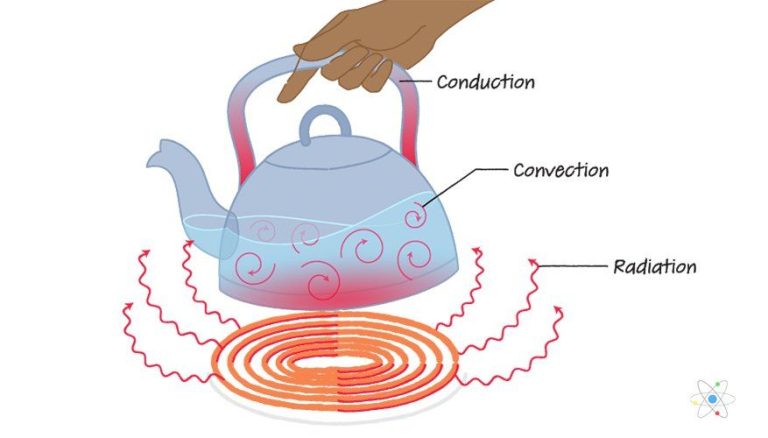
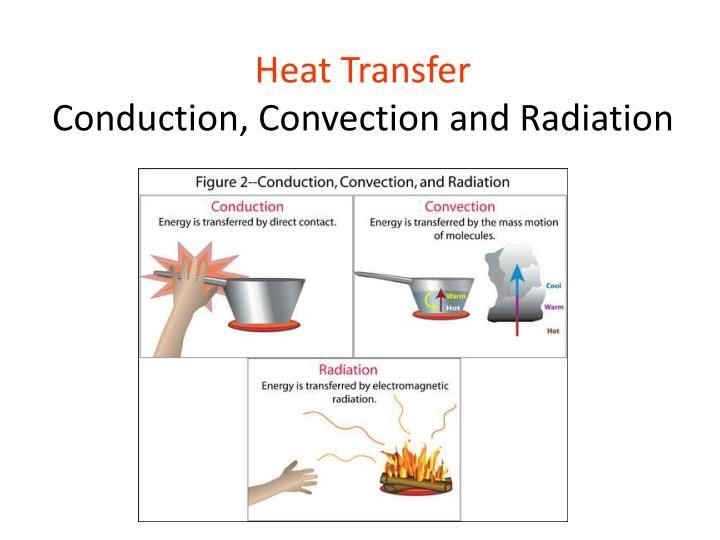
Where Does Most Heat Energy Come From?
Heat energy is a form of energy associated with the motion or vibration of atoms and molecules. It is generated by the movement and collision of these particles. Heat energy flows from a hotter object to a colder object – from high temperature to low temperature.
There are several main natural sources that generate most of the heat energy on Earth. These include the Sun, friction, electrical currents, chemical reactions, radioactive decay, and living organisms. Additional man-made sources come from the burning of fossil fuels and from mechanical work.
The Sun
The sun is the original source of most of the heat energy on Earth. Through a process called nuclear fusion, hydrogen atoms in the sun’s core are constantly fused into helium, releasing enormous amounts of energy in the form of solar radiation. This solar radiation travels over 150 million kilometers through space before reaching Earth’s atmosphere and surface. The sunlight that reaches and warms Earth’s surface provides the energy that drives weather, climate, ocean currents, the water cycle, photosynthesis in plants, and more. Without the constant outpouring of solar energy from the sun, there would be no life on Earth.
The rate at which the sun produces energy is extremely large and constant. Every second, the sun converts over 4 million tons of matter into energy, releasing 9.2 × 10^16 joules of energy primarily in the form of solar radiation. About 30% of this incoming solar radiation is reflected back into space by clouds, ice caps, and other reflective surfaces. The remaining 70% is absorbed by the oceans, lands, and atmosphere, heating Earth. This solar energy powers the planet’s climate and weather systems on an ongoing basis.
Friction
Friction generates heat energy whenever two surfaces move against each other. The friction force between the surfaces converts the mechanical energy of motion into thermal energy. Examples of frictional heating in everyday life include:
– Rubbing hands together to warm them up
– Tires heating up as they roll against the road
– Brakes heating up as they slow down a moving car
– Drilling into wood causing the drill bit to increase in temperature
– Feet getting hot in shoes from friction against the ground while walking
– Clothes in a dryer tumbling and rubbing to generate heat for drying
In each case, the mechanical energy exerted is dissipated into the environment as heat. The amount of heating depends on factors like the force pushing the surfaces together, the texture and hardness of the surfaces, and the speed of relative motion.
Electrical Current
Electricity flowing through wires and electrical devices produces heat through resistance. As electrons move through a conductor, they collide with atoms in the material, generating thermal energy. The amount of heat produced depends on the current (flow of electrons) and the resistance of the material. More current flowing through a higher resistance material will produce more heat. This is known as Joule heating.
Common electrical appliances and electronics rely on this principal to produce heat for various purposes. Electric stoves, ovens, and toasters use heating elements made of metals like nichrome that have high resistance. As electricity passes through them, they get hot enough to cook or toast food. Light bulbs also produce heat and light as electricity flows through their thin filament wires. Other electronics like computers and TVs generate heat as a byproduct when electricity powers their internal components.
Chemical Reactions
Chemical reactions can be a significant source of heat energy. Some chemical reactions release energy in the form of heat. These reactions are called exothermic reactions.
One of the most common examples is combustion reactions like burning wood, natural gas, gasoline, etc. These fuels undergo oxidation reactions with oxygen that result in the release of a large amount of heat energy. This heat energy can be captured and used for heating homes, cooking food, generating electricity in power plants, propelling automobiles, etc.
Another major source of heat from chemical reactions is cellular respiration and metabolism in living organisms. The breakdown of food molecules like glucose releases energy that is used by cells. Much of this energy is converted into heat and raises the temperature of the organism.
On a larger scale, heat production from chemical reactions drives major geological processes like volcanoes. Exothermic reactions also occur in manufacturing, such as the production of cement, steel, and many other materials.
Radioactive Decay
Radioactive decay of elements like uranium, thorium, and potassium in Earth’s interior produce a significant amount of geothermal energy. These radioactive elements are found in the planet’s crust and mantle, and as they decay, they release energy in the form of heat.
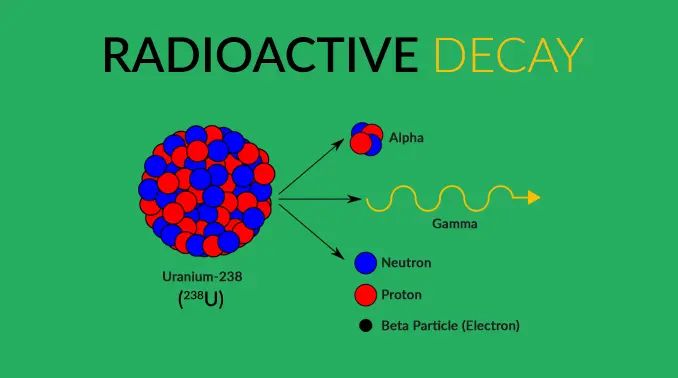
The decay of uranium-238 to lead-206 is a major source of heat, for example. As uranium atoms decay, they emit alpha particles and gamma rays and decay into other elements like radium and radon, producing heat in the process. Thorium-232 also decays in a similar radioactive chain to produce heat. Potassium-40 is another radioactive isotope that contributes to Earth’s internal heat.
This radiogenic heating from radioactive decay is estimated to contribute around 19% of the total heat flux from Earth’s interior. It helps drive convection currents in the mantle that are responsible for plate tectonics and volcanism on the surface. This geothermal energy from radioactivity plays a key role in powering geological processes and dynamics both deep within the planet and on the surface.
Fossil Fuels
Fossil fuels are a major source of heat energy globally. The burning of fossil fuels like coal, oil, and natural gas releases tremendous amounts of heat energy. When fossil fuels are combusted in power plants, factories, vehicles, and homes, the hydrocarbon molecules are oxidized, producing large quantities of heat.
Fossil fuels are formed from the buried organic matter of dead plants and animals from millions of years ago. Over time, heat and pressure converted the organic matter into fuels like oil, natural gas, and coal. Fossil fuels contain high concentrations of stored energy due to their hydrocarbon bonds.
The widespread burning of fossil fuels since the Industrial Revolution has released enormous amounts of carbon dioxide and other greenhouse gases, driving global climate change. The combustion of fossil fuels for energy is a major contributor to global heating and accounts for around 73% of total greenhouse gas emissions worldwide. Reducing dependence on fossil fuels is crucial to mitigate climate change and its impacts.
Living Organisms
Living organisms, including humans, animals, and plants, produce heat as a byproduct of metabolic processes that are essential for life. The chemical reactions involved in metabolism, such as the breakdown of food molecules, generate heat energy. In endothermic animals like mammals and birds, this metabolic heat is crucial for maintaining a constant internal body temperature.
The decomposition of organic matter by bacteria, fungi, and other organisms also releases heat. As microbes break down dead plant and animal material, the metabolic reactions involved give off heat. Compost piles are a good example of heat being produced from the microbial decomposition of organic waste. In large compost heaps, the heat generated by decomposition can raise temperatures substantially higher than the surrounding environment.
Mechanical Work
Mechanical work and motion can also produce heat energy through friction. Anytime two objects rub against each other, some of their kinetic energy is converted into thermal energy in the form of heat. For example, when you rub your hands together vigorously, the friction between your hands generates heat.
On a larger scale, most machines and vehicles generate heat from friction during operation. Internal combustion engines, like those in cars and power plants, convert chemical energy from fuel into mechanical energy to propel the engine’s moving parts. This motion inevitably creates heat from friction between engine components. The vast majority of global energy consumption comes from burning fossil fuels like gasoline and coal to power mechanical engines and machinery which produce usable energy, but also release tremendous amounts of waste heat.
Conclusion
In this article, we examined the main sources of heat energy on Earth and in the universe. The largest source by far is the Sun, which bombards the Earth with heat and light energy from nuclear fusion reactions. On Earth, the burning of fossil fuels like coal, oil, and natural gas also releases tremendous amounts of heat from chemical reactions. Friction converts mechanical energy into thermal energy and is another major way heat is generated on Earth, like when brakes get hot. Living organisms, radioactive decay, electrical currents, and mechanical work are other notable sources of heat energy.
The key applications for harnessing various heat sources are power generation, heating buildings and water, industrial processes, transportation, and cooking food. As fossil fuel reserves deplete over the next century, there will be increasing emphasis on utilizing renewable sources like solar and geothermal energy to meet humanity’s heating and electricity needs in a sustainable way.