What Is The Law Of Energy Exchange?
The law of energy exchange is a fundamental concept in physics and natural sciences that relates to the transfer of energy between systems. Simply stated, it says that in any interaction between systems, the total energy remains constant – energy can neither be created nor destroyed, but only changes form or transfers from one system to another. For example, when a ball drops, its potential energy transforms into kinetic energy. The origins of the law trace back to the 19th century and the work of pioneering scientists like Julius Robert Mayer, James Prescott Joule, and Hermann von Helmholtz who helped establish the principle of conservation of energy. Their experiments demonstrated that heat and mechanical energy are equivalent and interchangeable.
This law represents a core scientific truth about our universe and has profoundly shaped our understanding of physics, thermodynamics, chemistry, biology, ecology, and other fields. It shows that energy flows directionally whenever systems interact, and reminds us that energy is an essential driver behind all processes and phenomena. The law provides a universal accounting system for tracking energy transfers within and between complex systems. It has enabled great progress in harnessing energy for human ends, from steam engines to internal combustion to renewables. But there are limits to energy use, and the law cautions us against waste and overconsumption. By illuminating key energetic dynamics between human societies and the natural world, the law of energy exchange remains profoundly relevant today.
The Basics
The law of energy exchange states that energy cannot be created or destroyed, only transformed from one form to another. This is a fundamental principle of physics that has been found to apply across all natural systems. It means that the total amount of energy in a closed system always remains constant, even as energy changes form through different processes and interactions.
For example, when a ball falls to the ground, its potential energy gets transformed into kinetic energy as it speeds up due to gravity. The energy doesn’t disappear – it just changes from one type to another. This conservation of energy is always true, and it has profound implications for understanding our universe and the nature of energy flows within it.
The concept was first articulated in the 19th century as part of the development of thermodynamics. Scientists found that when carefully accounting for all energy inputs and outputs in a closed system, the total energy always remained the same. This principle has become a foundational concept in physics and other sciences.
Examples in Nature
The law of energy exchange is readily observed in many natural systems. Here are some key examples:
Photosynthesis: Plants absorb light energy from the sun and use it to convert carbon dioxide and water into sugars and oxygen. The chemical energy stored in the sugars, known as glucose, is then transferred up the food chain to provide energy to herbivores and omnivores when the plants are consumed.
The water cycle: The sun’s energy evaporates water from oceans, lakes, and rivers into water vapor which eventually condenses into clouds. This condensation releases energy that fuels cloud formation and rain. When the water falls as precipitation, it has potential energy that drives rivers and water flow systems. This energy is then dissipated through friction and enables transportation and nutrients to circulate in the environment.
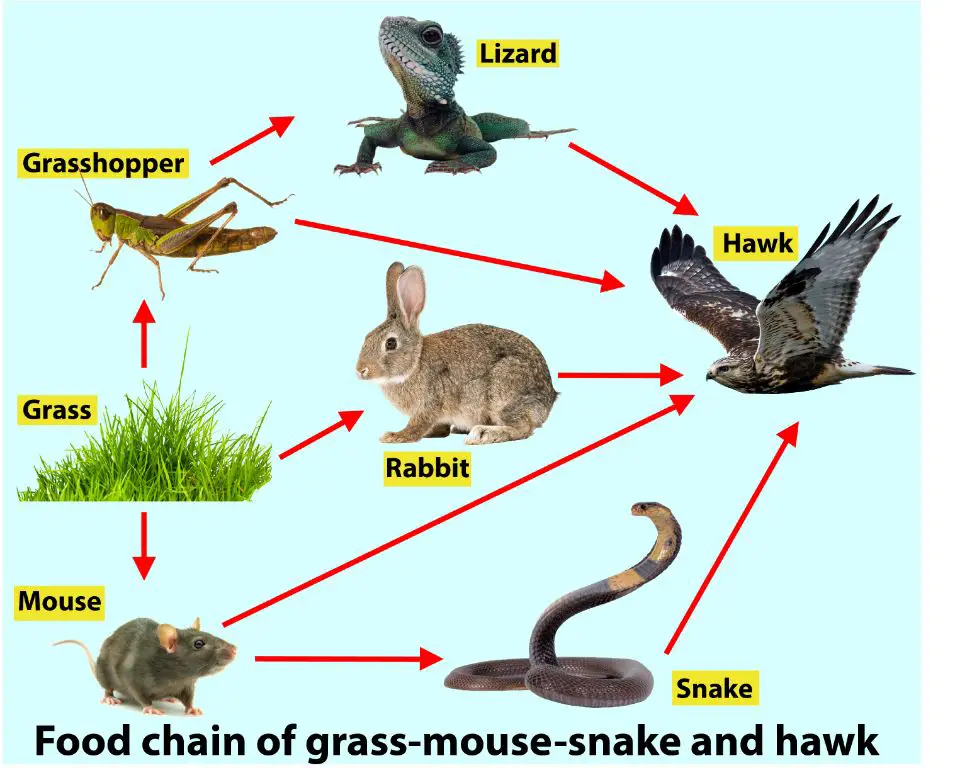
Food chains: All living organisms require energy to survive, grow and reproduce. This energy enters the ecosystem primarily through photosynthesis and is then transferred between organisms via consumption. For example, grass absorbs solar energy, mice eat the grass, snakes eat the mice, and hawks eat the snakes. At each trophic level, some energy is stored in biomass while the rest is dissipated as heat. This one-way flow forms a chain of energy transfer.
Examples in Human Systems
There are many ways humans see the law of energy exchange in action in everyday life. For example, electrical energy can be transformed into light, heat, or motion energy through different appliances and devices. Electrical energy powers light bulbs and produces light energy. Electric heaters and stoves transform electrical energy into heat energy for warming and cooking. Electric motors change electrical energy into the kinetic energy of motion to power fans, blenders, and cars.
Another key example is the chemical energy in food. Through digestion and metabolism, the chemical energy locked in food molecules gets transformed into kinetic energy that powers our bodies. The calories in food represent the potential chemical energy that can do work in the body.
On a larger scale, power plants convert the chemical energy stored in coal, oil, natural gas, or nuclear fuels into electricity. This generates the electrical energy that powers cities and the modern world.
In all these examples, one form of energy gets changed into another according to the law of energy exchange. The total energy remains constant, even as energy transforms into different states.
Historical Origins
The law of energy exchange has its roots in the 19th century discoveries in thermodynamics by scientists such as Julius Robert von Mayer, James Prescott Joule, and others. In 1842, Mayer first proposed that energy can be converted but not created or destroyed. This became known as the first law of thermodynamics. Joule later provided experimental proof of this principle through his famous paddle wheel experiments in 1843. He showed that mechanical energy is converted to heat energy without loss, thereby demonstrating the conservation of energy.
These foundational principles led to the establishment of the law of energy exchange as we know it today. The law states that energy cannot be created or destroyed, only changed from one form to another. For every energy exchange, the amount of energy input will equal the amount of energy output. While Mayer and Joule defined the essential scientific framework, other scientists and philosophers expanded on these concepts to develop a fuller understanding of energy dynamics. Hermann von Helmholtz clarified the difference between energy and force. William Thomson outlined principles of thermodynamic systems. Peter Tait and Balfour Stewart furthered investigations into the effects of energy exchanges. Through the collective work of these pioneers in physics and thermodynamics, the universal law of energy exchange emerged.
Applications
The law of energy exchange has many important applications in engineering and technology today. Engineers apply this law when designing systems to utilize renewable energy sources like solar, wind, and hydropower. The goal is to capture naturally occurring energy and convert it into useful forms like electricity. For example, solar panels capture photons from sunlight and convert them into an electric current.
The law also guides recycling efforts. Recycling gives discarded materials a new purpose, thereby extending the productive lifetime of the energy used to create them initially. Instead of ending up in a landfill, materials like paper, plastic, glass and metals can be reprocessed and manufactured into new products. This saves energy since it avoids extracting and transporting new raw materials.
More broadly, the law provides a framework for building sustainable systems that make effective use of available energy. Following the law helps identify opportunities to reuse waste energy and materials, rather than letting them dissipate. It also emphasizes maximizing efficiency to reduce unnecessary waste and energy expenditures.
Limitations
While the law of energy exchange describes an important rule that governs all dynamics in nature, it also has some limitations and challenges worth considering:
– In complex systems, it can be difficult to accurately measure and track all energy inputs and outputs. There may be unknowns or uncertainties that make the law hard to precisely apply.
– The law implies but does not guarantee equality or fairness in energy transactions. One party may benefit disproportionately from an exchange.
– The law focuses on quantitative energy exchanges but does not address qualitative factors that affect interactions.
– Strict adherence to the law can lead to transactional, tit-for-tat thinking that discourages generosity and goodwill.
– The law applies to closed systems but ecosystems and human societies are open systems with external energy sources that can disrupt balances.
– Some paradoxes in physics and thermodynamics seem to defy or complicate the basic law of energy exchange.
– The law does not account for man-made systems like currencies that attempt to regulate energetic transactions through pricing mechanisms.
– While useful as a concept, in practice no energy exchange achieves perfect equilibrium or equality.
– The law describes what is, not necessarily what should be. Moral and ethical factors may warrant deviation from strict energetic balance.
Related Laws
The law of energy exchange is closely related to other fundamental scientific laws that describe the natural world. Two of the most directly relevant are:
Conservation of Mass
The law of conservation of mass states that matter cannot be created or destroyed in a closed system. The total mass remains constant. This applies to energy exchanges where substances may change form but maintain the same overall mass.
Conservation of Momentum
The law of conservation of momentum says that in a closed system, the total momentum of objects before an interaction is equal to their total momentum after the interaction. Momentum must be transferred and conserved when energy is exchanged.
Like the law of energy exchange, these principles reflect the stable, balanced nature of mass and energy in closed systems. The universe itself appears to obey these conservation laws at its most fundamental levels.
Significance
The law of energy exchange is significant because it forms the basis for many scientific fields and technologies. Energy is a fundamental concept that underlies all of physics and chemistry. The law explains that energy can change forms but is never created or destroyed. This principle enabled the development of modern physics, chemistry, biology, and engineering. It allows us to understand how living organisms harness energy, how chemical reactions work, and how to build complex technologies.
The law provides a quantitative framework for analyzing energy transformations. We can use it to calculate the efficiency of machines, model chemical reactions, and predict biological processes. It allows us to harness various energy sources from fossil fuels to nuclear power. Technologies like batteries, engines, solar cells all rely on the law of energy exchange. Scientists apply it to design new energy systems and improve existing ones. Overall, this law has enabled major advances in science and engineering over the past two centuries. It remains essential for developing sustainable energy solutions and technologies needed for the future.
Conclusion
In summary, the law of energy exchange is a fundamental principle governing all systems in nature. It states that for energy to be transmitted or transformed, there must be a difference in energy potential between two systems or locations. This law underpins natural phenomena like heat transfer, electricity generation, and even the growth of living organisms.
While simple in concept, the implications of the law of energy exchange are profound. It helps explain the directional flow of energy throughout nature, the limits on efficiency for energy transfer processes, and the dependence of life on gradients and differences. This law guides designs from thermal power plants to biological enzymes.
Understanding the law provides insight into how our human systems for energy production and consumption align, or fail to align, with natural principles. It suggests that cooperation and symbiotic relationships, hallmarks of thriving natural systems, may offer models for more sustainable human technologies and economies. By illuminating deep patterns that connect living beings and their environments, the law of energy exchange reveals our interdependence with the natural world.