What Is Electric Source?
An electric source is a device or system that converts other forms of energy into electrical energy. Electricity is an extremely useful and versatile energy carrier that allows power to be transported easily and used in many applications. Almost all modern technology requires electricity to operate. Therefore, electric sources are vital components that enable virtually all electronics and electrical devices to function.
There are several different categories and types of electric sources, each with their own advantages and best uses. Some common examples include batteries, solar cells, fuel cells, thermoelectric generators, piezoelectric generators, electromagnetic induction, electrostatic generators, and radioisotope generators.
Electric sources allow us to generate electricity to power all kinds of important devices and systems. Some key applications that rely on electric sources include: powering consumer electronics like laptops, phones, and household appliances; storing energy from renewable sources like solar and wind power; powering electric vehicles and transportation; providing off-grid electricity access and backup power; enabling biomedical devices like pacemakers; and supplying consistent and reliable electricity for the electrical grid and infrastructure.
In summary, electric sources are essential devices that convert many forms of energy into usable electricity. The diversity of electric source technologies provides flexible options to generate electricity for an extensive range of applications and needs.
Batteries as Electric Sources
Batteries are electrochemical devices that convert stored chemical energy into electrical energy. They consist of one or more electrochemical cells, each containing two electrodes (a cathode and an anode) and an electrolyte that allows ions to move between them.
Batteries work because of reversible reduction-oxidation reactions that occur at the electrodes. At the anode, oxidation takes place, generating electrons that flow through the external circuit to the cathode. At the cathode, reduction takes place, consuming the electrons that flowed from the anode. The electrolyte allows ions to flow internally between electrodes to maintain charge balance.
Common battery types include:
- Lead-acid batteries – Used in vehicles and backup power systems. The anode is lead, the cathode is lead dioxide, and the electrolyte is sulfuric acid.
- Lithium-ion batteries – Used in consumer electronics and electric vehicles. The anode is graphite, the cathode is a lithium metal oxide, and the electrolyte is a lithium salt in an organic solvent.
- Alkaline batteries – Single-use batteries commonly used in flashlights and toys. The anode is zinc, the cathode is manganese dioxide, and the electrolyte is potassium hydroxide.
Advantages of batteries include portability, rechargeability (for secondary batteries), high specific energy, and low self-discharge. Disadvantages include limited lifetimes, slow recharge times, temperature sensitivity, and for primary batteries, non-rechargeability.
Solar Cells
Solar cells, also known as photovoltaic cells, convert sunlight directly into electricity. They are made of semiconductor materials like silicon that absorb photons from sunlight and release electrons. When these free electrons are captured, an electric current is generated that can be used as electricity.
There are three main types of solar cells:
- Monocrystalline solar cells – Made of single crystal silicon and have the highest efficiency rates of around 15-20%. They have a distinctive uniform look with an even coloring.
- Polycrystalline solar cells – Made of multiple crystals of silicon and have a slightly lower efficiency of around 12-15%. They have a speckled blue color and rectangular shapes.
- Thin-film solar cells – Made by depositing one or more thin layers of photovoltaic material onto a substrate. They have lower efficiencies of 7-13% but can be made very flexible and lightweight.
Solar cell efficiency is determined by how much of the sun’s energy can be converted into electricity. Current standard solar panels average 15-20% efficiency. Higher efficiency solar cells are being researched, with experimental cells reaching over 40% efficiency in lab conditions.
Solar cells have various applications in different sizes and scales. Small solar cells are used to power calculators and watches. Panels made of multiple solar cells can be installed on rooftops to power homes and businesses. Large solar farms can generate electricity on a utility scale and feed into the grid.
Fuel Cells as Electric Sources
Fuel cells are electrochemical devices that convert the chemical energy of a fuel directly into electricity. They work similarly to batteries, except they don’t run down and need recharging. Fuel cells will produce electricity continuously as long as fuel is supplied.
In a fuel cell, the fuel (typically hydrogen) is fed to the anode, while oxygen is fed to the cathode. At the anode site, the hydrogen molecules are split into protons and electrons. The protons pass through the electrolyte membrane to the cathode site, while the electrons travel in an external circuit, producing electricity. At the cathode, the electrons combine with oxygen and protons, producing water as the only byproduct.
There are several types of fuel cells, categorized mainly by their electrolyte material:
- Proton Exchange Membrane Fuel Cell (PEMFC) – Uses a solid polymer membrane electrolyte, operates at relatively low temperatures. Used for transport applications and small stationary uses.
- Phosphoric Acid Fuel Cell (PAFC) – Uses liquid phosphoric acid as the electrolyte, operates at higher temperatures. Used for stationary power generation.
- Molten Carbonate Fuel Cell (MCFC) – Uses a molten carbonate salt mixture as the electrolyte, operates at high temperatures. Used for stationary power generation.
- Solid Oxide Fuel Cell (SOFC) – Uses a solid ceramic electrolyte, operates at very high temperatures. For stationary and mobile power generation.
Fuel cells are very useful as electricity generators for stationary and portable applications. They have potential uses for powering buildings, data centers, vehicles, ships, airplanes and more. Compared to combustion generators, fuel cells produce electricity more efficiently and quietly, with minimal emissions.
Thermoelectric Generators
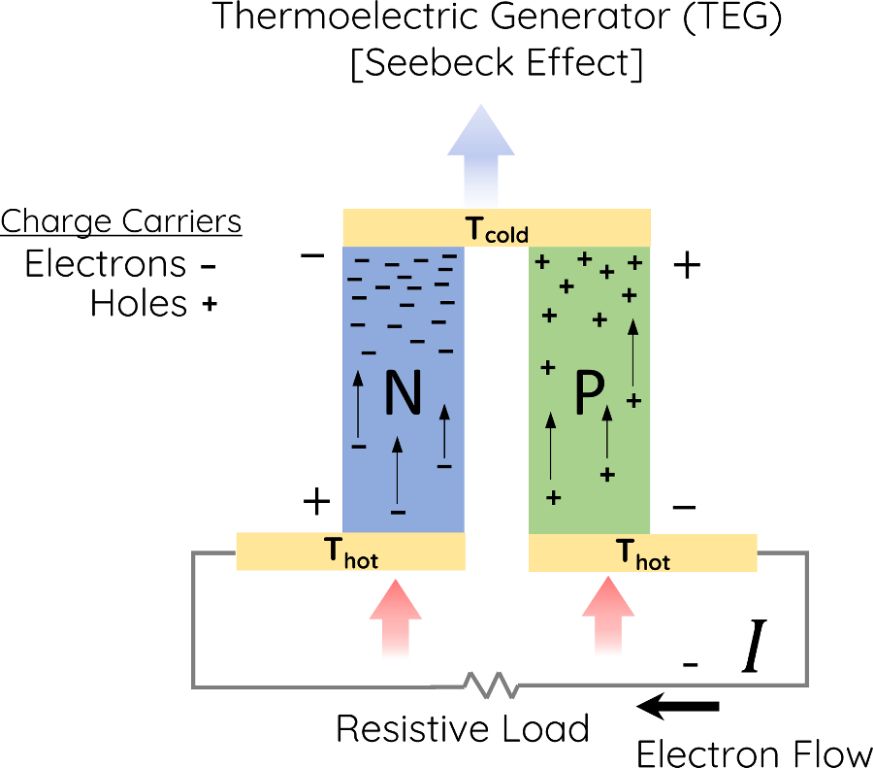
Thermoelectric generators (TEGs) are devices that convert temperature differences directly into electricity using the Seebeck effect. They function as heat engines that use electricity instead of mechanical power.
TEGs consist of thermocouples made from two different semiconductors, usually bismuth telluride or lead telluride, that are connected thermally in parallel but electrically in series. One side of the TEG is in contact with the heat source while the other side is cooled. When one side is hotter, charge carriers in the semiconductors diffuse from the hot side to the cold side, generating an electrical voltage.
Some key applications of TEGs include:
- Waste heat recovery – TEGs can capture waste heat from industrial processes, vehicle exhaust, etc. and convert it to usable electricity.
- Remote power generation – TEGs are used in remote sites like oil wells, buoys, and spacecraft to provide electricity from heat sources without any moving parts or maintenance.
- Portable generators – Small TEGs are used in camping stoves, gas stoves, outdoor grills to charge batteries or power electronics.
Research is ongoing to find more efficient thermoelectric materials to improve the conversion efficiency and applications of TEGs.
Piezoelectric Generators
Piezoelectricity refers to the ability of certain materials to generate an electric charge in response to applied mechanical stress. The piezoelectric effect occurs in crystalline materials that lack inversion symmetry. When the material is compressed or bent, the asymmetrical crystal structure results in a separation of electric charge, creating voltage. Conversely, applying voltage to a piezoelectric material can make it expand or contract.
Piezoelectric generators can harness this phenomenon to convert kinetic energy, vibration, or motion into useful electrical energy. Common piezoelectric materials used include quartz, some ceramics like barium titanate or lead zirconate titanate (PZT), and various polymers such as polyvinylidene fluoride (PVDF).
There are several ways piezoelectricity is utilized to generate electricity:
- Piezoelectric elements can be attached to structures and machines that vibrate or move to produce an AC output. This allows scavenging energy from existing motion.
- Stacking or connecting multiple piezoelectric layers can increase output voltage.
- Applying a compressive force cyclically on a piezoelectric material creates pulsating voltage.
- Cantilever beams made of piezoelectric substances can flutter in response to airflow, inducing flexing that generates electricity.
Applications of piezoelectric generators include powering small sensors and electronics, charging batteries, energy harvesting from walking, exploiting ocean wave motion, and even energy scavenging from breathing. Their simplicity and scalability make them attractive options for low power generation from mechanical energy sources.
Electromagnetic Induction
Electromagnetic induction is the process by which a conductor is exposed to a changing magnetic field and induces a voltage across itself. This phenomenon is utilized in electrical generators to convert mechanical energy into electrical energy.
The working principle behind electromagnetic induction is Faraday’s law of induction, which states that the induced electromotive force in a closed circuit is equal to the negative of the rate of change of magnetic flux through the circuit. When the magnetic flux through a conductor changes, it induces a voltage across the conductor. In an electrical generator, mechanical energy is used to rotate a magnet near a closed conductive loop, changing the magnetic flux through the loop and inducing a voltage.
Some common applications of electromagnetic induction in electrical generators include:
- Hydroelectric generators – Use falling water to spin turbines connected to magnets near wire coils
- Wind turbines – Use wind to spin turbines connected to magnets near wire coils
- Fossil fuel and nuclear power plants – Use steam to spin turbines connected to magnets near wire coils
- Gasoline/diesel generators – Use an internal combustion engine to spin magnets near wire coils
Electromagnetic induction allows generators to supply electricity from a variety of mechanical power sources. Generators at power plants supply electricity for power grids, while smaller portable generators are useful for providing electricity in remote locations or as emergency backup power.
Electrostatic Generators
Electrostatic generators produce electricity through electrostatic induction, which is the process of using electrostatic charge buildup to generate current flow. They operate on the principle that opposite charges attract while like charges repel. When charges build up on two separate conductors, it creates a voltage difference. This voltage can be tapped and directed through a circuit to do useful work.
A classic electrostatic generator is the Wimshurst machine, which consists of two counter-rotating disks with metal sectors. As the disks rotate, charge is induced on the sectors via electrostatic induction. Metal brushes collect this charge, building up large static voltages that can jump across a spark gap. The rapid switching of the charged sectors continually reverses the polarity of the output as the disks rotate, producing an oscillating high voltage.
More modern electrostatic generators utilize MEMS technology with microstructures that have moveable parts. As these parts move back and forth, they induce alternating charges which can be collected. Electrostatic generators are simple and have no moving parts besides the voltage-generating structures. They can effectively harness ambient energy sources like wind or vibration. However, they produce high voltage but low current electricity, so the output may need conditioning for practical use.
Radioisotope Generators
Radioisotope generators (RTGs) convert the heat released from the natural decay of radioactive materials into electricity. The radioactive materials most commonly used in RTGs are plutonium-238 and strontium-90. As these materials decay, they release alpha particles and beta particles, producing heat. This heat is then converted into electricity.
RTGs consist of two main components: the radioactive fuel source and the thermocouples. The thermocouples are made from metallic alloys and are placed next to the fuel source. As the fuel source decays and releases heat, the thermocouples absorb the thermal energy. They then convert this heat into electrical energy via the thermoelectric effect. The more significant the temperature difference between the hot side (next to the fuel) and cold side (exposed to the environment) of the thermocouples, the more electricity they can generate.
RTGs have long lifetimes and require no external power source, making them very reliable. They are ideal for providing power in remote locations like space probes and uncrewed lighthouses. Some of NASA’s deep space missions powered by RTGs include the Pioneer, Voyager, Galileo, Cassini, and New Horizons spacecraft. RTGs also provide auxiliary power on some Russian Federal Space Agency satellites.
Conclusion
Electricity can be generated from a variety of sources that convert different forms of energy into electrical energy. The key electric source types covered in this article include:
- Batteries – Electrochemical cells that convert chemical energy into electricity.
- Solar cells – Photovoltaic cells that convert sunlight into electricity.
- Fuel cells – Electrochemical cells that produce electricity from fuel sources.
- Thermoelectric generators – Devices that convert heat differentials into electricity.
- Piezoelectric generators – Materials that generate electricity from mechanical stress.
- Electromagnetic induction – Coils and magnets used to induce electric current.
- Electrostatic generators – Devices that use static electricity to generate power.
- Radioisotope generators – Systems that utilize radioactive decay to produce electricity.
Research and development continues to enhance existing technologies like solar cells, batteries, and thermoelectrics. Emerging electric source technologies like triboelectric and hybrid generators are also being explored. With global electricity demand projected to grow substantially, improving the efficiency, sustainability and accessibility of electric power generation remains an important focus area.