What Is A Short Note On Thermal Physics?
Thermal physics is a branch of physics that deals with heat and temperature and their relation to energy, work, radiation, and properties of matter. It explores concepts such as thermal expansion, thermodynamics, kinetic theory, and phase changes. Some of the major topics covered in thermal physics include:
Temperature – This is a measure of how hot or cold an object is. Thermal physics studies methods of measuring temperature and scales such as Celsius, Fahrenheit, and Kelvin.
Heat – Heat is energy transferred between objects due to a temperature difference. Thermal physics examines heat capacity, mechanisms of heat transfer, and the relationship between heat and thermodynamic work.
Thermal Expansion – Most materials expand upon heating and contract when cooled. Thermal physics seeks to quantify the thermal expansion of solids, liquids, and gases.
Thermodynamics – The laws of thermodynamics govern fundamental physical quantities like temperature, energy, and entropy. Thermal physics explores how to apply these laws to heating, cooling, and heat engines.
Phase Changes – Phase changes, like the melting of ice or boiling of water, absorb or release heat. Thermal physics studies these transitions between solid, liquid, and gas phases.
Some applications of thermal physics include refrigerators, air conditioners, internal combustion and jet engines, power plants, and even the weather and climate patterns of planets. The principles of thermal physics are crucial across engineering disciplines like mechanical, chemical, aerospace, and electrical engineering.
Temperature
Temperature is a measure of how hot or cold an object is. It is a measure of the average kinetic energy of the molecules in an object. Temperature is what we feel as hot or cold when we touch an object.
There are three main temperature scales used to measure temperature:
- Celsius – The Celsius scale (°C) is the most commonly used scale. On this scale, the freezing point of water is 0°C and the boiling point is 100°C.
- Fahrenheit – The Fahrenheit scale (°F) is mostly used in the United States. On this scale, the freezing point of water is 32°F and the boiling point is 212°F.
- Kelvin – The Kelvin scale (K) is the scientific standard used in thermodynamics. On this scale, 0 K (-273.15°C/ -459.7°F) is known as absolute zero, the theoretical lowest possible temperature where no more heat can be extracted from a system.
Temperature can be measured using various devices such as thermometers, thermocouples, and infrared thermometers. These devices work by correlating a measured property such as expansion or electrical resistance to temperature.
Heat
Heat is the transfer of thermal energy between systems due to a temperature difference. It refers specifically to the energy in transit, not the energy before or after the transfer takes place. Heat is a form of energy, but it does not represent the stored internal energy of an object.
Heat is different from temperature. Temperature measures the average kinetic energy of molecules within a system, expressed on a standard scale. Heat is the transfer of energy driven by a temperature difference, and it occurs spontaneously from a hotter to a colder body. An object does not “contain” heat, it contains internal energy. But heat is the energy transferred between objects due to their temperature difference.
The amount of heat required to change a substance’s temperature by one degree is called heat capacity. It is an intensive property, meaning it does not depend on the amount of material. The higher the heat capacity, the more energy is required to increase the temperature. Heat capacity depends on the material, its mass, and other factors. Understanding heat capacity allows us to engineer materials and systems that can effectively store and release heat for various applications.
Thermal Expansion
Thermal expansion refers to the tendency of matter to change its shape, area, volume, and length in response to a change in temperature. When most materials are heated, they expand, and when cooled, they contract.
On the molecular level, when matter is heated, the molecules and atoms gain kinetic energy and start vibrating and moving more. Since they take up more space while vibrating, the overall volume expands. This increase in size due to heating is known as thermal expansion. The degree of expansion varies based on the material and amount of temperature change.
Some common examples of thermal expansion include:
- Railroad tracks or bridges expand due to hot summer temperatures. Small gaps are left between the sections to allow for expansion.
- Heat causes metals like iron and copper to expand slightly. This is why plumbers leave space between pipes and take expansion into account.
- Glass or ceramic plates that suddenly change in temperature may crack or shatter from uneven expansion and stress.
Thermal expansion is used in various applications and devices. For example:
- Thermostats use thermal expansion of metal strips to control heating/cooling systems.
- Liquid thermometers rely on the thermal expansion of liquids in a sealed glass tube.
- Expansion joints and couplings are used in pipes, engines, boilers, ships, and buildings to allow for expansion and contraction.
Heat Transfer
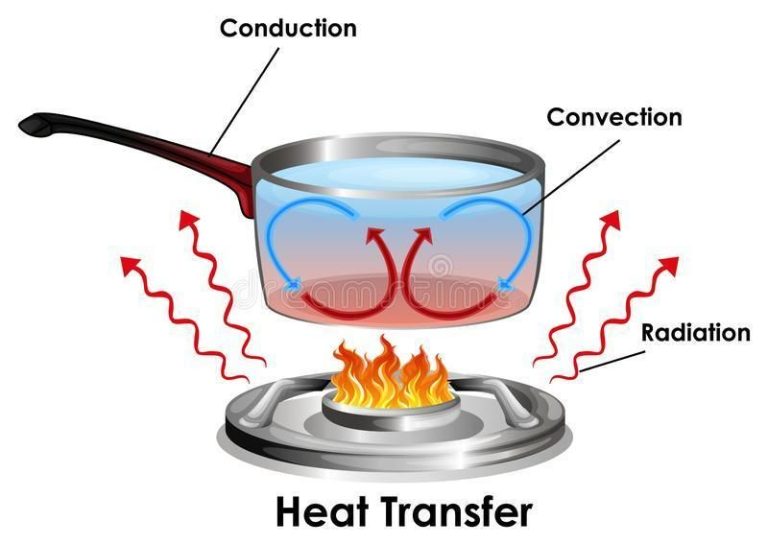
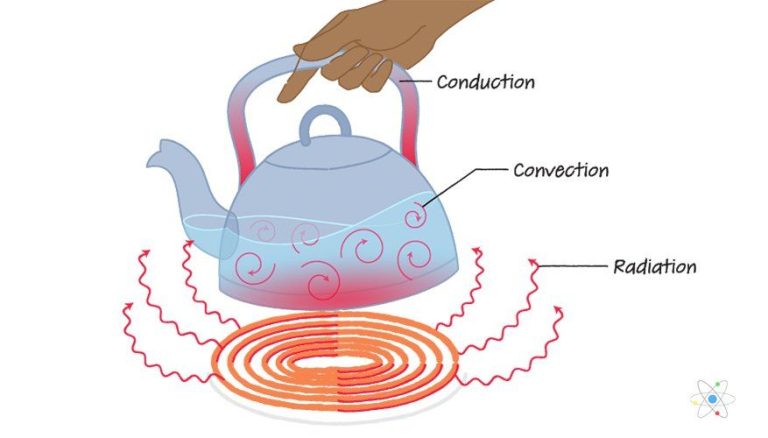
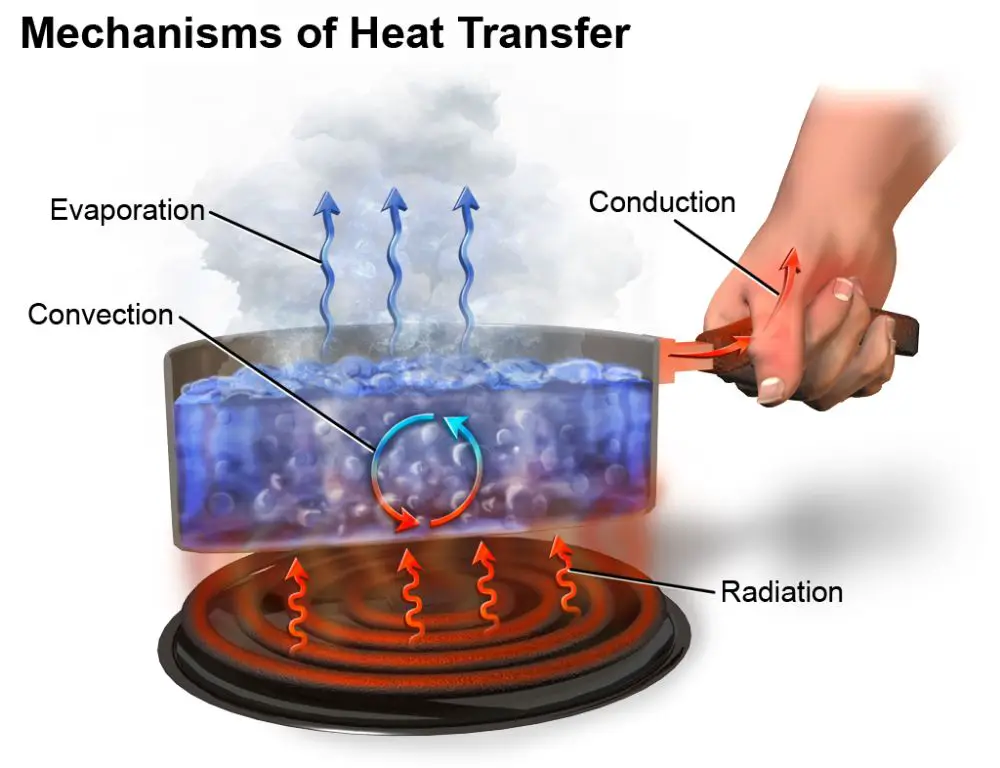
Heat transfer is the exchange of thermal energy between physical systems. There are three main mechanisms of heat transfer:
Conduction
Conduction is the transfer of heat between substances that are in direct contact with each other. It occurs when electrons from warmer areas interact with cooler areas, transferring kinetic energy. Materials like metals are good conductors. Insulators like wood or plastic do not readily conduct heat.
Convection
Convection is the heat transfer due to bulk movement of molecules within fluids such as liquids or gases. Hotter areas are less dense and rise, while cooler areas sink, causing circulation. Convection can be natural, driven by temperature differences, or forced, via fans or pumps.
Radiation
Radiation is the transfer of heat via electromagnetic waves directly between matter without requiring a medium to travel through. All matter with a temperature above absolute zero radiates thermal energy. Radiation does not require direct contact between source and destination.
Laws of Thermodynamics
Thermodynamics describes the relationships between heat, work, and energy. There are four laws of thermodynamics that provide an understanding of these concepts.
Zeroth Law of Thermodynamics
The zeroth law of thermodynamics states that if two systems are in thermal equilibrium with a third system, then they are in thermal equilibrium with each other. This law helps define temperature.
First Law of Thermodynamics
The first law of thermodynamics, also known as the law of conservation of energy, states that energy can neither be created nor destroyed in an isolated system. The change in the internal energy of a system is equal to the amount of heat supplied to the system minus the amount of work done by the system.
Second Law of Thermodynamics
The second law of thermodynamics states that heat flows spontaneously from a hotter to a colder body. It also states that it is impossible for a process to have as its sole result the transfer of heat from a colder to a hotter body. Another way of stating this is that the entropy of an isolated system can never decrease over time.
Third Law of Thermodynamics
The third law of thermodynamics states that the entropy of a perfect crystal at absolute zero is zero. This law provides an absolute reference point for the determination of entropy.
Engines and Refrigerators
Some of the most important applications of thermal physics relate to heat engines and refrigerators. A heat engine is a device that converts heat into mechanical work. It operates on a thermodynamic cycle, such as the Carnot cycle.
The Carnot cycle consists of four reversible processes:
- Isothermal expansion – The gas expands and does work on the surroundings while absorbing heat at a constant temperature.
- Adiabatic expansion – The gas continues to expand and do work, but without transfer of heat.
- Isothermal compression – The gas is compressed and work is done on it while expelling heat at a constant temperature.
- Adiabatic compression – The gas is compressed and work is done on it, but without transfer of heat.
This cycle provides the maximum possible thermodynamic efficiency for a heat engine operating between two specific temperatures. All heat engines are subject to the efficiency limits imposed by the Second Law of Thermodynamics.
A refrigerator or heat pump operates in reverse – using work to move heat from a low-temperature reservoir to a high-temperature one. The ideal refrigerator cycle is essentially a Carnot cycle operating in reverse. Real refrigerators have efficiencies below the Carnot limit, but still well above unity.
Thermal physics principles allow us to understand, design and improve various heat engines and refrigerators that are part of modern technology and everyday life.
Phase Changes
Phase changes occur when the physical state of a substance changes from solid, to liquid, to gas or vice versa. The three main phase changes studied in thermal physics are melting/freezing, evaporation/condensation, and sublimation.
Melting and Freezing
Melting refers to the phase change of a substance from solid to liquid state, which occurs when the kinetic energy of the molecules increases enough to overcome the intermolecular forces keeping it solid. The reverse process, liquid changing to solid, is called freezing. The temperature at which a substance melts or freezes is called its melting point or freezing point.
Evaporation and Condensation
Evaporation is when a liquid turns into a gas or vapor. It occurs when the kinetic energy of the liquid molecules is high enough to break away from the intermolecular attractive forces and enter the gaseous state. Condensation is the reverse process, when a gas transforms into a liquid state after the gas molecules lose enough kinetic energy.
Sublimation
Sublimation is when a substance changes directly from solid to gas or vice versa, without entering the liquid phase. The process of a solid turning directly into a gas is called sublimation, while deposition refers to the reverse process of a gas transforming directly into a solid.
Thermal Properties of Matter
There are three important thermal properties of matter that are useful for describing how materials respond to changes in temperature. They are:
Specific Heat Capacity
Specific heat capacity (Cp) is the amount of heat energy required to raise the temperature of 1 kg of a substance by 1°C. Substances like water have a high specific heat capacity, meaning they require a lot of heat to change temperature. Metals generally have low specific heat capacities.
Thermal Conductivity
Thermal conductivity (k) measures a material’s ability to conduct heat. Materials like metals are good thermal conductors, allowing heat to easily flow. Insulators like wood have low thermal conductivity, impeding heat flow.
Coefficient of Thermal Expansion
The coefficient of thermal expansion (α) describes how much a material expands upon heating. Most materials expand when heated and contract when cooled. A high coefficient means a material expands more for a given temperature change. Expansion needs to be considered in things like railroad tracks and heat sinks.
Knowing the thermal properties of materials is important for designing systems and components that involve heat transfer and thermal management. Engineers use these properties to predict how materials and structures will perform under different temperature conditions.
Applications of Thermal Physics
Thermal physics has many practical applications in everyday life as well as cutting-edge technologies. Here are some examples:
Everyday Examples
– Cooking – Applying heat to food to alter its chemical properties. Different cooking methods like baking, frying, etc. use thermal physics principles.
– Heating and air conditioning – Creating and regulating comfortable temperatures in homes and buildings.
– Engines – Internal combustion engines rely on exploding fuel to move pistons and generate power.
– Thermometers – Measuring temperature based on how heat energy affects substances like mercury or alcohol.
Advanced Technologies
– Power generation – Most electricity is generated by heating water to produce steam that spins turbines connected to generators.
– Refrigeration – Removing heat via compression and expansion of gases to lower temperatures for food storage, AC, cryogenics etc.
– Thermal imaging – Detecting infrared radiation to see variances in heat and temperature not visible to the eye.
– Semiconductors – Controlling electrical conductivity in chips and computing devices by carefully regulating heat dissipation.