What Are The 5 Properties Of Energy?
Energy is an important concept across all fields of science, from physics to biology. Energy makes life and civilization possible. At its most basic, energy is the ability to do work or cause change. Energy exists in different forms that can be converted from one to another, like kinetic and potential energy. The properties of energy describe how it behaves and interacts with matter.
Understanding the fundamental properties of energy provides insights into the physical world. From the growth of plants to electricity generation, the applications of energy principles are all around us. By studying the properties of energy, scientists gain knowledge to develop new technologies and solve global challenges. Appreciating energy as a conserved, convertible, stable-yet-dynamic quantity underlying all natural processes allows clearer thinking across scientific disciplines.
Conservation of Energy
The law of conservation of energy states that energy cannot be created or destroyed, only converted from one form to another. This means the total energy in an isolated system always remains constant. For example, when a ball drops, its potential energy is converted to kinetic energy as it falls. The total amount of energy before and after the drop remains the same. According to this fundamental law of physics, energy can change forms between kinetic, potential, thermal, chemical, nuclear and other types, but the total quantity of energy in a closed system does not change.
The law of conservation of energy is important because it allows us to calculate the amounts of different forms of energy before and after energy is converted from one form to another. We know that if 100 J of potential energy is converted to kinetic energy, there will be 100 J of kinetic energy. This law allows us to analyze energy transformations quantitatively. It is one of the most important laws governing energy and its behavior.
Potential Energy
Potential energy is the energy an object has due to its position, composition, or arrangement. In other words, it is energy that is stored and held in readiness. For example, energy is stored in the water held behind a dam or in a battery. This stored energy has the potential to do work when released.
Some common examples of potential energy include:
- Gravitational potential energy – Objects positioned high above the ground, such as a water reservoir or a rock on a cliff, contain gravitational potential energy due to the force of gravity acting on their mass.
- Elastic potential energy – Compressed or stretched springs and elastic bands contain elastic potential energy due to the restoring force that tries to bring them back to their original shape.
- Chemical potential energy – Energy stored in the bonds between atoms and molecules in fuels, batteries, food, and living organisms contain chemical potential energy that can be released in chemical reactions.
Potential energy is converted into kinetic energy when the object falls or when the chemical bonds are broken. Understanding potential energy is crucial for harnessing renewable energy sources like water, wind, and the sun.
Kinetic Energy
Kinetic energy is the energy of motion. Any object that has motion – whether it is vertical or horizontal motion – has kinetic energy. The amount of kinetic energy depends on the mass and velocity of the object. The greater the mass and velocity, the greater the amount of kinetic energy.
Some examples of kinetic energy include:
- A car driving down the road
- A child swinging on a swing
- Heat, which is the motion of molecules
- Light, which is energy that travels in the form of electromagnetic waves
- Sound, which is the motion of sound waves
- Wind, which is the motion of air
In physics, the kinetic energy of an object is calculated using the equation:
KE = 1/2 mv2
Where m is the mass of the object and v is its velocity. This shows that an increase in either mass or velocity will result in an increase in the object’s kinetic energy.
Chemical Energy
Chemical energy is the energy stored in the bonds between atoms and molecules. It is the energy that holds these particles together. This energy can be released or absorbed in chemical reactions when chemical bonds are broken or formed.
Some common examples of stored chemical energy include:
- Food – The cells in our bodies break down the chemical bonds in food to release energy that allows our bodies to function.
- Fuel – Gasoline, diesel, and other fuels contain high amounts of stored chemical energy in their molecular bonds. This energy is released through combustion, such as in car engines.
- Batteries – Batteries store chemical energy in the bonds of their chemicals. This energy is converted to electrical energy when the chemical reaction inside the battery takes place.
So in summary, chemical energy is energy contained in the structure of molecules and released in chemical processes. It’s an essential form of energy that powers many biological, industrial, and mechanical systems.
Electrical Energy
Electrical energy is energy that comes from the movement of electrical charges, usually electrons. Some common examples of electrical energy include the electricity that comes from wall outlets to power various appliances and devices, lightning, and powering computers and other electronics.
Electrical energy is generated when electrical charges flow through a conductor. For example in power plants, generators spin magnets around conductive coils to induce an electric current and voltage. Electricity is then distributed through power lines and transmission cables to deliver it to homes, businesses, and factories. Electricity flowing through wires or circuits can then power many different devices once that energy is converted by the device into other usable forms.
Electrical energy powers much of the modern world. It allows appliances like refrigerators, lights, and televisions to operate. It runs machinery and electronics of all kinds. While electricity flows silently and imperceptibly, it involves extremely high levels of energy that do measurable work each second. The convenience and versatility of electrical energy to produce heat, light, motion, or computing makes it one of the most useful forms of energy today.
Radiant Energy
Radiant energy is a form of energy that travels through space or matter in waves. The most familiar form of radiant energy is visible light, but radiant energy also includes ultraviolet light, infrared radiation, radio waves, microwaves, X-rays and gamma rays.
All forms of radiant energy are types of electromagnetic radiation that move energy from one place to another through electromagnetic waves that can travel through space without requiring a medium like air or water. Electromagnetic waves are composed of oscillating electric and magnetic fields moving perpendicular to each other and to the direction of the wave.
The fundamental unit of radiant energy is the photon, which carries a fixed amount of energy based on its wavelength. Shorter wavelength electromagnetic radiation like gamma rays and X-rays carry higher energy photons than longer wavelength radiation like radio waves and microwaves.
Radiant energy is produced through atomic and molecular interactions. Common sources include the sun, lightbulbs, radio transmitters, nuclear reactions, and X-ray machines. Radiant energy can generate electrical currents when absorbed, heat surfaces through infrared radiation, cause chemical reactions, and damage organic tissue through ionizing radiation like ultraviolet rays.
Radiant energy travels at the speed of light and can be reflected, refracted, or absorbed when interacting with matter. It follows the inverse square law – intensity decreases rapidly with increasing distance from the source. Radiant energy is considered a renewable energy source and plays a fundamental role in the physics of the universe.
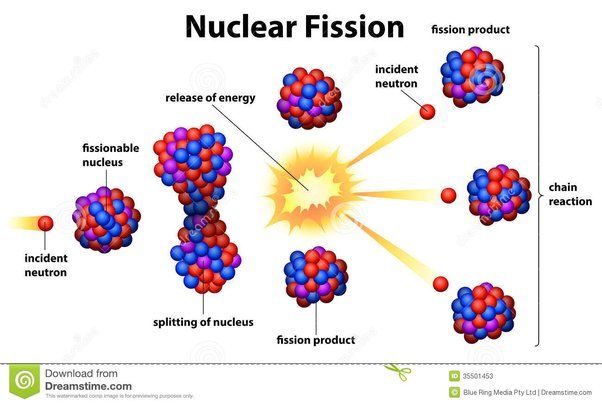
Nuclear Energy
Nuclear energy comes from the splitting (fission) or fusion of atomic nuclei. Nuclear fission occurs when an atomic nucleus like uranium or plutonium splits into smaller nuclei, releasing energy in the process. This energy can be harnessed to generate electricity in nuclear power plants. Fission reactions also produce radiation, which must be contained. Nuclear fusion joins together light atomic nuclei to form heavier nuclei, releasing energy. The sun produces energy through fusion reactions.
In nuclear power plants, nuclear fission reactions are used to heat water and produce steam that spins turbines to generate electricity. Over 400 nuclear power plants supply about 11% of the world’s electricity. Nuclear power is considered a relatively clean energy source since it does not directly emit air pollutants or carbon dioxide. However, there are concerns about accidents, radioactive waste disposal, and security risks with nuclear plants.
Radioactivity refers to the particles and radiation released by decaying atomic nuclei. Exposure to high levels of radiation can damage human cells and cause health effects like cancer. Nuclear facilities have multiple safety systems and radiation shielding to protect workers and the environment from exposure. But accidents can release significant radiation. Nuclear waste remains radioactive for thousands of years, so safe long-term disposal is required.
Thermal Energy
Thermal energy refers to the internal energy of a system that arises from the random motion of molecules and atoms. It is directly associated with the temperature of matter. The higher the temperature, the greater the thermal energy since the atoms and molecules are moving faster and colliding more frequently.
Thermal energy can be transferred between objects through processes like conduction, convection, and radiation. For example, when you heat up a pot of water on the stove, thermal energy is transferred from the hot stove burner to the pot through conduction. The water molecules absorb this thermal energy, increasing their kinetic energy and thus raising the temperature. Thermal energy always moves from higher temperature matter to lower temperature matter if they are in contact.
We can calculate thermal energy using the heat capacity and temperature change of a substance. Heat capacity is the amount of energy needed to raise a substance’s temperature by 1 degree. By measuring the temperature change of a substance as thermal energy is added or removed, we can quantify the thermal energy using its specific heat capacity. This allows us to track the gain or loss of thermal energy during heating and cooling processes.
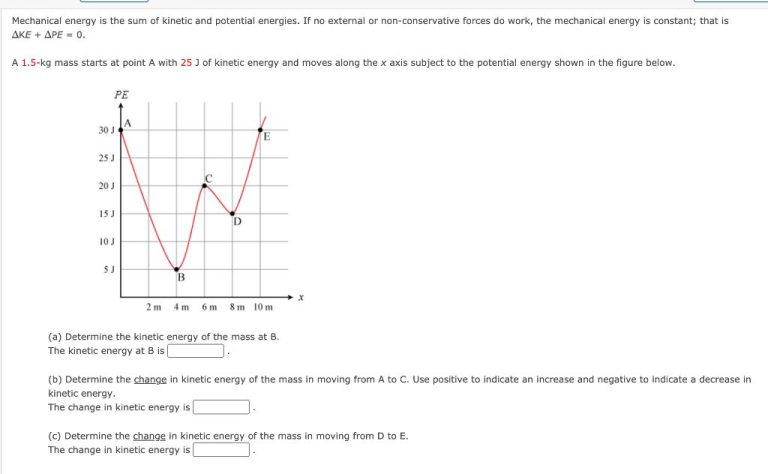
Mechanical Energy
Mechanical energy is the sum of an object’s kinetic energy and potential energy. Kinetic energy is the energy associated with motion, while potential energy is stored energy that depends on an object’s position or shape.
Some examples of mechanical energy include:
- A stretched or compressed spring contains mechanical energy in the form of potential energy.
- A bowling ball rolling down a lane contains mechanical energy in the form of kinetic energy.
- engines contain mechanical energy in the forms of kinetic energy, potential energy, and thermal energy from the combustion of fuel.
Mechanical energy can be converted between potential and kinetic forms. For example, when a ball is thrown upwards, its initial kinetic energy is converted into gravitational potential energy at the balls highest point. As it falls back down, this potential energy is converted back into kinetic energy.