Is There A Physical Form Of Energy?
What is Energy?
Energy is the capacity to do work and cause change. It is needed to make things move, grow, or happen in general. Energy enables all activity and life. On a fundamental level, energy is the ability to cause a transformation or transition of some kind. It exists in various forms that can be harnessed to produce motion, light, heat, growth, or other phenomena.
Some key characteristics of energy include:
- Energy is the ability to do work or cause action.
- Energy comes in different forms that can be converted from one to another.
- The law of conservation of energy states that energy cannot be created or destroyed, only transformed.
- Energy flows and is transferred between objects through work and heat.
- The total energy of an isolated system remains constant.
In physics, energy is measured in various units like joules, kilowatt-hours, calories, etc. But fundamentally, energy is the driver of all activity in the universe. It’s the invisible “fuel” powering everything from stars to cells to technology. Understanding energy is key to understanding our world.
Forms of Energy
There are many different forms that energy can take. The major forms of energy include:
- Thermal Energy – The energy that comes from the motions of atoms and molecules in a substance. The greater the temperature of a substance, the more thermal energy it possesses.
- Radiant Energy – The energy carried by electromagnetic waves such as light. It can transfer from one location to another without heating the intervening space.
- Kinetic Energy – The energy possessed by an object due to its motion. The faster an object moves, the more kinetic energy it has.
- Potential Energy – The stored energy an object has due to its position or chemical composition. There are types like gravitational potential energy and elastic potential energy.
- Electrical Energy – The energy from the flow of electrons. It is used to power devices and can easily be converted to other forms.
- Chemical Energy – The energy stored in the bonds between atoms and molecules. It is released in chemical reactions.
- Nuclear Energy – The energy that holds nucleons together in the nucleus of an atom. It can be released in nuclear reactions.
Energy is constantly being converted from one form into another. For example, chemical energy in gasoline is converted to kinetic energy in a moving car. Understanding the different forms energy takes is crucial to utilizing it effectively.
Energy Conversion
Energy can change from one form to another, but the total amount of energy in a closed system remains constant. This is known as the law of conservation of energy. For example, chemical energy stored in a battery can be converted into electrical energy to power a light bulb. The light bulb then converts some of that electrical energy into heat and light. The total amount of energy at the start (chemical energy in the battery) is equal to the total amount of energy at the end (heat, light, and any remaining electricity).
Some common energy conversions include:
- Chemical to electrical (battery)
- Chemical to thermal (burning fuel)
- Electrical to thermal (electric stove)
- Electrical to mechanical (electric motor)
- Mechanical to electrical (generator)
- Nuclear to thermal (nuclear reactor)
- Solar to electrical (solar panel)
Energy conversions allow us to transform energy from one form to another to meet our needs. For example, a car engine burns gasoline to release chemical energy, which is converted into mechanical energy that powers the wheels. Even though energy changes form, the total amount remains the same due to conservation of energy.
Is Energy Physical?
Whether energy has physical properties or not has been debated by scientists and philosophers for centuries. On one hand, energy does seem to have some physical characteristics. For example, energy can be transferred from one object to another through radiation, conduction, or mechanical work. We can measure and quantify energy in units like joules or kilowatt-hours. Energy also follows the laws of thermodynamics and must be conserved in any process or transformation.
However, energy itself is an abstract concept that is not tangible like matter. Energy manifests itself in various forms like kinetic, potential, thermal, or electromagnetic energy. But the energy itself is not a physical object that we can see or touch. We cannot destroy or create energy, only convert it from one form to another. This immaterial nature makes some argue that energy is a mathematical abstraction and not something physical.
Ultimately, there are good arguments on both sides of the debate. Energy does have some physical properties and can be measured empirically. But it also has an intangible, abstract essence that is not like traditional matter. The nature of energy continues to be explored by modern physics through quantum field theory and other frameworks. While a definitive answer may not be reached, studying the physical characteristics of different energy forms remains an important area of scientific research.
Energy and Matter
Energy is not the same thing as matter, but energy does need matter in order to be transferred or transformed. Matter refers to anything that has mass and takes up space. Energy, on the other hand, is the capacity to do work or produce heat. Energy itself does not have mass or take up space like matter does.
While energy is an abstract quantity, it needs some physical material or matter in order to be measurable or observable. For example, energy in the form of heat needs materials like air or water to transfer the warmth from one place to another. Chemical energy stored in a battery requires materials like lithium and copper to hold and release the energy. Even light, which can travel through empty space as electromagnetic waves, requires charged particles or matter to be generated in the first place.
So in summary, energy relies on matter to be harnessed, used, and transferred. But energy itself does not physically take up space like traditional matter. Energy is an abstract property that exists as a capacity or potential, one that can do work within matter when converted from one form to another.
Energy Storage
Energy can be stored in various physical forms and systems. Some key ways that energy is stored physically include:
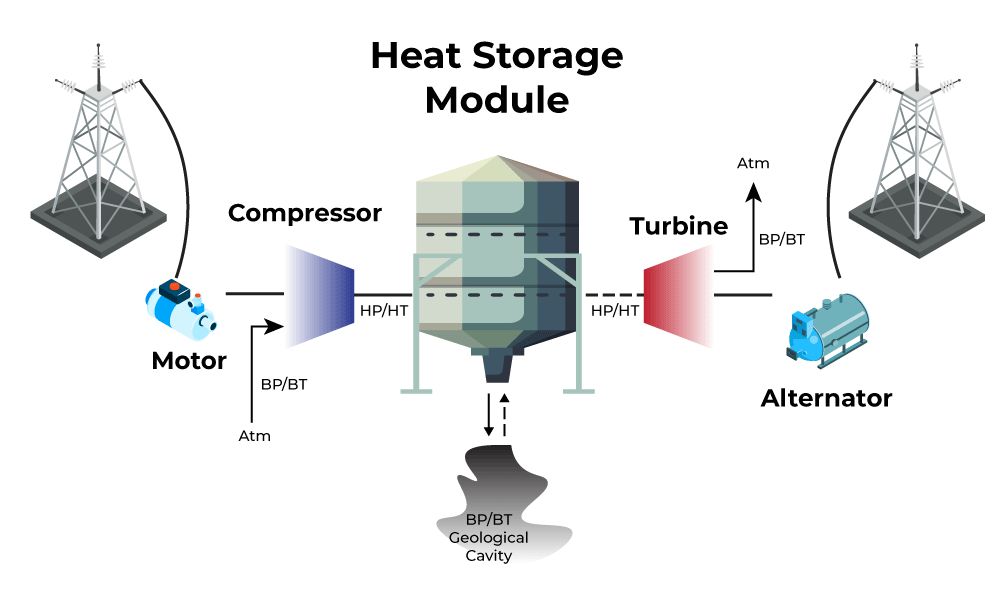
Chemical energy – Energy stored in the bonds between atoms and molecules. Batteries and fuels like gasoline, natural gas, and coal contain stored chemical energy that can be released through chemical reactions.
Electrical energy – Energy stored in electric fields between charges or in magnetic fields created by electric current. Capacitors and inductors can store electrical energy.
Mechanical energy – The energy stored in objects by tension, compression or motion. Springs and flywheels are common mechanical energy storage devices.
Nuclear energy – Energy stored in the nuclei of atoms through nuclear binding forces. Nuclear fuel contains stored nuclear energy that can be released in nuclear reactions.
Thermal energy – Energy stored in the molecular motion of a substance. Water tanks, molten salt, and materials with high specific heat capacity allow storing thermal energy.
Gravitational potential energy – Energy stored in objects elevated against gravity. Pumped hydro facilities store energy in elevated water.
In summary, energy storage always involves a physical process or system that can accumulate energy and release it on demand. Batteries, fuels, capacitors, flywheels, elevated water, and heated materials represent some of the many physical forms energy storage can take.
Energy Transfer
Energy can be transferred between objects through various processes and mechanisms. Some key ways that energy is transferred include:
-
Conduction – The transfer of energy as heat between objects in direct physical contact. Heat energy transfers from higher temperature to lower temperature objects when they touch, like a pot on a stove heating up.
-
Convection – The transfer of heat by the movement of heated fluid or gas. Hot air or liquid rising carries thermal energy from the heat source and heats up the surrounding area, like hot air from a furnace circulating in a room.
-
Radiation – The transfer of energy by electromagnetic waves or photons. Heat and light from sources like the Sun radiate outwards in all directions. Objects absorb the radiation to gain thermal energy.
-
Mechanical Work – The transfer of energy when a force causes movement or displacement. Physical forces acting on an object over a distance do work to transfer kinetic and potential energy.
During energy transfers, the total amount of energy remains constant. Energy changes from one form into another, but is never created or destroyed. The processes and mechanisms allow energy to spread out and balance in the universe.
Measuring Energy
The ability to accurately quantify energy is essential to understanding it and putting it to use. Energy is measured using a variety of units and methods based on the form or domain being examined. Some key ways energy is measured include:
Units: The standard SI unit for energy is the joule (J). Other common units include calories, British thermal units (BTUs), kilowatt-hours (kWh), and tons of oil equivalent (toe). These units allow numeric values to be assigned to energy amounts in different contexts.
Calorimetry: This involves directly measuring thermal energy by observing temperature changes in a calorimeter. It is used to quantify energy content in fuels and foods.
Electricity meters: Utilities use these devices to monitor and bill for electric energy usage in homes and businesses based on kilowatt-hours consumed.
Power meters: These measure the instantaneous energy conversion rate of electrical devices in watts or kilowatts.
Fuel gauges: These measure the amount of chemical energy remaining in a fuel tank be it gasoline, diesel, or other liquid fuels.
Spectroscopy: This set of techniques uses the interaction of electromagnetic radiation with matter to measure energy transitions and quanta at atomic and molecular levels.
The diversity of energy measurement approaches reflects the diverse manifestations of energy in our world. But the ability to assign definitive numerical values through proper measurement techniques provides the foundation for deeper Energy analysis and insights.
Perception of Energy
Energy can seem abstract and intangible from a human perspective. We cannot directly see, hear, taste, smell, or touch energy itself. Yet we can perceive its effects all around us – the warmth of the sun, the force of wind, the brightness of a lightbulb. This contrast between subjective perception and the physics view of energy as a quantifiable, measurable property of matter can create confusion.
Physicists define energy as the ability to do work or transfer heat. But in daily life, we associate energy with feelings of vigor, enthusiasm, and liveliness. We might say someone has “good energy” or “bad energy.” This ties energy to emotions and personality traits, which physics does not support. While our perceptions shape how we think about energy, from a physics standpoint, energy itself has no inherent moral or emotional qualities.
The human tendency to project sensory attributes onto energy can also lead to misconceptions. We describe types of energy as “clean” or “dirty,” “warm” or “cold,” “bright” or “dark.” But energy itself has no color, warmth, or texture. We are merely describing our subjective sensory experience of different energy manifestations. This anthropomorphizing can create confusion between metaphor and physical reality.
Ultimately, energy remains an abstract concept that we cannot directly experience through our senses. While our perceptions and metaphors for energy differ from the physics view, they help us connect and engage with something inherently intangible. Bridging the gap between subjective experience and scientific objectivity is key to demystifying energy.
Conclusions
In summary, while energy itself is an abstract concept representing the potential to do work or cause change, all forms of energy have some physical basis or manifestation. Kinetic and potential energy exist as properties of physical matter and systems. Thermal, chemical, nuclear and electromagnetic energy involve the motion or arrangement of particles and fields. Even seemingly empty space contains fluctuating quantum fields with real physical effects.
While energy cannot be directly observed or touched, every change in the physical world is accompanied by changes in energy. Energy is intrinsically linked to matter and always requires some physical carrier or system. Ultimately, the physicality of energy lies in the fact that it is measurable, quantifiable and has tangible effects in the material world. Without any physical manifestation or influence, energy would remain an abstract idea without context.
In summary, energy is indeed physical – not as a discrete tangible substance, but through its real manifestations, conversions and interactions with matter. All observable forms of energy have some basis in the physical world. Energy undeniably exists as a real physical property, influence and driver of change in physical systems.