Is Production Of Light A Chemical Or Physical Change?
Chemical changes and physical changes are different types of changes that can occur in matter. Chemical changes involve a substance being changed into a new substance through chemical reactions. The composition and properties of the original substance are altered. Physical changes simply change the shape, size, or state of a substance, but do not create a new substance. Understanding the differences between chemical and physical changes is fundamental to the study of chemistry and physics.
In a chemical change, chemical bonds are broken and formed, resulting in one or more new substances. This often involves an energy transfer, such as through heat, light, or electricity. Examples of chemical changes include burning wood, cooking an egg, rusting metal, and mixing baking soda and vinegar. The original substances are changed into completely new ones.
In contrast, a physical change does not produce a new substance. It simply results in a physical rearrangement of the atoms in an object or substance. Examples of physical changes include freezing water into ice, tearing paper, bending metal, and evaporating salt water to obtain salt. No matter how dramatic the change may appear, the underlying atomic structure remains fundamentally the same.
Examples of Chemical Changes
Chemical changes involve the formation of new chemical substances. The molecular structures of the original substances are changed and new molecules are created during a chemical change. Here are some common examples of chemical changes:
Burning: Burning is a chemical change that occurs when a substance combines with oxygen rapidly. This produces heat and light energy. The chemical composition of the substance is altered in burning. For example, when wood burns, it combines with oxygen to form carbon dioxide, water vapor and ash.
Rusting: Rusting is an example of oxidation, which is a chemical change. When iron rusts, the iron atoms react with water and oxygen to form rust, which is hydrated iron(III) oxide. Rust has a different chemical composition than pure iron.
Cooking: Cooking food involves many chemical reactions that change the molecular structures of substances. When meat is cooked, proteins denature, fats melt, and complex carbohydrates break down. New volatile molecules are created, producing the flavors and aromas of cooked food.
Digestion: Digestion is a series of chemical reactions that break down large food molecules into smaller molecules that can be absorbed and utilized by the body. Enzymes in the digestive tract catalyze these chemical changes. For example, starches are broken down into simple sugars, proteins into amino acids.
Examples of Physical Changes
Physical changes are transformations in which no new chemical substances are created. Common examples of physical changes include melting, freezing, cutting, and folding.
Melting is a physical change because even though the solid melts into a liquid, its chemical composition stays the same. Water is still H2O whether it’s frozen as ice or melted into a liquid. Freezing is also considered a physical change because while water transitions from a liquid to a solid state, it retains its chemical identity as H2O.
Cutting an object into pieces is a physical change because the material itself remains unchanged; only its shape is altered. When paper is cut with scissors, it is still made of cellulose fibers. Folding is another example of a physical change. Folding paper into a paper airplane does not change the chemical makeup of the paper.
In summary, physical changes affect the appearance of a substance but do not produce any new chemical products. Melting, freezing, cutting, and folding are all examples of transformations that are physical, rather than chemical, changes.
Light Production in Fireflies
Fireflies produce light through a chemical reaction called bioluminescence. Bioluminescence is a form of chemiluminescence where a living organism produces light through a chemical reaction.
In fireflies, bioluminescence occurs in specialized light-emitting organs called lanterns, located under their abdomens. The lantern contains chemiluminescent luciferin, the light-emitting pigment, as well as luciferase, the enzyme that catalyzes the reaction.
When a firefly wants to signal, its nervous system sends a signal to the lantern. This causes the luciferase enzyme to convert the luciferin into an excited-state form called oxyluciferin. As the oxyluciferin returns to its ground state, it emits energy in the form of light. This reaction does not produce heat, and nearly all the energy input is emitted as light.
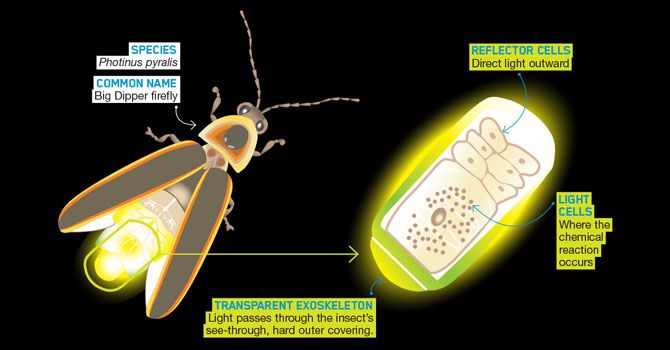
The exact color of the light depends on the structure of the luciferin molecule. In most fireflies, the light is yellow-green, although some species can produce orange, red, or green bioluminescence. Since bioluminescence in fireflies requires a chemical reaction, it is considered a chemical process rather than a physical one.
Light Production in Glow Sticks
Glow sticks produce light through a chemical reaction called chemiluminescence. Inside a glow stick are two separate compartments containing different chemicals. One compartment contains the chemical diphenyl oxalate and the other contains hydrogen peroxide.
When the glow stick is bent or snapped, the barrier between the two compartments breaks, allowing the chemicals to mix together. The hydrogen peroxide oxidizes the diphenyl oxalate, providing energy for the diphenyl oxalate molecules to reach an excited electronic state.
As the excited diphenyl oxalate molecules return to their ground state, they emit photons of light. The photons are emitted in different wavelengths giving the glow stick its colorful glow. The light emission continues as long as the chemical reaction proceeds. Once the reactants are used up, the glow stick stops producing light.
So in summary, the light production in glow sticks is a chemical process, where a chemical reaction provides energy for light emission. No combustion or burning takes place, distinguishing it from a physical process like fire.
Light Production in Stars
Stars produce light through the process of nuclear fusion. At the core of stars, hydrogen atoms fuse together under extremely high temperatures and pressures to form helium. This nuclear fusion reaction releases energy in the form of photons or light particles.
Specifically, when two hydrogen nuclei collide and fuse, a positron and a neutrino are released. The positron finds and annihilates an electron resulting in two gamma ray photons being emitted. Meanwhile the fusion of the two hydrogen nuclei produces a deuterium nucleus which rapidly fuses with another hydrogen nucleus to form a helium-3 nucleus. The helium-3 nucleus then fuses with another hydrogen nucleus to make a regular helium-4 nucleus and two more protons.
The protons interact with electrons and positrons to produce more gamma ray photons. The resulting gamma ray photons make their way from the core of the star to the surface and are emitted as visible light that we see. This entire fusion process converts some of the mass of the hydrogen fuel into the energy of starlight.
Analysis
Analyzing whether light production is a chemical or physical change requires looking at the process by which light is created and emitted in different scenarios. In fireflies, glow sticks, and stars, light production involves chemical reactions at the atomic level. Atoms and molecules interact and rearrange to create energy that is released as light.
Specifically, in fireflies, the enzyme luciferase catalyzes a reaction between luciferin, magnesium ions, ATP, and oxygen that generates an excited intermediate compound. As this compound decays to a lower energy state, it releases energy as light. The chemical bonds and structure of the reactants change to form products, indicating a chemical change has occurred.
Similarly, inside glow sticks, hydrogen peroxide and phenyl oxalate ester react to form an unstable peroxyester which then decomposes to a lower energy state, releasing energy as light. The chemical composition of the reactants is altered in the process.
In stars, nuclear fusion converts hydrogen to helium, releasing energy that initially takes the form of gamma rays which then degrade into visible light. Atoms are changed into different atomic elements, demonstrating a chemical change.
Since light production involves the rearrangement of atomic bonds and the conversion of some chemical species into others, it can definitively be categorized as a chemical change rather than a physical change.
Conclusion
After considering examples of chemical and physical changes, as well as looking closer at light production in various scenarios, it’s clear that the production of light is primarily a chemical change. While there are some instances where light can be produced through physical changes, like heating an object, the most common and brightest examples of light production occur due to chemical reactions.
In fireflies, glow sticks, and stars, light is created when chemicals combine and energy is released. The chemicals change their atomic structure to form new compounds, giving off photons in the process. Since new substances are formed, this confirms it is a chemical change. The light emitted from fire, on the other hand, is caused by heating rather than new chemical bonds.
So in summary, light production is predominately driven by chemical changes at the atomic level, with some exceptions where physical changes create forms of light through other processes like heating. When bright light occurs, you can be fairly confident a chemical reaction is taking place.
References
Woodford, Chris. “How do fireflies and glow-worms produce light?” ExplainThatStuff!, 4 Jun. 2021, https://www.explainthatstuff.com/how-fireflies-glow.html.
Ophardt, Charles E. “Chemiluminescence of Luminol.” Virtual Chembook, Elmhurst College, 2003, https://chem.libretexts.org/Bookshelves/Introductory_Chemistry/Map%3A_Introductory_Chemistry_(Tro)/12%3A_Solids_and_Liquids/12.8%3A_Chemiluminescence_of_Luminol.
Moldowan, J. Michael, et al. “The role of molecular fossils in astrobiology.” Precambrian Research, vol. 111, no. 1-4, 2001, pp. 311–329., https://doi.org/10.1016/s0301-9268(01)00154-9.
Further Reading
While this article covers the topic of whether light production is a chemical or physical change in depth, there are some other resources that readers may find interesting for learning more about this topic:
- Light and Colour – This resource from Science Learning Hub provides more details on the science behind how light is produced and perceived.
- Is Fire a Chemical or Physical Change? – An article examining if fire, another source of light production, is a chemical or physical change.
- Bioluminescence – An overview from Encyclopedia Britannica on the biological production of light in organisms like fireflies and deep sea creatures.
- Light Emission – A section from an online physics textbook explaining light emission from excited atoms.
For those interested in learning more about chemical vs. physical changes in general:
- Physical and Chemical Changes – A lesson providing an introduction to identifying chemical and physical changes.
- Examples of Chemical and Physical Changes – Lists and explains examples of different types of chemical and physical changes.
Readers looking for additional academic sources can search scientific journals and databases like Google Scholar.