Does More Electrons Mean More Electricity?
Electricity is a fundamental part of our everyday lives. From powering our homes and devices to enabling communications, transportation, and technology, electricity is essential to modern society. But what exactly causes electricity? How does electricity flow from power plants to the outlets in our walls? At the core of these questions lies perhaps the most basic query of all: do more electrons mean more electricity?
On a microscopic scale, electricity arises from the movement of tiny particles called electrons. Electrons carry a negative electric charge. The flow of these charged electrons in a unified direction produces electricity, such as the current that powers a lightbulb. This raises a logical hypothesis: more electrons must mean more electricity, right? The relationship is understandable but too simplistic. In truth, many complex factors determine conductivity and the strength of an electric current. Examining core electrical concepts helps unpack whether more electrons necessarily equate to more electricity.
Electric Current Basics
Electric current refers to the flow of electric charge. Electric charge is carried by tiny particles called electrons that orbit the nucleus of atoms. Electrons can be made to move from one place to another when an electric potential difference is applied. This potential difference provides the electromotive force that causes electrons to flow through a conductor. This electron flow is what we call electric current.
Electrons are negatively charged, so they will flow towards a positive electric potential and away from a negative potential. The potential difference is measured in volts (V). A higher voltage means a greater potential difference, which will cause more electrons to flow. So in a simplified sense, more electrons flowing means a higher electric current.
Electrons and Electric Charge
Electricity is the flow of electric charge, which is carried by tiny particles called electrons. Atoms contain equal amounts of positively charged protons and negatively charged electrons, making them electrically neutral overall. However, electrons can become unbalanced and separate from their atoms, allowing them to move freely.
Electrons carry a negative electric charge. When electrons build up in one area, it becomes negatively charged, while a lack of electrons results in a positive charge. The flow of electrons is what we call electricity. Metals are good conductors of electricity because they contain many loose electrons that can move freely when voltage is applied.
The more excess electrons there are in a conductive material like a wire, the more electric current that can flow through it. So in a sense, more electrons do mean more electricity. However, there are other factors that affect conductivity, like material composition and temperature. But in general, increasing the number of free electrons allows more electric charge to flow.
Ohm’s Law
Ohm’s law is one of the basic relationships that describes electrical conductivity. It states that the current (I) flowing through a conductor is directly proportional to the voltage (V) applied across it, for any given temperature. This relationship is expressed by the equation:
I = V/R
Where R is the resistance of the conductor material, measured in ohms. Resistance is a property of the material that impedes the flow of electric current. So for any given voltage, higher resistance leads to lower current, while lower resistance leads to higher current.
Ohm’s law is important because once you know two of the variables (voltage, current, resistance), you can calculate the third. This allows us to predict and control the flow of electricity in circuits. The resistance of materials is generally constant over a wide range of voltages. So if we increase the voltage, the current will increase proportionally.
Resistivity and Conductivity
Materials can be classified as conductors or insulators based on how easily electrons can flow through them. In conductors like metals, electrons flow freely, allowing electric current to pass through easily. In insulators like rubber, electron flow is difficult, making them resistant to electric current.
Resistivity is a measure of how strongly a material opposes electric current. Materials with high resistivity (like insulators) have a large resistance to current flow. Materials with low resistivity (like conductors) allow current to flow more freely.
Conductivity is the inverse of resistivity – it measures how easily current passes through a material. Highly conductive materials like copper have many free electrons that can carry current. Less conductive materials like glass have fewer free electrons, inhibiting current flow. Conductivity depends on properties like a material’s electron configuration, temperature, and purity.
By understanding resistivity and conductivity, we can predict how easily electricity will flow through different materials. This allows us to design circuits and components that utilize conductors and insulators in ways that control electrical current.
Superconductors
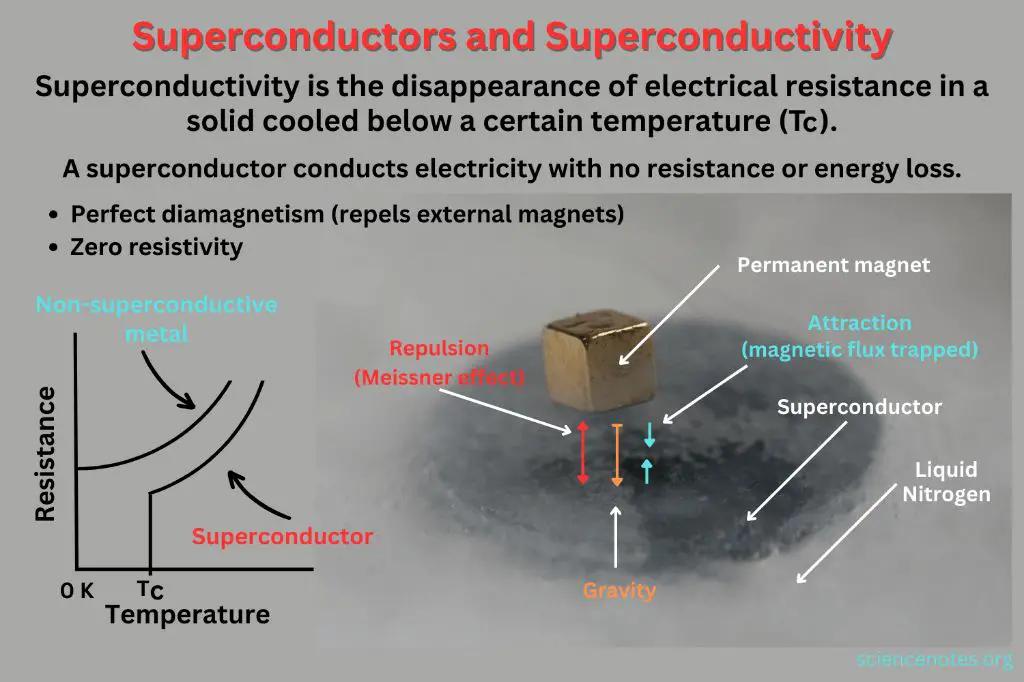
Superconductors are materials that exhibit zero electrical resistance and the ability to expel magnetic fields when cooled below a critical transition temperature. In a normal conductor, resistance arises from collisions between electrons and ions in the material. However, in a superconductor, electrons form pairs known as Cooper pairs, which are able to move through the material without resistance.
The transition temperature at which a material becomes superconducting depends on the material composition and structure. Superconductivity was first discovered in mercury in 1911 at 4 Kelvin (-269°C). Since then, high-temperature cuprate superconductors have been developed with transition temperatures over 130 K (-143°C). This allows superconductors to operate using cheaper liquid nitrogen cooling rather than liquid helium.
The zero resistivity of superconductors allows electrical current to persist indefinitely with no applied voltage. Superconductors are used to make powerful electromagnets and for applications like MRI machines, particle accelerators, magnetic levitation trains, and ultra-sensitive devices for measuring magnetic fields or radio frequencies. They have the potential to greatly improve energy efficiency if used for power transmission lines or generators.
Semiconductors
Semiconductors are materials that have conductivity properties between conductors like metals and insulators like glass or rubber. This means semiconductors can act as either insulators or conductors depending on conditions.
In their natural state, semiconductors have a small number of free electrons that can carry electric current. However, they can be “doped” with impurities to greatly increase the number of free electrons or “holes” to improve conductivity.
Semiconductors are used to make transistors, integrated circuits, solar cells, LEDs, and other electronic devices. By controlling the doping and structure of semiconductors, engineers can fine-tune their electrical properties for different applications.
Some common semiconductor materials are silicon, germanium, gallium arsenide, and graphene. The conductivity of semiconductors can be increased by raising temperature or exposure to light, allowing them to switch between insulating and conducting states.
Overall, semiconductors have intermediate conductivity that allows them to transition between conductor and insulator behavior. This makes them extremely useful for controlling electric current in electronic devices.
Conductivity in Metals
Metals tend to have very high electrical conductivity compared to other materials. This is because of their unique atomic structure. Metals contain a sea of free electrons that are not bound to any particular atom. These free electrons can move easily through the atomic lattice of the metal when an electrical voltage is applied. The free electrons serve as charge carriers, transporting electric charge through the metal very efficiently.
In most materials, electrons are tightly bound to their atoms and cannot move freely. But in metals, the outermost electrons are loosely bound and can jump from atom to atom with very little resistance. This ability for free electrons to flow makes metals highly conductive. The more free electrons a metal contains, the higher its conductivity generally is. Silver, copper, gold, and aluminum are examples of very conductive metals with a high concentration of free electrons.
Other Factors Affecting Conductivity
While the number of free electrons is a key factor in determining the conductivity of a material, it is not the only factor. Other aspects can also affect how well a material conducts electricity.
One such factor is temperature. For metals, conductivity generally decreases as temperature increases. This is because higher temperatures cause more vibrations of the metal atoms, which interferes with electron mobility. So even if a metal has a high number of free electrons, its ability to conduct electricity will be reduced at higher temperatures.
The cross-sectional area of the conducting material also matters. For a given material, a larger cross-sectional area provides more room for electron flow, increasing conductivity. This is why wires with larger diameter tend to have higher conductivity than thinner wires of the same metal.
In summary, while the number of free electrons plays a major role, factors like temperature and physical dimensions can also affect the conductivity of a material. So more free electrons alone does not necessarily mean more electricity. The interaction between electrons and the material’s atoms, as well as the available area for electron flow, must also be considered.
Conclusion
In summary, the number of free electrons in a material does affect its conductivity and ability to carry an electric current. Materials like metals contain many free electrons that can move through the material, allowing electricity to flow more easily. The more free electrons in a material, the higher its conductivity.
However, resistivity also plays a key role. Even materials with lots of free electrons can have high resistivity that inhibits electron flow. Resistivity depends on factors like the structure and temperature of the material. Substances like superconductors have very low resistivity, allowing electricity to flow freely even though they may not have the most free electrons.
So while more free electrons allows for more electric current, the intrinsic resistivity of the material also matters. Both factors need to be considered when determining the conductivity of a substance and its ability to transmit electricity.