Does Heat Flow From High To Low Energy?
Heat flow refers to the transfer of thermal energy from one object or system to another as a result of their temperature difference. Objects and systems naturally tend towards thermal equilibrium, which is the state where interacting objects or systems are at the same temperature and no net heat transfer occurs between them.
The key principle that governs heat flow is the 2nd Law of Thermodynamics. This law states that heat will spontaneously flow from hotter objects to colder objects, or more scientifically, from higher energy states to lower energy states. So heat flows from high energy (high temperature) to low energy (low temperature).
This allows us to define high and low energy – high energy refers to higher temperatures, while low energy refers to lower temperatures. Heat always flows from high to low temperature objects until equilibrium is reached. The 2nd Law of Thermodynamics governs this behavior of heat flow and energy transfer on the microscopic scale.
Heat Flow Basics
There are three main mechanisms by which heat flows between objects or regions:
Conduction occurs when heat transfers between objects or regions that are in direct contact with each other. The better the conductor, the more rapidly heat will flow. Metals are good conductors. Conduction happens as higher energy particles collide with lower energy particles, transferring kinetic energy between them.
Convection is the heat transfer due to the bulk movement or circulation of a fluid (liquid or gas). Hotter regions of the fluid become less dense and rise, while cooler regions sink, creating circulation patterns that transfer heat. Convection occurs in liquids like water or air.
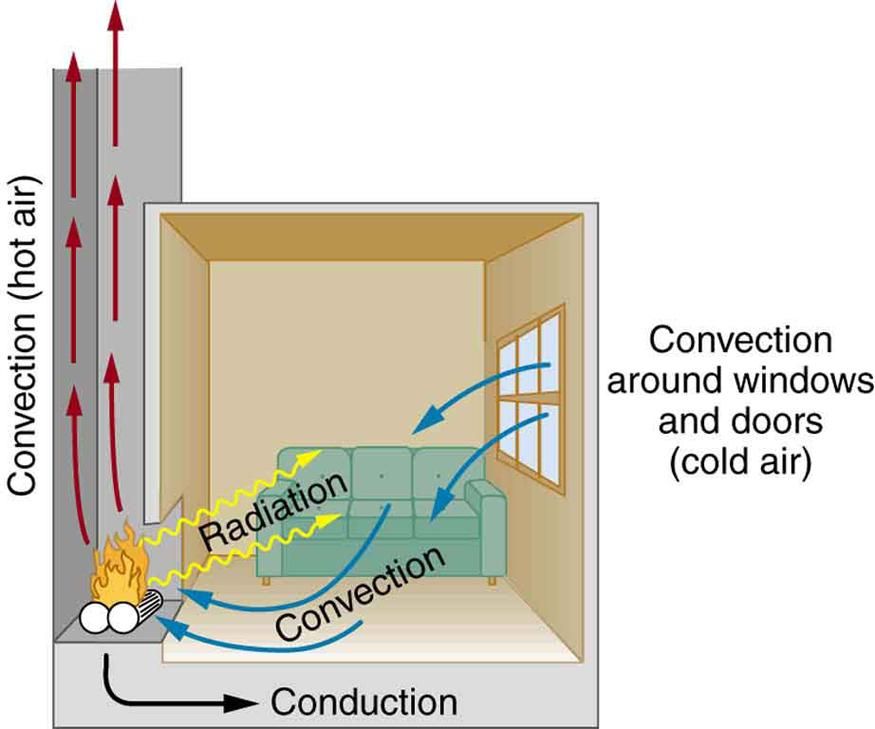
Radiation is the transfer of heat via electromagnetic waves, like infrared radiation. All objects emit thermal radiation related to their temperature. Hotter objects emit more intense radiation than cooler objects. Radiation does not require direct contact between objects, it can travel through empty space.
Thermal Equilibrium
Thermal equilibrium refers to the state where two objects that are in contact with each other are at the same temperature. When two objects at different temperatures come into contact, heat will flow from the object at higher temperature to the object at lower temperature until the temperatures equalize.
For example, when you add hot water to a cold bath, the hot water will transfer heat to the cold water until both reach the same intermediate temperature. The net heat flow is from areas of high temperature to areas of low temperature, as the system tries to reach thermal equilibrium where the objects are at the same temperature.
This process occurs because heat is a form of energy, and systems tend to move towards lower energy states. Transferring heat from high to low temperature allows the total energy in the system to spread out and become more uniform. Thermal equilibrium is the lowest energy state for the combined system.
2nd Law of Thermodynamics
The 2nd law of thermodynamics explains why heat flows from high energy to low energy. This law states that the total entropy of an isolated system can never decrease over time. Entropy is a measure of disorder and randomness. An isolated system will tend to evolve toward thermodynamic equilibrium, which is the state of maximum entropy. At equilibrium, the energy in the system is distributed uniformly.
Since energy naturally spreads out, heat will flow spontaneously from higher concentrations of energy to lower concentrations. For example, if you put a hot object next to a cold object in a thermally isolated room, heat will flow from the hot object to the cold object until they reach the same temperature. The hot object has higher thermal energy, and the cold object has lower thermal energy. Heat moves from high (hot) to low (cold) energy states.
The 2nd law explains why it is impossible for heat to flow from a cold object to a hot object without an external energy source. The natural direction of heat flow is from hot to cold. This is why a cup of hot coffee cools down to room temperature over time, rather than heating up the room. In summary, the 2nd law of thermodynamics mandates that heat flows from high energy to low energy.
Real-World Examples
Heat flow is happening all around us every day. Here are some common examples of heat transferring from high to low energy:
– A hot cup of coffee left on a table will transfer heat to the surrounding air, cooling down over time while the air heats up slightly.
– An oven cooking food transfers heat energy to the colder food inside, raising its temperature.
– On a cold day, the higher energy heat from your warm body will flow to the lower energy cold air, making you feel chilled.
– A just-used clothes iron left sitting on an ironing board will transfer its higher heat energy to the board, slowly cooling down as the board absorbs the heat.
– Sunlight striking a solar panel heats it up as higher energy photons are absorbed, allowing electricity to be generated.
– An automobile engine running hotter than the surrounding air will have heat flow out through the metal engine block and radiator into the lower temperature environment.
– Steam rising from a pot of boiling water on the stove is heat energy transferring into the cooler air of the room.
Heat Flow Direction
Heat generally flows from high energy to low energy systems until an equilibrium is reached. This means that heat will naturally transfer from high temperature objects and regions to low temperature objects and regions. For example, heat will flow from a hot cup of coffee into the surrounding room until the coffee and room reach the same temperature.
This heat flow occurs due to temperature gradients. Heat moves down the temperature gradient, from high to low, seeking to minimize the gradient over time. So in the coffee example, the large temperature difference between the hot coffee and cool room creates a gradient that drives the heat transfer. As the coffee cools and the room warms up, the gradient gets smaller until both reach the same uniform temperature.
On a molecular level, heat is the kinetic energy of molecules. Hotter objects have faster moving molecules with more kinetic energy. When a hot and cold object interact, the higher energy molecules collide with the lower energy molecules, transferring kinetic energy and effectively heating up the colder object. This continues until the molecular kinetic energies reach equilibrium.
So in summary, heat predictably flows from higher temperature/higher energy systems to lower temperature/lower energy systems due to temperature gradients and molecular kinetics. Understanding this basic high to low directionality of heat flow allows us to engineer more efficient heat transfer systems and make accurate thermodynamic calculations.
Exceptions to the Rule?
While the general rule is that heat flows from higher to lower temperature, there are some notable exceptions and caveats to consider:
Superconductors: In superconducting materials, heat flow can be anomalous and may not always flow “downhill” from higher to lower temperatures due to quantum effects.
Laser cooling: Under special laboratory conditions, lasers can be used to cool objects below the temperature of their surroundings, temporarily causing heat to flow “uphill.”
Transient heat flow: When systems are out of thermal equilibrium, heat may temporarily flow from cold to hot as the system approaches equilibrium.
Small scale effects: At very small microscopic scales, random thermal fluctuations can cause short-lived hot and cold spots, leading to momentary reverse heat flow.
Quantum effects: Quantum tunneling and other phenomena can lead to exceptions to classical heat flow rules in nanoscale systems.
While macroscopically heat predominantly flows downhill from hot to cold, these examples illustrate special cases where more complex physics can come into play.
Impact and Applications
The second law of thermodynamics has immense importance and a wide range of real-world applications. Here are some of the key impacts and uses of this fundamental law of physics:
Thermodynamic efficiencies – The second law sets fundamental limits on the maximum efficiency of heat engines and refrigeration cycles. This allows us to properly evaluate and optimize real heat transfer systems.
Entropy calculations – We can use the second law’s focus on entropy to calculate the entropy change in chemical, thermal, and other processes. This provides insights into spontaneity and equilibrium.
Life processes – Living organisms rely on low entropy inputs and high entropy waste outputs to sustain organized states. The second law explains why this flow is essential for life.
Evolution of the universe – The second law points to an overall increase in entropy as the universe evolves. This helps describe the arrow of time and origins of structure in the cosmos.
Technology advances – Advances that convert thermal energy to work more efficiently or recapture wasted heat rely critically on the second law.
In summary, the second law of thermodynamics and concept of entropy are hugely significant in physics, engineering, chemistry, biology, cosmology, and beyond.
Future Research
Heat transfer remains an active area of scientific research. Scientists continue to develop more advanced mathematical models and simulations to better understand heat flow. Microscale and nanoscale heat transfer are cutting-edge research topics with applications in microelectronics, thermoelectrics, and energy conversion. Researchers are also studying how to manipulate heat flow in ways that could lead to improved thermal management technologies. Phase change materials, thermally conductive materials like graphene, and metamaterials with designed thermal properties are also being explored. Work is ongoing to develop methods of thermal imaging with greater resolution and sensitivity. Real-world testing of heat flow theories under extreme conditions is another important research direction. Overall, heat transfer science continues to evolve through the dedicated efforts of researchers around the world.
Conclusion
In summary, heat does tend to flow from higher temperature objects to lower temperature objects until thermal equilibrium is reached. This aligns with the 2nd law of thermodynamics, which states that the entropy of an isolated system can never decrease over time. While there are some exceptions, such as with refrigerators and air conditioners, overall heat flow follows the rule of going from high energy to low energy. This principle impacts everything from your morning coffee cooling down, to the Earth radiating heat into space. Additional research on manipulating heat flow could lead to more efficient energy usage and innovative technologies in the future.
To conclude, the answer to the original question is yes, heat does primarily flow from high energy to low energy objects and systems, with some interesting exceptions.