Which Is The Best Example Of How Electromagnetic Energy?
Electromagnetic energy is a form of energy that is created by oscillating electric and magnetic fields. It is one of the most common forms of energy we regularly encounter. Electromagnetic energy refers to both light energy and radio waves on the electromagnetic spectrum. The electromagnetic spectrum encompasses all types of electromagnetic radiation, from radio waves to gamma rays. All forms of electromagnetic energy exhibit wave-like properties and travel at the speed of light, which is approximately 186,000 miles per second.
In its most basic form, electromagnetic energy is created when an electric field couples with a magnetic field. An electric field is produced by a charge, while a magnetic field is produced by the motion of a charge, which creates a current. When an electric field varies, it produces a varying magnetic field. Likewise, a changing magnetic field generates a changing electric field. These oscillating fields transmit energy through space in the form of electromagnetic waves. This allows electromagnetic energy to travel immense distances, even from millions of light years away.
Types of Electromagnetic Energy
Electromagnetic energy exists in many different forms that have a wide range of applications in our everyday lives. Some of the main types of electromagnetic energy include:
Visible Light
Visible light is the electromagnetic energy that is detectable by the human eye. It makes up the portion of the electromagnetic spectrum with wavelengths between 380-700 nanometers. Visible light allows us to see the world around us through illumination and reflection off objects.
Radio Waves
Radio waves have the longest wavelengths in the electromagnetic spectrum, ranging from around 1 millimeter to hundreds of kilometers. They are widely used for communications through radio, television, mobile phones, WiFi, Bluetooth, and more.
Microwaves
Microwaves have wavelengths from around 1 millimeter up to 1 meter and oscillate at frequencies between 300 MHz and 300 GHz. They are commonly used for point-to-point communications, radar, and heating in microwave ovens.
Infrared
Infrared waves are located just beyond the red end of the visible spectrum, with wavelengths from about 700 nanometers to 1 millimeter. Infrared radiation is used for thermal imaging, weather satellites, spectroscopy, remote controls, and more.
Ultraviolet
Ultraviolet (UV) light sits between visible violet light and x-rays on the spectrum, with wavelengths from 10 to 400 nanometers. It has uses including disinfection, fluorescence, detecting UV damage, and in blacklights.
X-Rays
X-rays have wavelengths shorter than ultraviolet light but longer than gamma rays, generally between 0.01 to 10 nanometers. They are useful for medical imaging, material science, astronomy, and security screening applications.
Gamma Rays
Gamma rays occupy the highest frequency end of the electromagnetic spectrum with the shortest wavelengths, less than 0.01 nanometers. They are produced by nuclear reactions and high-energy events in space, and used in cancer treatment and imaging.
Properties
Electromagnetic energy is characterized by its wavelength, frequency, speed, and energy. These properties are all interrelated:
Wavelength refers to the distance between successive wave peaks. It is commonly represented by the Greek letter lambda (λ). Wavelength determines how electromagnetic radiation interacts with matter. Longer wavelengths (lower frequencies) tend to penetrate matter better. Shorter wavelengths (higher frequencies) tend to get blocked or absorbed more easily.
Frequency refers to how many wave cycles pass a fixed point per unit of time. It is commonly measured in Hertz (Hz), which is cycles per second. Frequency is inversely proportional to wavelength – higher frequencies have shorter wavelengths.
Speed is the rate at which electromagnetic waves propagate through space. All electromagnetic radiation, regardless of wavelength or frequency, travels through a vacuum at the speed of light – approximately 3 x 10^8 meters per second.
Energy is directly proportional to frequency, so higher frequency electromagnetic radiation carries more energy per photon. The energy of photons is quantized and depends on their frequency according to Planck’s equation (E=hf).
Generation
Electromagnetic energy is generated when charged particles are accelerated. This acceleration can come from oscillating electric charges or the movement of electric charges in a magnetic field.
Electric charges that oscillate or accelerate emit oscillating electric and magnetic fields. These oscillating fields propagate outward from the accelerating charges as electromagnetic waves. Common ways to accelerate electric charges and generate electromagnetic waves include:
- An antenna in which oscillating electric currents are applied.
- The motion of electrons in the electron beam of a cathode ray tube TV or computer monitor.
- The motion of electrons in a wire as they “fall” from higher to lower atomic orbitals and emit light (as in a neon light).
So in summary, electromagnetic energy arises whenever charged particles undergo acceleration, producing oscillating electric and magnetic fields that radiate outward. Manipulating and accelerating charges allows us to generate many useful types of electromagnetic energy.
Applications
Electromagnetic energy has many important practical applications in modern technology. Some of the most significant uses are in communications, imaging, spectroscopy, medicine, and manufacturing.
In communications, electromagnetic waves are used to transmit information over long distances. Radio, television, cell phones, and wireless internet all rely on encoding information onto carrier waves in the radio frequency portion of the spectrum. Fiber optic communication uses pulses of infrared light transmitted through glass fibers.
Medical imaging techniques like X-rays, MRI, and PET scans allow us to see inside the human body by detecting different forms of electromagnetic radiation. These important diagnostic tools have revolutionized medicine.
Spectroscopy analyzes the unique spectrum of electromagnetic radiation emitted or absorbed by materials to study their composition and properties. Various types of spectroscopy are used in chemistry, physics, and astronomy.
In manufacturing, microwave energy can be used to heat and cure materials, while lasers have many applications in cutting, welding, and precision machining of products. Electromagnetic radiation is also used for process control and quality assurance.
Visible Light
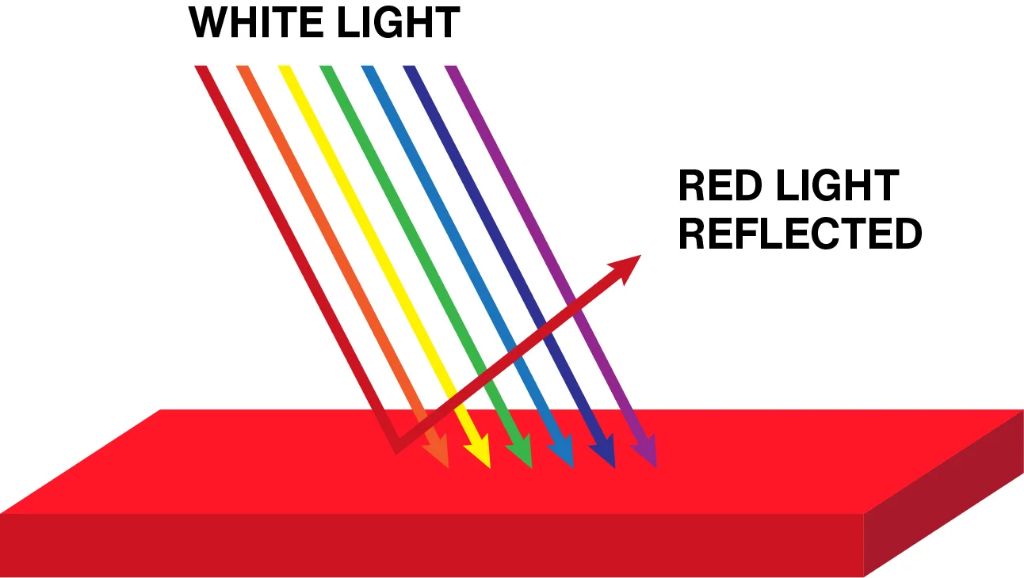
Visible light is the range of electromagnetic radiation that can be perceived by the human eye. It occupies wavelengths from approximately 380 to 740 nanometers. The human eye contains photoreceptor cells called rods and cones that respond to different wavelengths of light. The different responses of these photoreceptors to light allow our eyes to see different colors.
The eye’s perception of color is determined by the combination of wavelengths reflected off objects and entering the eye. For example, visible light with wavelengths around 700 nm appears red, while light around 500 nm appears green. The three primary colors in human color vision are red, green, and blue. By mixing different amounts of these three colors, our eyes can perceive a wide array of other colors.
Visible light plays an essential role in human vision and illuminating our surroundings. We rely on visible light from the sun and artificial light sources to see the world around us. Our eyes have evolved to be highly sensitive to visible light, as this range of wavelengths carries the most visual information in natural daylight conditions on Earth.
Overall, visible light represents a very small slice of the full electromagnetic spectrum, yet it is a profoundly important one for us. Our experience of color and sight depends fundamentally on those wavelengths our eyes can detect as visible light.
Infrared Radiation
Infrared radiation, sometimes simply called infrared, refers to electromagnetic waves with wavelengths longer than visible light but shorter than microwaves. Although infrared is invisible to the human eye, we can feel this type of radiation in the form of heat. There are several important applications of infrared radiation.
Thermal Imaging
Infrared cameras can detect infrared radiation emitted or reflected from objects, producing images called thermograms. Because infrared radiation is emitted by all objects based on their temperatures, thermograms allow you to see variations in temperature. Thermal imaging cameras are used for applications like night vision, surveillance, inspection, and firefighting.
Spectroscopy
Infrared spectroscopy takes advantage of the fact that molecules absorb specific frequencies of infrared radiation. By analyzing the wavelengths absorbed by a material, its molecular composition can be identified. Infrared spectroscopy is commonly used in chemistry, astronomy, industry, and medicine.
Remote Controls
Most remote controls for electronics like televisions, DVD players, and garage door openers rely on infrared LEDs to transmit signals. These LEDs emit modulated infrared light detected by sensors on the device being controlled. Infrared remote controls allow wireless control without interfering with visible light.
Microwaves
Microwaves are a type of electromagnetic radiation that have wavelengths ranging from about one millimeter to one meter. Some key uses of microwaves include heating, radar, and communications.
One of the most common uses of microwaves is for heating food. Microwave ovens work by emitting microwaves that interact with water molecules in food, causing them to vibrate rapidly and generate heat. This allows food to be cooked very quickly. Only certain materials that contain water molecules, like food, absorb microwave radiation, which is why the containers in microwave ovens stay cool.
Microwaves are also used in radar technology. Radar systems emit pulses of microwave radiation and analyze the reflected signals to detect the presence, location, and speed of objects like aircraft and weather formations. The short wavelength of microwaves allows radar to detect small objects and provide high resolution.
In communications, microwaves allow for the transmission of information over long distances. Microwave frequencies are used for radio, television, mobile phones, satellite communications and wireless networking. Compared to longer radio waves, microwaves can carry more information by supporting a higher data transmission rate.
X-Rays
X-rays are a form of high-frequency electromagnetic radiation with wavelengths that range from about 10 to 0.01 nanometers. They have a wide variety of useful applications, particularly in the medical and security fields.
One of the most common uses of X-rays is in medical imaging. When X-ray beams pass through the body, they are absorbed in different amounts depending on the density of the material they pass through. This allows X-rays to create images of bones and organs, showing contrasts between soft and hard tissues. Doctors use X-ray imaging for a variety of diagnostic purposes such as detecting fractures, pneumonia, cancer, and even dental cavities. Medical imaging accounts for the majority of total global X-ray use.
X-rays are also commonly used for security screening purposes. Airport security checkpoints often employ X-ray scanners to detect weapons and other prohibited items within luggage and bags. Customs agencies also use X-rays to examine cargo containers and vehicles crossing borders to uncover contraband. The ability of X-rays to penetrate materials makes them ideal for seeing inside objects without physically opening them.
In addition, X-rays are invaluable for crystallography, the study of crystal structures and atomic spacing. When X-ray beams interact with crystalline materials, they create distinctive diffraction patterns that reveal information about the crystal shape, size, and orientation of molecules. Analyzing these patterns enables determination of molecular structures, which is hugely important across physics, chemistry, biology, and materials science.
Thanks to their unique properties, X-rays have become indispensable tools for medicine, security, and science. Their ability to pass through matter revolutionized medical imaging and border security, while also providing insight into the atomic structure of materials.
Conclusion
In summary, electromagnetic energy encompasses many different types of waves and radiation that are part of the electromagnetic spectrum. From radio waves to gamma rays, these energies differ in wavelength, frequency, and energy levels. While we cannot see most forms of electromagnetic radiation, they have many important properties and uses that impact our daily lives. Understanding the fundamentals of electromagnetic energy is key to advances in telecommunications, medical imaging, computing, and many other fields. Appreciating the broad spectrum of these invisible energies around us provides insight into how our increasingly technology-driven world functions on a fundamental scientific level.
The importance of electromagnetic energy in both nature and engineered applications cannot be overstated. Our ability to harness various wavelengths for information transmission, medical diagnosis, power generation, and innumerable other uses underpins key technologies we rely on today. As our scientific understanding progresses, we continue finding new ways to utilize the electromagnetic spectrum to benefit society. Whether through increasingly powerful broadband networks, advanced medical scans, or more efficient photovoltaic solar power, the possibilities from electromagnetic radiation seem endless. By maintaining a curiosity and appreciation for these unseen energies, we ensure continued innovation and an improved future for generations to come.