When Energy Is Transformed From One Form To Another What Is Released?
Forms of Energy
Energy comes in many forms that can be categorized into two main types: potential energy and kinetic energy. Potential energy is stored energy that has the potential to do work, while kinetic energy is energy in motion. The main forms of energy include:
- Potential Energy
- Chemical – stored in the bonds between atoms and molecules
- Nuclear – stored in the nucleus of an atom
- Gravitational – stored in an object’s height
- Elastic – stored in compressed or stretched objects
- Kinetic Energy
- Radiant – energy in electromagnetic waves like light
- Thermal – energy from the kinetic motion of particles
- Motion – energy of moving objects
- Electrical – energy from electric current flow
Energy is constantly being converted from one form to another. For example, chemical energy in gasoline is converted to kinetic energy to move a car forward. Understanding the different forms energy takes is key to harnessing energy efficiently.
Energy Transformation
Energy transformation refers to the change of energy from one form into another. This occurs constantly in the universe as energy flows and circulates. According to the law of conservation of energy, the total energy in a closed system remains constant. Energy is never created or destroyed but merely transformed.
A common example of energy transformation is the chemical potential energy stored in the bonds of molecules changing into thermal energy. This occurs through exothermic reactions, where chemical reactions release heat. For instance, when wood burns, the chemical energy stored in the wood’s organic molecules transforms into fire’s thermal energy and light. Another example is cellular respiration, where cells break down glucose to harness its stored chemical energy, releasing some thermal energy in the process.
Other types of energy transformations include electrical energy converting into light and thermal energy in appliances and electronics. Mechanical energy changing into thermal energy due to friction is also a form of energy transformation. The possibilities for energy converting from one type to another are endless, but the total quantity of energy stays the same.
Law of Conservation of Energy
The law of conservation of energy states that energy can neither be created nor destroyed, only transformed from one form to another. This means the total energy in an isolated system always remains constant.
For example, when a freight train brakes, the kinetic energy of the train is transformed into thermal energy in the form of heat due to the friction from the brake pads rubbing against the wheels. The total amount of energy before and after the train brakes is the same, but the form of energy changes from kinetic to thermal.
This law is fundamental to understanding energy transformations. When energy changes form, the quantity of energy stays the same. The energy is neither lost nor gained, but simply converted into a new type of energy. This principle applies across all energy transformations, from chemical reactions to photosynthesis to the metabolism of food in our bodies.
The law of conservation of energy is a key concept that underlies the study of thermodynamics. It provides a mathematical framework for analyzing energy transformations and calculating the efficiency of energy conversion processes. This law represents one of the most fundamental laws of physics and is considered to be inviolable.
Heat and Work
When energy is converted from one form to another in a closed system, two main ways that energy is transferred are through heat and work. Heat is energy transferred between objects or systems due to a temperature difference. Work is energy transferred when a force causes an object or system to move through a distance. For example, the chemical energy stored in gasoline is converted into heat and work when burned in a car engine. The exploding gasoline applies force to move the pistons, doing mechanical work. Heat is also produced which is absorbed by the engine and exhaust. The total energy before and after remains the same, even though the energy changes form. This demonstrates the law of conservation of energy.

Exothermic Reactions
An exothermic reaction is a chemical reaction that releases energy in the form of heat. During exothermic reactions, energy is transferred from the chemical bonds in the reactants to the surroundings. This results in the products having less energy than the reactants.
Some examples of exothermic reactions include:
- Combustion – When a hydrocarbon fuel like methane or gasoline burns in oxygen, a large amount of heat is released.
- Thermite reaction – Iron oxide and aluminum react to form aluminum oxide and iron. This reaction reaches temperatures over 2000°C.
- Acid-base neutralization – When an acid and a base react, water and a salt are formed. For example, when hydrochloric acid reacts with sodium hydroxide, water and sodium chloride are produced.
The energy released by exothermic reactions can be seen in the form of light, sound, motion, and heat. Exothermic reactions play an important role in chemical processes that generate energy, like batteries and metabolism. The energy released is proportional to how exergonic the reaction is.
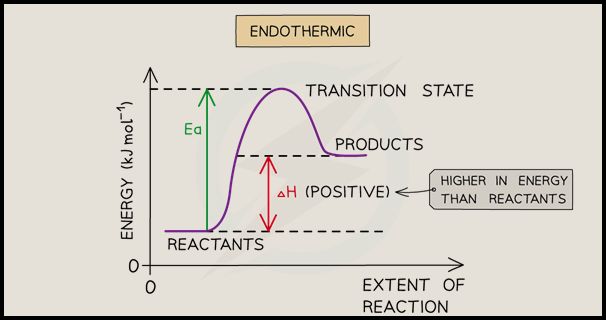
Endothermic Reactions
Endothermic reactions are chemical reactions that absorb heat energy from the surroundings. These reactions require an input of energy to proceed, unlike exothermic reactions which release energy. The absorption of heat by endothermic reactions leads to a decrease in temperature of the surroundings.
Some examples of endothermic reactions include:
- Thermal decomposition reactions – where a chemical compound breaks down into simpler substances upon heating. For example, calcium carbonate (CaCO3) decomposing into calcium oxide (CaO) and carbon dioxide (CO2).
- Neutralization reactions between an acid and a base – such as between hydrochloric acid (HCl) and sodium hydroxide (NaOH) to form water (H2O) and sodium chloride (NaCl).
- Solvation processes – when solids dissolve into a solvent, such as table salt (sodium chloride) dissolving in water.
- Photosynthesis in plants – where carbon dioxide (CO2) and water (H2O) are converted into glucose (C6H12O6) and oxygen (O2), using energy from sunlight.
In all these examples, heat energy is absorbed from the surroundings to break down chemical bonds, dissolve substances, or enable reactions to proceed. This makes them endothermic processes.
Energy Conversion Efficiency
Energy conversion efficiency refers to the ratio of useful output energy to the total input energy in a system. When energy transforms from one form to another, some energy is often lost or wasted, usually in the form of heat. The efficiency of a system indicates how much of the initial energy is actually converted into usable work or output energy.
For example, in an electrical power plant, the chemical energy stored in fuel is converted into electricity. However, not all the chemical energy can be transformed into electrical energy. Some is lost as waste heat during combustion and energy transformations. The energy conversion efficiency of a power plant refers to the percentage of the fuel’s energy content that is actually converted into electrical energy. A higher percentage indicates greater efficiency.
Improving energy efficiency is an important strategy for conserving energy resources and reducing energy waste and environmental impacts. More efficient systems need less input energy to produce the same amount of useful output energy. By maximizing efficiency, less fuel and energy need to be consumed to achieve the same tasks and processes.
Wasted Energy and Heat
When energy transformations occur, there is often a significant amount of wasted energy released as heat. This happens because most energy transformations are not 100% efficient. For example, when burning gasoline in a car engine, only about 25% of the chemical energy in the gasoline is converted into mechanical energy to move the car. The other 75% is released as heat and sound energy, which is not harnessed and is essentially wasted.
The same is true for other energy transformations like converting electrical energy into light in an incandescent light bulb – only about 10% of the initial electrical energy gets converted into light, while the other 90% is given off as heat. This waste heat is a natural byproduct of irreversible processes and limits how much useful energy we can extract. Thermodynamics tells us that we cannot convert 100% of input energy into useful output energy in any real-world transformation.
This wasted heat is released into the environment and contributes to global warming if it comes from carbon-based fuels like coal, oil, and natural gas. Finding ways to utilize waste heat, instead of releasing it unused into the atmosphere, is an important consideration for improving overall energy efficiency and reducing environmental impacts.
Implications and Impact of Wasted Energy
When energy transformations are inefficient, significant amounts of heat and wasted energy are produced. This wasted energy can have major environmental and climate impacts.
The greenhouse gases emitted from the burning of fossil fuels like coal, oil and natural gas are a major contributor to global warming and climate change. As energy demand rises globally, more greenhouse gases are pumped into the atmosphere. Increased temperatures lead to melting ice caps, rising sea levels, more extreme weather events, mass extinctions and destroyed ecosystems.
Wasted energy in the form of heat pollution also disrupts local environments. Thermal pollution from industries, vehicles and appliances exhausts large amounts of heat that warm rivers, lakes and oceans. This affects oxygen levels and harms aquatic life. Urban heat islands caused by dark paved surfaces, buildings and wasted heat alter weather patterns and rainfall in cities.
Improving energy efficiency with better technology, renewable energy, and conservation practices can help curb greenhouse gas emissions and thermal pollution. Wise energy use benefits the environment, climate, ecosystems and public health. More efficient energy transformations reduce resource and financial costs as well.
Improving Efficiency
There are various ways we can improve the efficiency of energy transformations and reduce wasted energy:
- Proper insulation in homes and buildings to prevent heat loss
- Energy-efficient appliances and lighting
- Waste heat recovery systems to capture and reuse excess heat
- Preventative maintenance on equipment and systems
- Using combined heat and power systems to utilize waste heat
- Upgrading to higher efficiency heating and cooling systems
- Improving manufacturing processes to minimize energy waste
- Using renewable energy sources that do not release waste heat
- Educating consumers and businesses on energy efficiency practices
- Implementing energy standards and regulations for appliances and equipment
With thoughtful design and proper implementation of these methods, the efficiency of energy conversions can be optimized, reducing wasted energy and its impacts on the environment.