What Process Can Produce Chemical Energy?
Chemical energy is the energy that can be derived from the chemical bonds present in molecules. It is particularly important because living organisms rely on molecules like carbohydrates, lipids, and proteins from food to provide the chemical energy needed to power life processes. Without chemical energy, organisms would not be able to grow, move, reproduce, and maintain homeostasis.
Chemical energy enables metabolic processes to occur by driving chemical reactions that break and form chemical bonds. Some common examples of chemical energy include the energy stored in fuels like gasoline and natural gas, batteries, and the food we eat. When chemical bonds are broken during chemical reactions, chemical energy is released which can then be harnessed to do useful work. The breakdown and formation of chemical bonds are key to life processes, as chemical energy is required for muscle contraction, nerve impulse propagation, biosynthesis, and transport of molecules across cell membranes.
Photosynthesis
Photosynthesis is the process by which plants and some bacteria and algae convert sunlight into chemical energy that can be used by the organism for growth and metabolism. During photosynthesis, carbon dioxide and water are converted by sunlight into glucose (a sugar) and oxygen. The glucose provides the energy that allows plants to grow, and the oxygen is released as a byproduct.
The overall chemical reaction for photosynthesis is:
6CO2 + 6H2O + sunlight –> C6H12O6 + 6O2
The process of photosynthesis occurs in two main stages: light reactions and the Calvin cycle. During light reactions, photons of light excite electrons in chlorophyll, generating ATP and NADPH, which are used in the Calvin cycle to fix carbon into sugar. The light reactions take place in the thylakoid membranes in chloroplasts, while the Calvin cycle reactions take place in the stroma.
Photosynthesis is vital for nearly all life on Earth. The process converts the energy in sunlight to chemical energy in food, providing the base of most food chains and fueling ecosystems. Additionally, the oxygen released as a byproduct is essential for most organisms’ respiration.
Cellular Respiration
Cellular respiration is one of the key chemical processes that produces energy within cells. It involves a series of metabolic pathways that break down glucose molecules into carbon dioxide and water, releasing energy in the form of ATP. This process occurs in the cytoplasm and mitochondria of eukaryotic cells and the cytoplasm of prokaryotic cells.
The overall chemical equation for cellular respiration is:
C6H12O6 + 6 O2 → 6 CO2 + 6 H2O + Energy (ATP)
There are four key stages in cellular respiration:
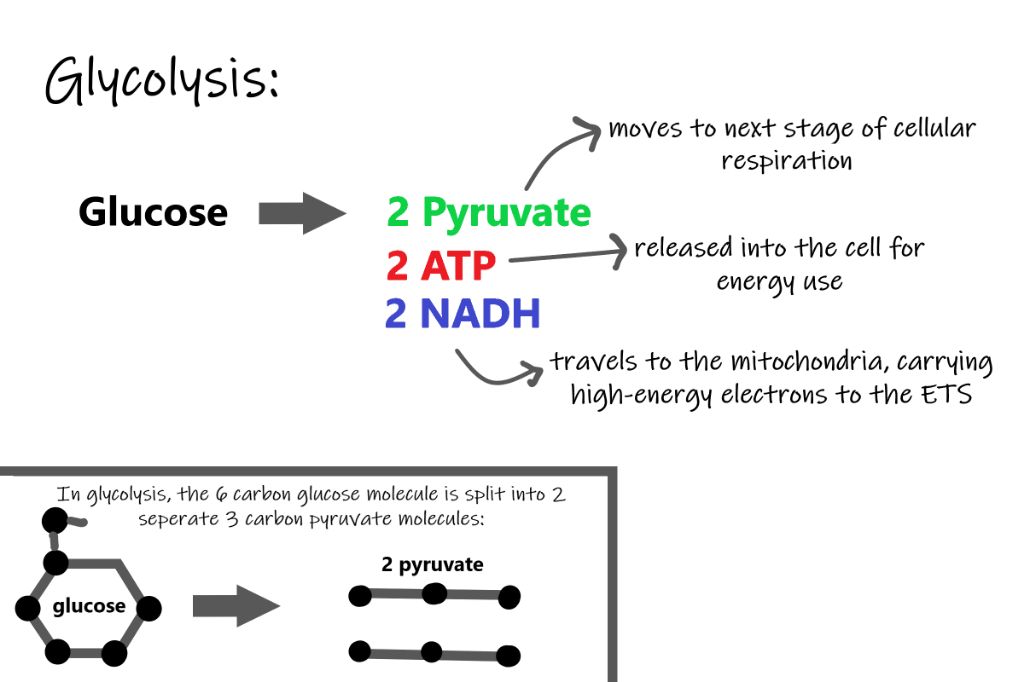
- Glycolysis – Glucose is broken down into pyruvate in the cell’s cytoplasm, producing a small amount of ATP and NADH.
- Pyruvate Oxidation – Pyruvate is transported into the mitochondria and converted into acetyl CoA while producing NADH.
- Citric Acid Cycle – Acetyl CoA enters the citric acid cycle and is further oxidized, producing more ATP, NADH, and FADH2.
- Oxidative Phosphorylation – The electron transport chain uses the NADH and FADH2 to create a proton gradient, which drives ATP synthase to produce the majority of ATP.
In summary, cellular respiration oxidizes glucose and harvests the energy in its bonds to generate ATP molecules, which provide usable chemical energy for cells. This efficient process generates up to 38 ATP molecules per glucose molecule.
Fermentation
Fermentation is a process that converts sugar into acids, gases or alcohol. It occurs naturally in some bacteria and yeasts. There are two main types of fermentation: alcoholic fermentation and lactic acid fermentation.
In alcoholic fermentation, yeasts break down sugars, creating ethanol and carbon dioxide as byproducts. For example, brewing beer involves yeast fermenting sugars derived from grains like barley. Wine production also relies on yeast fermenting the sugars in grapes.
In lactic acid fermentation, bacteria like lactobacilli convert sugars into lactic acid. This type of fermentation is used in the production of foods like yogurt, cheese and sourdough bread. The lactic acid gives these foods their sour taste. Lactic acid fermentation also occurs in animal muscles during intense exercise, causing muscle stiffness and fatigue.
Both alcoholic and lactic acid fermentation produce chemical energy in the form of ATP. They allow the breakdown of sugars to occur in the absence of oxygen. Fermentation is critical for producing foods, beverages and even energy within certain cells.
Combustion
Combustion is a high-temperature exothermic chemical reaction between a fuel and an oxidant, such as oxygen gas. The combination of these substances results in the production of heat, light, and various reaction products. Some common examples of combustion reactions include:
- Burning of wood, coal, natural gas, or other organic fuels
- Internal combustion in engines, such as in cars and trucks
- Burning of candles, oil lamps, or gas stoves
- Explosions involving gunpowder, dynamite, or other explosives
- Rocket propulsion
- Forest fires
In each case, the fuel source reacts rapidly with oxygen, generating significant amounts of heat energy. This exothermic process converts the chemical potential energy stored in the molecular bonds of the fuel into thermal energy and light. The reaction takes place so quickly that it is often accompanied by a flame. Combustion requires an ignition temperature to get started and an adequate supply of oxygen to sustain the reaction. It is a common method of producing energy for human needs.
Hydrogen Fuel Cells
Hydrogen fuel cells are electrochemical devices that combine hydrogen and oxygen to produce electricity, water, and heat. Hydrogen acts as an energy carrier, storing the chemical energy extracted from other substances. This allows hydrogen to be produced from any energy source and allows the chemical energy to be converted to electrical energy through a fuel cell.
The main components of a hydrogen fuel cell are the anode, cathode, electrolyte, and catalyst. Hydrogen is fed into the anode side of the fuel cell. At the anode site, the hydrogen molecules are split into protons and electrons through a process called oxidation. The protons pass through the electrolyte membrane to the cathode side, while the electrons travel in an external circuit, producing an electric current. Oxygen is fed into the cathode side, where it reacts with the electrons and protons to produce water. The only byproduct of this reaction is water, so hydrogen fuel cells are emissions-free.
Different electrolyte materials can be used in hydrogen fuel cells, each with their own advantages and disadvantages. Polymer electrolyte membrane (PEM) fuel cells operate at relatively low temperatures, are lightweight, and have high power density. Solid oxide fuel cells (SOFCs) operate at very high temperatures, so they can utilize waste heat and are not as sensitive to fuel impurities. Alkaline fuel cells (AFCs) use alkaline electrolytes and can provide high efficiency and power density, but are sensitive to carbon dioxide. Overall, hydrogen fuel cells provide a clean and efficient way to convert the chemical energy in hydrogen into usable electrical energy.
Batteries
Batteries produce chemical energy through electrochemical reactions. Within a battery, chemical energy is converted to electrical energy as electrons move from the negative electrode, called the anode, to the positive electrode, called the cathode.
In a typical battery, the anode is made of a metal like zinc or lithium. The anode undergoes a chemical oxidation reaction and loses electrons. The freed electrons flow through an external circuit to generate electricity. Meanwhile, the cathode is made of a material that can accept electrons, like manganese dioxide. The cathode undergoes a chemical reduction reaction as it gains electrons coming from the anode through the circuit.
In order for the electrochemical reaction to continue, the electrons must be able to flow from the anode to the cathode. This flow of electrons produces an electric current that can be used to power electrical devices. The movement of ions through the electrolyte also balances the flow of electrons to maintain overall charge neutrality within the battery.
Different battery chemistries involve different anode and cathode materials that determine the voltage and capacity of the battery. But in all cases, chemical energy is converted to electrical energy through spontaneous oxidation-reduction reactions.
Digestion
Digestion is a process that allows animals to chemically break down their food and extract energy from it. This involves a complex set of chemical reactions that begin in the mouth and stomach, and are completed further down in the small intestine and colon.
The digestion process starts when enzymes in saliva begin to break down carbohydrates in the mouth. When food is swallowed, it enters the stomach, where gastric juices containing hydrochloric acid and digestive enzymes continue breaking it down further. The partially digested food then moves into the small intestine, where bile from the liver and digestive enzymes from the pancreas help to further break down fats, proteins and carbohydrates.
As food molecules are broken down into their individual components, they can be absorbed through the intestinal walls and transported to cells throughout the body. The cells use these simple components to fuel their metabolic processes and produce energy in the form of ATP via cellular respiration. This ATP powers all other cellular functions.
Overall, the complex digestive process allows animals to unlock the chemical energy stored in food and convert it into a form they can use. Without this important ability to extract energy from external sources, animals would not be able to power all the biochemical reactions necessary for life.
Chemosynthesis
Chemosynthesis is a process where organisms obtain their energy from certain chemical reactions rather than through photosynthesis or by digesting other organisms. Many of these organisms live in extreme environments such as hydrothermal vents deep in the ocean where sunlight cannot reach. The organisms that utilize chemosynthesis are bacteria and archaea that use chemical reactions like oxidation of inorganic molecules such as hydrogen sulfide or methane to produce energy in the form of adenosine triphosphate (ATP). The bacteria and archaea are able to convert carbon dioxide into organic compounds like carbohydrates through chemosynthesis, providing themselves a source of nutrition. Overall, chemosynthesis allows certain organisms to thrive in harsh environments through a simple yet effective internal process.
Conclusion
Chemical energy plays an indispensable role in sustaining life and fueling society. The various processes that produce chemical energy, such as photosynthesis, cellular respiration, digestion, combustion, and more demonstrate the diversity of pathways that convert other forms of energy into chemically stored energy. This chemical energy is essential for cells and organisms to carry out their vital functions and activities. It powers everything from molecular processes to mechanical motion. Further research into improving the efficiency and sustainability of chemical energy production will be crucial as society works to meet rising energy needs while protecting the planet. Understanding the science behind transforming, transferring, and harnessing chemical energy will pave the way for innovations and solutions to global energy challenges.