What Is Work And Energy Short Note?
Work and energy are two fundamental concepts in physics that are closely related. Work involves applying a force to move an object, while energy is the ability to do work. Understanding work and energy is key to explaining how the physical world operates.
Work and energy concepts help describe interactions between objects and allow us to analyze complex mechanical systems. We can use work and energy principles to solve real-world problems, build machines and technology, and gain insights into the behavior of matter and motion.
Studying work and energy provides a foundation for other important physics topics like power, efficiency, momentum, thermodynamics, and more. Mastering these foundational ideas equips students with analytical skills applicable across STEM fields.
In short, work and energy are essential physics concepts with broad relevance. Examining these principles reveals deep insights into how our universe functions at the most fundamental level.
Work
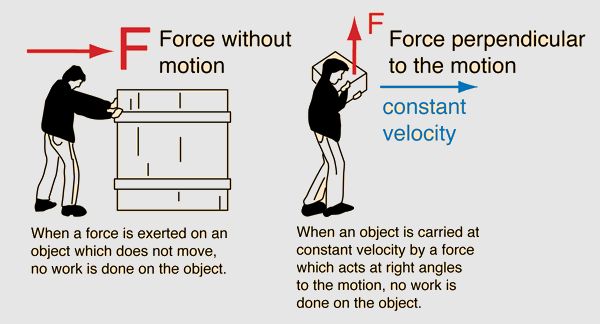
Work is defined as the energy transferred to or from an object via the application of a force. Work is done when an object is moved over a distance by an applied force. The mathematical formula for work is:
W = F x d
Where:
W = Work done (Joules)
F = Force applied (Newtons)
d = Distance moved (Meters)
The SI unit for work is the Joule (J), which is equal to 1 Newton-meter. Work is a scalar quantity, meaning it has magnitude but no direction.
Some examples of work include:
– Lifting a box from the ground. The force of your arms does work to move the box upwards.
– Pushing a cart across a floor. The force propelling the cart does work to move it.
– Stretching a spring. The force applied stretches the spring, doing work to increase its potential energy.
Energy
Energy is the capacity to do work. It exists in various forms that can be converted from one form to another. The main forms of energy include:
- Kinetic energy – the energy possessed by a body by virtue of its motion. For example, the kinetic energy of a moving car.
- Potential energy – stored energy possessed by a body by virtue of its position or state. For example, the potential energy stored in a compressed spring.
- Chemical energy – energy stored in the bonds of atoms and molecules. The energy released during chemical reactions like combustion.
- Electrical energy – energy due to electric charges. The energy that powers electrical devices.
- Nuclear energy – energy stored within atomic nuclei, released during nuclear reactions.
- Thermal energy – the internal energy present in substances due to molecular motion. Heat is a form of thermal energy.
- Radiant energy – energy propagated by electromagnetic waves. Light is a form of radiant energy.
Energy can be converted from one form to another, but it cannot be created or destroyed according to the law of conservation of energy. Examples of energy conversion include a battery converting chemical energy to electrical energy, or a light bulb converting electrical energy to radiant energy. The different forms of energy can be used to perform useful work.
Kinetic Energy
Kinetic energy is the energy an object possesses due to its motion. It is defined as the work needed to accelerate a body of a given mass from rest to its stated velocity. Having gained this energy during its acceleration, the body maintains this kinetic energy unless its speed changes. The same amount of work is done by the body when decelerating from its current speed to a state of rest.
In classical mechanics, the kinetic energy of a non-rotating object of mass m traveling at a speed v is 1⁄2 mv2. In relativistic mechanics, this is only a good approximation when v is much less than the speed of light.
Kinetic energy is directly proportional to the mass of the object and to the square of its velocity:
KE = 1⁄2 mv2. The velocity in this equation is the true velocity before the reference frame transformation.
This formula shows that the greater the mass of an object, the greater its kinetic energy at a given velocity. It also shows that the greater the velocity of an object, the greater its kinetic energy.
When an object is pushed or pulled through a distance by an applied force, it is moved by a certain amount of energy. The work done on the object by the force gives it kinetic energy equal to the work required to achieve its final state of movement.
Potential Energy
Potential energy is stored energy an object has because of its position or state. There are several types of potential energy:
- Gravitational potential energy – The energy an object has because of its height relative to the ground. The gravitational potential energy is equal to the object’s mass multiplied by the acceleration due to gravity and its height:
GPE = mgh
Where m is mass, g is gravitational acceleration, and h is height.
- Elastic potential energy – The energy stored in elastic materials that are stretched or compressed. It is equal to the force exerted multiplied by the distance the material is deformed:
EPE = 1/2kx2
Where k is the spring constant and x is the displacement distance.
- Chemical potential energy – Energy stored in the bonds between atoms and molecules. It is released or absorbed during a chemical reaction.
- Nuclear potential energy – Energy stored in the bonds between nucleons (protons and neutrons) in an atomic nucleus. It is released in nuclear fission or fusion reactions.
- Electric potential energy – Energy stored between two electrically charged particles or systems of charges. It depends on the electric potential and amount of charge.
The key thing about potential energy is that it is not motion, but stored energy ready to be released to do work. Understanding the different forms of potential energy is crucial in physics.
Work-Energy Theorem
The work-energy theorem states that the net work done on an object is equal to its change in kinetic energy. This theorem establishes the equivalence between work (W) and kinetic energy (K).
The work-energy theorem can be expressed mathematically as:
W = ∆K
Where:
- W is the net work done on the object
- ∆K is the change in kinetic energy of the object
This theorem shows that the net work done on an object by external forces changes its kinetic energy. Any net work adds to the object’s kinetic energy. If positive work is done, the kinetic energy increases. If negative work is done (by opposing forces), the kinetic energy decreases.
In summary, the work-energy theorem states that the net work on an object equals its change in kinetic energy. Work and kinetic energy are equivalent quantities connected through this important relationship.
Power
Power is defined as the rate at which work is done or energy is transferred. The mathematical formula for power is:
Power = Work / Time
Where:
- Power is measured in watts (W)
- Work is measured in joules (J)
- Time is measured in seconds (s)
Power represents the rate at which work is performed or energy is converted. For example, a lightbulb that uses 100 joules of energy over 10 seconds is converting energy at a power of 10 watts. The more work a machine can do in a shorter amount of time, the more powerful it is.
Power is an important concept because it allows us to understand how quickly work can be done or energy can be transferred. Power levels indicate how much useful work a device can perform and are a key specification for motors, engines, and other machines.
Energy Conservation
The principle of conservation of energy states that energy can neither be created nor destroyed, it can only be transformed from one form to another. This means that the total amount of energy in an isolated system always remains constant.
For example, when a ball falls, its potential energy gets transformed into kinetic energy. The initial potential energy of the ball gets converted into its kinetic energy as it falls. However, the total amount of energy before and after the fall remains the same. There is no creation or destruction of energy in this process.
This principle applies to all isolated systems where no energy crosses the boundaries. In real world systems that are not perfectly isolated, some energy may enter or escape, but the total energy change is equal to the difference between energy inputs and outputs.
The conservation of energy is an important concept in physics that can be applied to analyze mechanical, thermal, chemical, nuclear and other processes. It is a fundamental law of nature that holds true across all fields of science.
Real World Applications
The concepts of work, energy, and power are crucial in many real world technologies and systems. Here are some examples of how these ideas are applied:
Vehicles: The engine in a car or truck does work on the wheels to accelerate the vehicle. The energy produced by burning fuel is converted into kinetic energy that propels the vehicle forward. Power measures how quickly a vehicle’s engine can do this work.
Batteries: Batteries store chemical potential energy and convert it into electrical energy that can power devices. The bigger the battery, the more energy it can hold to allow longer use of devices before recharging.
Hydropower: Dams convert the potential energy of water high up in a reservoir into kinetic energy as it flows down through turbines, which convert that motion into electricity through generators.
Energy Efficiency: Engineers analyze where energy is being used and wasted in systems like buildings and factories. Applying concepts of work and power allows them to find ways to reduce energy consumption.
Nutrition: Food provides chemical potential energy for our bodies. The number of Calories measures how much energy various foods contain to power our metabolism.
Summary
Work and energy are two fundamental concepts in physics that are closely related. Work refers to the process of transferring energy to an object via the application of a force over a distance. Energy comes in multiple forms, but the two most relevant to work are kinetic energy, which is the energy of motion, and potential energy, which is stored energy due to an object’s position or structure.
The work-energy theorem states that the net work done on an object is equal to its change in kinetic energy. This shows that work done on an object transfers energy to that object. Power is the rate at which work is done or energy is transferred. Energy can never be created or destroyed, only converted from one form to another, which is the law of conservation of energy.
Understanding work and energy allows us to analyze physical systems and processes, from the motion of objects to the operation of machines. Key takeaways include the relationship between work and kinetic energy, the different forms of energy, work-energy theorem, power as the rate of doing work, and the principle of energy conservation in closed systems.