What Is Thermal Radiation Transferred By?
Definition of Thermal Radiation
Thermal radiation is electromagnetic radiation generated by the thermal motion of particles in matter. All matter with a temperature above absolute zero emits thermal radiation. Examples of thermal radiation include the infrared radiation emitted by hot objects, the heat you feel from a fire, and the glow from red-hot and white-hot objects.
Thermal radiation is part of the electromagnetic spectrum, which includes radio waves, microwaves, infrared, visible light, ultraviolet, X-rays, and gamma rays. The key difference between thermal radiation and other types of electromagnetic radiation is that thermal radiation is emitted by matter because of its temperature, rather than emissions caused by atomic transitions. Thermal radiation has longer wavelengths than ultraviolet, X-rays, and gamma rays.
While all objects emit thermal radiation, the wavelength and intensity of the radiation depends on the object’s temperature. Objects at room temperature emit low-intensity infrared radiation, while extremely hot objects like the sun emit higher intensity radiation across the visible spectrum as well.
How Thermal Radiation Works
Thermal radiation is electromagnetic radiation that is emitted from the surface of objects due to their internal temperature. All objects with a temperature above absolute zero emit thermal radiation. The amount and wavelength of the radiation depends on the temperature of the object.
The hotter an object is, the more thermal radiation it emits per unit surface area. This is known as the Stefan-Boltzmann law. For example, a red-hot stove element emits more thermal radiation than a cool sidewalk.
Hotter objects also emit radiation at shorter wavelengths than cooler objects. For example, a red-hot element may glow visibly red, while a slightly cooler one may emit invisible infrared radiation. This is known as Wien’s displacement law. The peak wavelength of emission shifts to shorter wavelengths as temperature increases.
According to Interesting Engineering, the thermal radiation emitted by objects is the mechanism by which heat travels directly between objects, unlike conduction or convection which require a medium. The emitted electromagnetic waves carry energy away from the hot object towards cooler surroundings.
Wavelengths of Thermal Radiation
The wavelengths associated with thermal radiation span from 0.1 to 100 μm, though the primary wavelengths are in the infrared region of the electromagnetic spectrum (Chegg). Infrared radiation refers to electromagnetic radiation with wavelengths longer than visible light but shorter than microwaves. For objects around room temperature, the peak of thermal radiation emitted is around 10 μm (Biology Forums).
Different temperature ranges are associated with different wavelengths of thermal radiation. For example, the sun has a surface temperature of nearly 6,000°C and emits most strongly at a wavelength of 0.5 μm. The human body emits thermal radiation peaked around 9.5 μm at a temperature of 37°C (ResearchGate). In general, hotter objects emit radiation at shorter wavelengths than cooler objects, following Wien’s displacement law.
Examples of Thermal Radiation
Thermal radiation is emitted by all objects above absolute zero temperature. Here are some common examples of thermal radiation we encounter in everyday life:
The Sun emits thermal radiation over a wide spectrum of wavelengths, including visible light, infrared, and ultraviolet. The Sun’s thermal radiation is the primary source of energy that heats the Earth and makes life possible.
The Earth itself emits thermal radiation as infrared light, which is known as terrestrial or Earth radiation. The Earth radiates heat into space because it is warmer than absolute zero.
Any object that is heated up, such as an oven burner or stove top, emits thermal radiation. You can feel this radiant heat on your skin when you stand near a hot object.
Fires emit thermal radiation, which allows the heat to be felt from a distance. The hot embers and flames of a campfire send out thermal radiation that warms people sitting around it.
Incandescent light bulbs work by heating up a tungsten filament inside the bulb, which makes the filament emit visible light. This is an example of thermal radiation in the visible spectrum.
The human body emits thermal radiation in the infrared spectrum, which is how infrared cameras are able to detect people in the dark.
Applications of Thermal Radiation
Thermal radiation has many practical applications in everyday life and science.
One major application is in night vision and thermal imaging. Objects emit infrared radiation that is invisible to the human eye. Special cameras can detect this radiation to see in dark conditions or to measure temperature variations. Night vision goggles for military use rely on detecting thermal radiation. Thermal imaging cameras are used for tasks like building inspections, locating heat loss, and identifying warm-blooded animals at night.
Thermal radiation is also widely used for cooking and heating. The radiation emitted by hot objects like stovetops, toasters, and space heaters is absorbed by cooler objects, heating them up. Microwave ovens specifically utilize microwave radiation to deliver energy used for cooking food.
Climate science relies on measuring thermal radiation emitted and absorbed by the Earth system. Satellites monitor outgoing radiation from Earth to determine the global energy budget and track climate change. The greenhouse effect is caused by gases that absorb infrared radiation, thereby affecting Earth’s surface temperatures.
Overall, thermal radiation underlies many essential technologies and scientific processes in fields like engineering, medicine, astronomy, and meteorology. Understanding and controlling thermal radiation unlocks new capabilities across these disciplines.
Measuring Thermal Radiation
Thermal radiation is invisible to the human eye, so specialized instruments are needed to detect and measure it. Two common devices used are infrared cameras and thermometers. Infrared cameras can visualize infrared radiation and create images based on temperature differences. The cameras detect infrared wavelengths and convert them into visible light that shows up as varying colors or shades representing hot and cold areas. This allows one to see thermal patterns and heat flows that are otherwise invisible. Infrared thermometers are a simpler device that measures temperature from a distance using emitted infrared radiation, displaying just a temperature reading. They are pointed at a surface to gauge its temperature.
Key quantities measured in thermal radiation include radiant exitance and irradiance. Radiant exitance (M) is the total power per unit area emitted from a surface via radiation. It indicates how much thermal energy radiates off a surface over time. Irradiance (E) measures the power of electromagnetic radiation per unit area received by a surface. It quantifies the incident radiant energy on a surface. Radiant exitance and irradiance are measured using specialized radiometer instruments designed to capture thermal radiation in infrared wavelengths.
Some pioneers in developing devices to measure thermal radiation include John Leslie, who created an early differential thermometer in the 1800s, and William Herschel who used prisms and thermometers to examine infrared radiation in sunlight. Modern infrared cameras and thermometers continue to improve, with greater temperature precision, resolution, and ability to visualize detailed thermal patterns enabling new applications.
Controlling Thermal Radiation
Thermal radiation can be controlled through various methods to either reduce or increase its transfer. Some common techniques involve using insulation, reflective coatings, and materials with low emissivity.
Insulation works by trapping air pockets to slow down heat transfer. Common insulating materials include fiberglass, plastic foams, wool, and ceramics. Proper insulationinstallation in buildings can help reduce heat loss in winter and heat gain in summer. Spacecraft and satellites also use insulation to maintain optimal internal temperatures (Ribbing, 2013).
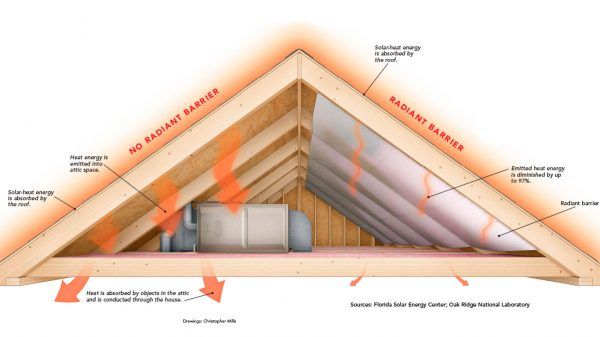
Reflective coatings are applied to surfaces to reflect thermal radiation rather than absorb it. Polished metals like aluminum are commonly used as radiation reflectors. Special paints containing aluminum flakes or other reflective particles can alsoreduce absorption and reradiate heat away from a surface. Reflective barriers are often added to attics to minimize solar heating (Ribbing, 2013).
Emissivity refers to an object’s effectiveness in emitting thermal radiation. Materials with low emissivity tend to be poor absorbers and good reflectors of radiant heat. For instance, shiny metals have lower emissivity than rough, dark surfaces. Advanced coatings with emissivity control are being developed to optimize heat transfer in applications like solar collectors and high-temperature materials (Phys.org, 2024).
Health Effects
Thermal radiation exposure can have various health effects on the human body. High levels of thermal radiation, especially in the infrared wavelengths, can cause burns to the skin and eyes (cite). Infrared radiation is absorbed strongly by water in human tissue, which converts the radiation into heat that can damage cells. Even short-term exposure to high-intensity infrared sources can cause first-degree or second-degree burns.
Infrared radiation is invisible to the human eye, so damage can occur before it is noticed. This radiation can penetrate and damage the cornea and lens of the eye, potentially leading to conditions like cataracts. Protective eyewear should be worn when working with infrared emitters.
However, infrared radiation also has some health benefits when used properly. It can help increase blood circulation and relieve muscle and joint pain. Many studies have found that infrared saunas may provide cardiovascular benefits. But overexposure carries risks like dehydration and overheating. Infrared radiation sources should be used cautiously (cite).
Thermal Radiation in Earth’s Energy Balance
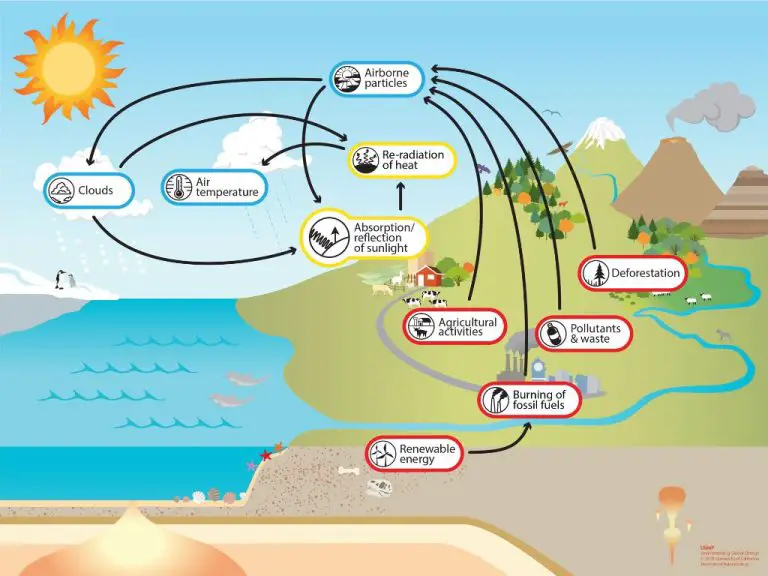
The Earth’s energy budget describes the balance between incoming and outgoing radiation on Earth. Of the approximately 340 watts per square meter (W/m2) of solar radiation received by Earth, about 77 W/m2 is reflected back to space by clouds and the atmosphere while 23 W/m2 is absorbed by the atmosphere (NASA, https://earthobservatory.nasa.gov/features/EnergyBalance). The remaining solar radiation is absorbed by the land and oceans. The Earth itself emits outgoing longwave thermal infrared radiation with an average power of around 240 W/m2 according to atmospheric temperature and emissivity (Wikipedia, https://en.wikipedia.org/wiki/Earth%27s_energy_budget).
Greenhouse gases in the atmosphere, especially carbon dioxide, absorb some of the outgoing thermal infrared radiation and re-radiate it back towards the surface. This results in a higher surface temperature than if there were no greenhouse gases. The increased absorption of infrared radiation by greenhouse gases due to human activities, especially the burning of fossil fuels, has enhanced the natural greenhouse effect and contributed significantly to global warming and climate change.
History and Pioneers
The study of thermal radiation has a long history going back to the 17th century when scientists first began to investigate heat and temperature. Some key developments include:
In 1600, the Italian scientist Giordano Bruno proposed that heat and light both consisted of tiny particles. This was an early conception of thermal radiation.
In 1701, the English physicist Sir Isaac Newton published his treatise on light and colors, Opticks, in which he theorized that heat and light were related phenomena. This laid important groundwork for the understanding of thermal radiation.
In 1738, the Swiss physicist Daniel Bernoulli conducted pioneering experiments on radiant heat, using concave mirrors to concentrate sunlight and measure its heating effect. This demonstrated that radiant heat followed the laws of optics.
In 1760, the Welsh scientist Joseph Priestley focused sunlight with a lens, discovering that different colors (wavelengths) of light had different heating abilities. He showed red light had the least heating effect.
In the 1770s, the Swedish chemist Carl Wilhelm Scheele made key distinctions between heat transfer by thermal radiation, conduction, and convection. This advanced the specific study of radiant heat.
In the 1800s, major experiments by John Leslie, John Herschel, Macedonio Melloni, and others furthered the understanding of radiant heat absorption, reflection, and transmission through different media.
In 1879, the Austrian physicist Josef Stefan derived what is now known as the Stefan-Boltzmann law, quantitatively relating thermal radiation to temperature. This was a major milestone.
In the late 19th and early 20th centuries, scientists like Wilhelm Wien, Max Planck, and others helped fully quantify the properties of thermal radiation across the electromagnetic spectrum.