What Is The Unit For Measuring Energy?
Energy is a fundamental concept in physics that describes the capacity to do work and cause change. Energy comes in many different forms such as electrical, thermal, nuclear, gravitational, chemical, kinetic, potential, and more. To quantify, compare and measure different forms of energy, a standard unit of measurement is needed. This article will discuss the history and applications of the joule, which is the standard international unit used for measuring energy.
Defining Energy
Energy is the ability to do work or produce heat. It is a quantitative property that can be transferred between objects or converted into different forms. Energy comes in many varieties, including kinetic energy, potential energy, electrical energy, thermal energy, chemical energy, radiant energy, and nuclear energy. For example, a moving car has kinetic energy, a battery stores electrical energy, food contains chemical energy for our bodies, and the sun radiates electromagnetic energy.
When energy is transferred between objects or changes form, it is still conserved according to the law of conservation of energy. The total energy in a closed system remains constant. This means one form of energy can be converted into another, but energy cannot be created from nothing or destroyed. For instance, a light bulb converts electrical energy into radiant energy in the form of light and heat. The total energy before and after remains the same.
History of the Joule
The joule unit was named in honor of James Prescott Joule, a British physicist who lived from 1818 to 1889. Joule made important contributions to the fields of thermodynamics and electromagnetism. His most notable achievement was determining the mechanical equivalent of heat – the amount of work required to produce a certain amount of heat.
In the 1840s, Joule conducted experiments involving mechanical work and the transfer of energy as heat. He demonstrated that the energy to produce a certain amount of heat was constant, regardless of the process or materials involved. This became known as the mechanical equivalent of heat. Joule’s work showed that heat and mechanical work were two forms of the same type of energy, rather than separate substances as previously thought.
To recognize Joule’s contributions, the name “joule” was officially adopted as the unit of energy in the International System of Units (SI) in 1960. The joule represented the international standard base unit of measurement for all forms of energy. Joule’s research formed the basis of the first law of thermodynamics and helped establish the principle of conservation of energy.
What is a Joule?
The joule is the International System of Units (SI) derived unit of energy. It is defined as the work done, or energy transferred, when an applied force of one newton moves an object one meter in the direction of the force.
This definition was established at the first General Conference on Weights and Measures in 1889 and was named after the English physicist James Prescott Joule. The joule measures the amount of energy that is expended when a force of one newton is applied over a distance of one meter.
For example, when you lift a book from the floor onto a table, you are moving the book through a vertical distance while gravity acts on it with a downward force equal to the book’s weight. The amount of energy transferred during this process, from the chemical energy in your muscles to the potential energy of the book on the table, is measured in joules.
The joule is used to quantify a wide range of forms of energy, including thermal, mechanical, electrical and chemical energy. Having a standard unit of measurement for energy allows precise comparisons between different energy quantities and conversions between different energy types.
Measuring Energy in Joules
Energy is measured in joules, with the joule being the standard SI unit for all forms of energy. Some examples of measuring energy amounts in joules:
- A 100 watt lightbulb uses 100 joules of electrical energy per second. Over the course of an hour, it would use 100 x 3600 = 360,000 joules.
- A 200 calorie food item contains 200 calories x 4.184 joules/calorie = 836.8 joules of chemical energy that can be released through digestion.
- A moving car with a mass of 1500 kg moving at 50 km/hr (13.9 m/s) has a kinetic energy of 0.5 x 1500 x 13.9^2 = 1,361,425 joules.
- A 75 kg person climbing a flight of stairs 10 meters high gains potential energy of 75 x 9.8 x 10 = 7,350 joules.
As these examples show, joules are used to measure the total energy content or expenditure across mechanical, electrical, chemical, nuclear and thermal energy forms.
Converting Between Energy Units
While the joule is the standard SI unit for measuring energy, there are several other common energy units used around the world. It’s important to understand how to convert between joules and these other units:
Calories
The calorie (cal) is a unit commonly used to measure energy in food. 1 calorie is equal to 4.184 joules.
To convert calories to joules, multiply the calories by 4.184. For example, 200 calories is equal to 200 x 4.184 = 836.8 joules.
To convert joules to calories, divide the joules by 4.184. For example, 836.8 joules is equal to 836.8 / 4.184 = 200 calories.
Kilowatt-hours
The kilowatt-hour (kWh) is a unit commonly used to measure electrical energy consumption. 1 kilowatt-hour is equal to 3,600,000 joules.
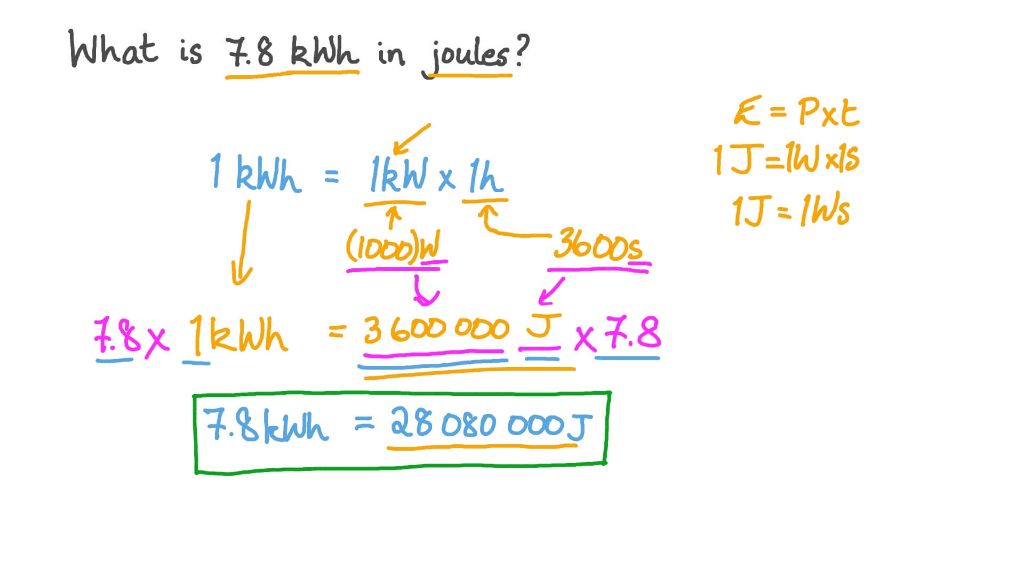
To convert kilowatt-hours to joules, multiply the kWh by 3,600,000. For example, 1 kWh is equal to 1 x 3,600,000 = 3,600,000 joules.
To convert joules to kWh, divide the joules by 3,600,000. For example, 3,600,000 joules is equal to 3,600,000 / 3,600,000 = 1 kWh.
Knowing how to convert between energy units allows you to evaluate and compare energy usage across different contexts. With practice, these unit conversions can become second nature.
Applications of the Joule
The joule is used to quantify energy in many scientific and engineering applications:
-
Physics – The joule is the standard unit for work, energy, and quantity of heat in the International System of Units (SI). It is used in calculations involving thermodynamics, electromagnetism, mechanics, and optics.
-
Engineering – Joules are used by electrical, mechanical, civil, and chemical engineers to measure energy usage, convert between energy types, and analyze processes. For example, a joule can describe the energy required to lift an object or the kinetic energy of a moving vehicle.
-
Nutrition – Food labels use joules along with calories to quantify the metabolizable energy content of foods and beverages. This helps people monitor their caloric intake.
-
Medicine – In respirometry tests, a joule measures the amount of oxygen used by organisms during cellular respiration. This helps determine metabolic rates.
-
Chemistry – Joules describe the energy released or absorbed during chemical reactions. Exothermic reactions like combustion release energy, while endothermic reactions absorb energy.
-
Ecology – Ecologists may measure the productivity of ecosystems in joules to quantify energy flows between trophic levels.
In summary, the joule unit enables the measurement of energy across many scientific, engineering, medical, and practical applications.
Importance of Standardization
Having an internationally recognized and standardized unit for measuring energy provides substantial benefits for science, technology, and industry. Prior to the joule, there was no unified system for quantifying energy. Scientists and engineers in different countries used disparate units like the calorie, foot-pound, and kilogram-meter. This made it extremely difficult to communicate and compare energy values across national borders.
The formal adoption of the joule by international scientific bodies in the late 19th century finally provided a single, well-defined unit. Now scientists around the world can seamlessly share research and calculations using the same energy measurements. Standardization enables clear collaboration in physics, engineering, chemistry, and other technical fields.
In industry, universal energy units facilitate international trade and commerce. Companies can buy and sell products across global markets using consistent specifications and testing methodologies with joules. Standardization also aids regulation, safety standards, and quality control across national boundaries. Consumers benefit from standardized energy ratings for appliances, vehicles, and devices.
Overall, the joule provides a critical foundation for technological advancement worldwide. By agreeing on a shared definition of energy, the international community has made great strides in scientific discovery, industrial development, and consumer awareness.
Fun Facts About the Joule
The joule unit of energy has some interesting facts and trivia associated with it:
-
The joule is named after English physicist James Prescott Joule. Joule determined the mechanical equivalent of heat through experiments such as stirring water to measure heat increases.
-
One joule is defined as the amount of work required to produce one watt of power for one second. A watt is defined as one joule per second.
-
A typical apple contains about 520 joules of energy. An average banana contains about 370 joules.
-
A single lightning bolt contains roughly 1 billion joules of energy. That’s enough to power a 100-watt light bulb for 3 months!
-
It takes approximately 4.2 joules to raise the temperature of 1 gram of water by 1°Celsius.
-
The average daily energy requirement for an adult human is about 8400 kilojoules.
-
One kilowatt-hour (kWh) equals 3600 kilojoules of electrical energy. Most electricity usage is measured and billed in kilowatt-hours.
These fun facts help provide some tangible examples of just how much energy is represented by a joule. It’s an important standard unit that allows us to universally measure and compare energy usage across many contexts.
Conclusion
The joule is an important base unit of measurement in the International System of Units for quantifying energy. As we’ve explored, one joule is defined as the amount of work done by a force of one newton moving an object one meter. While James Prescott Joule did not invent the joule, his pioneering work in the 1840s on the mechanical equivalent of heat helped establish the joule as the standard international unit of energy. Using the joule allows us to uniformly measure, compare and convert between different forms of energy including electrical, thermal, solar and mechanical energy.
The applications of the joule are far-reaching, from calculating the potential energy in food to determining the power usage of electrical appliances. Having a standardized unit for energy facilitates scientific study and commerce worldwide. While people may be more familiar with kilojoules or calories on nutrition labels, the joule remains the base SI unit. Understanding what a joule represents and how to convert between energy units is key to quantifying energy transfers in our universe.
In summary, the joule as a derived unit for energy enables precise measurement and comparison of energy across disciplines and contexts. Joule’s empirical experiments provided the foundation for the joule as the international standardized unit of energy.