What Is The Relation Between Potential And Energy?
Potential and energy are related concepts in physics. Potential refers to stored or latent energy that has the possibility of becoming active. Energy is the capacity to do work and overcome resistance. While energy is needed to perform work, potential represents the maximum amount of energy available to perform work.
There are various forms of potential energy, like chemical, nuclear, gravitational, elastic, and electrical. These represent energy stored in a system by virtue of an object’s position or arrangement. This stored energy can be transformed into kinetic energy, which is the energy associated with motion.
The relationship between potential and energy is that potential energy can be harnessed and turned into usable kinetic energy. Potential energy becomes kinetic energy when the objects are released to move. Understanding the connection between potential and kinetic energy is key to explaining many physical phenomena.
Potential Energy
Potential energy is the stored energy an object has due to its position or chemical composition. There are several types of potential energy:
Gravitational Potential Energy
Gravitational potential energy is the energy an object possesses due to its height above the ground. For example, a book sitting on a high shelf has more gravitational potential energy than the same book sitting on the floor. As the book rises, its potential energy increases. When the book falls, this potential energy converts into kinetic energy.
Elastic Potential Energy
Elastic potential energy refers to the energy stored in elastic materials that are deformed. For example, when a rubber band is stretched, it gains elastic potential energy. When released, this energy converts into kinetic energy as the rubber band snaps back to its original shape. Springs also exhibit elastic potential energy.
Kinetic Energy
Kinetic energy is the energy of motion. It refers to the energy an object has due to its motion. The kinetic energy of an object depends on two variables: mass (m) and velocity (v). The kinetic energy (KE) can be calculated using the following formula:
KE = 1/2 * m * v^2
Where m is the mass of the object and v is its velocity. Some examples of kinetic energy in motion include:
- A moving car
- A kicked soccer ball
- Wind
- A flowing river
The faster the object moves, the more kinetic energy it possesses. Kinetic energy is directly proportional to the object’s mass and the square of its velocity. For instance, doubling the velocity of a moving object quadruples its kinetic energy.
Work Energy Theorem
The work energy theorem is one of the most important concepts in physics. It relates the net work done on an object to its change in kinetic energy. The theorem states that the net work done by all forces acting on an object equals its change in kinetic energy. In equation form:
Wnet = ΔK
Where Wnet is the net work done on the object and ΔK is the change in the object’s kinetic energy. Kinetic energy is energy associated with motion and is defined as:
K = 1/2mv2
Where m is mass and v is velocity.
The work-energy theorem is based on the principles of conservation of energy and the work-kinetic energy theorem. It shows that work done on an object transfers energy to the object, manifesting as kinetic energy. This transfers mechanical energy to the object, increasing its speed and kinetic energy.
Some examples where the work-energy theorem applies:
- Pushing a box across a floor – the work done to push the box increases its kinetic energy.
- Lifting an object upwards – work done against gravity increases the object’s potential energy.
- An externally powered machine or engine does work upon an object to increase its kinetic energy.
The work energy theorem is foundational in physics and engineering, enabling the analysis of mechanical systems and motion through the relationship between work and energy.
Conservation of Energy
The law of conservation of energy states that the total mechanical energy in a closed system remains constant. This means that energy can change forms within a system, but it cannot be created or destroyed. There are two main types of mechanical energy to consider – potential energy and kinetic energy.
Potential energy is the stored energy an object has due to its position or shape. For example, a ball held at a height above the ground has gravitational potential energy. As the ball falls, this potential energy gets converted into kinetic energy, the energy of motion. By the time the ball hits the ground, all of its potential energy has been converted into kinetic energy.
Kinetic energy is the energy of motion. The faster an object moves, the more kinetic energy it has. In the ball example, the kinetic energy increases as the ball picks up speed while falling. The gravitational potential energy gets transferred into kinetic energy.
So in a closed system, the total amount of mechanical energy remains the same. However, energy can shift between potential and kinetic. As an object gains potential energy, it loses kinetic energy. And as it loses potential energy, it gains kinetic energy. The conservation of mechanical energy is why things like pendulums and rollercoasters work.
Understanding the relationship between potential and kinetic energy helps explain how many mechanical systems function. It is a fundamental concept in physics that illustrates the principle of conservation of energy.
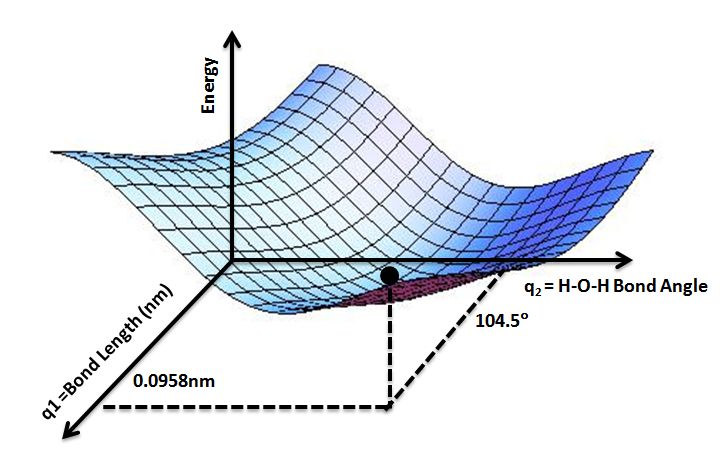
Potential Energy Curves
Potential energy curves, also known as potential energy diagrams or surfaces, are graphical representations showing how the potential energy of a system changes with the position of its components. They illustrate the energy landscape of a system and are very useful for visualizing molecular interactions and chemical reactions.
For a simple two-particle system like a diatomic molecule, the potential energy curve plots the potential energy as a function of the internuclear distance between the two atoms or nuclei. As the two atoms are brought closer together from an infinite separation, the potential energy decreases and reaches a minimum at the equilibrium bond length. This corresponds to the most stable configuration of the molecule. As the atoms are pushed closer, the potential energy increases sharply as the repulsive forces start to dominate, until the two nuclei finally fuse.
The shape of the potential energy curve provides a wealth of information about the forces between the particles and the stability of the system. The depth of the potential energy well indicates the bond strength, while the curvature reflects the stiffness or rigidity of the bond. A steeper curve means a stronger, shorter and more rigid bond. The positions and heights of local minima and maxima show the locations and energies of stable and unstable configurations.
For more complex molecular systems and chemical reactions, multi-dimensional potential energy surfaces can be plotted, with the axes representing different structural parameters like bond lengths, angles and dihedral angles. These hypersurfaces illustrate how the potential energy landscape changes along different reaction coordinates. The valleys and ridges on the surface determine the lowest energy reaction pathway from reactants to products.
In summary, potential energy curves and surfaces are indispensable for understanding the energetics and dynamics of chemical systems. They provide deep insights into molecular structure, stability, reactivity and more. Analyzing potential energy curves is critical for revealing transition states, intermediates, and activation barriers for chemical reactions.
Electric Potential Energy
Electric potential energy arises from electric fields and charged particles. An electric field surrounds any charged particle or collection of charged particles. The electric field contains energy in the form of voltage. When a charged particle moves through this field, its electric potential energy changes.
For example, consider two oppositely charged particles located near each other. They have an inherent electric potential energy due to their separated charges. If allowed to move closer together, the particles will accelerate towards each other, converting their electric potential energy into kinetic energy. Upon contact, the charges neutralize and the kinetic energy dissipates as heat.
This example demonstrates the relationship between electric potential energy, electric fields, particle charges, and motion. Electric potential represents stored energy available to do work on charged particles. The higher the electric potential (voltage) and amount of charge, the greater the potential energy. As charges accelerate or move in an electric field, their electric potential energy converts into kinetic energy or other forms.
Electric potential energy powers electrical devices and equipment. Batteries, for instance, separate charges to sustain an electric field. This field contains readily available energy to deliver power. Understanding electric potential energy provides key insights into electrical and electromagnetic phenomena.
Chemical Potential Energy
Chemical potential energy is the energy stored within the bonds of atoms and molecules. It is the energy that can be released or absorbed during a chemical reaction. When a chemical reaction occurs, the atomic bonds between reactants are broken and new bonds form between products – this requires an input or output of energy.
The amount of chemical potential energy stored in a substance depends on the types of atoms involved and how they are bonded. Some chemical bonds store more potential energy than others. For example, the bonds in gasoline molecules contain a lot of stored chemical energy that can be released through combustion. The energy released comes from breaking the bonds in the fuel molecules and forming new bonds in the product molecules (carbon dioxide and water). This release of energy is what powers car engines.
Food is another example of stored chemical energy. Food contains organic molecules like carbohydrates, proteins and fats. Digestion breaks these molecules down, releasing energy that cells can use to power biological processes. The amount of potential energy stored in food is commonly measured in Calories.
In summary, chemical potential energy exists in the bonds between atoms. Chemical reactions can release this energy when bonds are broken and new bonds form. The potential energy stored in substances like gasoline and food can be harnessed to do useful work.
Thermal Energy
Thermal energy, also known as heat energy, is the energy associated with the random motion of molecules within an object. As molecules vibrate or move faster, the object’s temperature increases. This molecular motion is a form of kinetic energy at the microscopic scale.
When heat is added to an object, the molecules absorb energy and begin to vibrate and move faster. The increased molecular motion corresponds to an increase in thermal energy and temperature. For example, heating water on a stove causes the water molecules to vibrate faster and move with greater kinetic energy. This increases the thermal energy and temperature of the water.
Likewise, removing heat from an object causes the molecules to vibrate slower, decreasing their kinetic energy at the molecular level. This corresponds to a decrease in the thermal energy and temperature of the object. For instance, putting ice cubes in a drink removes thermal energy, causing the drink molecules to move slower and lowering its temperature.
Thermal energy is directly proportional to the average kinetic energy of molecules within an object. When thermal energy increases, so does molecular kinetic energy, and vice versa. Therefore, thermal energy and temperature are macroscopic manifestations of the kinetic energy present at the microscopic molecular level.
Conclusion
The key relation between potential and kinetic energy is that they are two forms of energy that can convert between one another. Potential energy is stored energy based on an object’s position or configuration, while kinetic energy is energy associated with motion. According to the work-energy theorem, when net work is done on an object, the object’s kinetic energy changes by an amount equal to the work. This means potential energy can be converted into kinetic energy when net work is done. The law of conservation of energy states that total energy (potential + kinetic) remains constant in a closed system. Energy is never created or destroyed but only converted between different forms. An object at a high position has more potential energy and less kinetic energy. As it falls, potential energy is converted to kinetic energy until all potential energy is depleted when the object hits the ground. Potential energy curves show how potential and kinetic energy vary for interactions like molecular bonds. The ability to interconvert potential and kinetic energy explains many everyday phenomena and is a fundamental concept in physics.