What Is The Process Of Converting Chemical Energy To Light Energy?
All living organisms require energy to survive and carry out processes. Energy comes in different forms that can be converted from one to another. Two key types of energy important for life are chemical energy and light energy.
Chemical energy is the energy stored within the bonds of molecules like ATP, glucose, and fatty acids. Light energy is electromagnetic radiation in the visible spectrum. Through bioluminescence, chemical energy can be converted into light energy.
This process involves storing chemical energy, utilizing a luciferase enzyme, going through an oxidation reaction, reaching an excited state, relaxing to a lower energy state, and emitting a photon of light at a specific wavelength. Understanding the bioluminescence process provides insights into this energy conversion and how it can be applied.
Store Chemical Energy
Some molecules, called luciferins, are able to store chemical energy within their molecular bonds. Luciferins are most commonly found in bioluminescent organisms like fireflies, jellyfish, and deep sea creatures. The firefly luciferin molecule, for example, contains chemical energy stored in the bonds between its carbon, nitrogen, and oxygen atoms.
This stored energy comes from the luciferin molecule being energized through a chemical reaction like oxidation. The energized luciferin is essentially like a charged battery, with the high energy electrons ready to be released. Once the right reaction occurs, triggered by an enzyme like luciferase, those energized electrons get discharged and release energy in the form of light.
Luciferase Enzyme
The key enzyme involved in bioluminescence is called luciferase. Luciferase catalyzes the oxidation reaction of the luciferin substrate, which provides the energy to produce light. There are different types of luciferase enzymes found in various bioluminescent organisms, with each one specifically matched to oxidize a particular luciferin molecule. For example, firefly luciferase acts on firefly luciferin, Renilla luciferase acts on coelenterazine, and bacterial luciferase acts on a long chain aldehyde luciferin.
Luciferase enzymes and luciferins evolved in tandem, so that a specific luciferase only reacts with its paired luciferin substrate. The luciferase enzyme lowers the energy needed for the oxidation reaction to occur, making bioluminescence viable. Without luciferase, the energy required to oxidize luciferin would be too high for the reaction to proceed efficiently at ambient temperatures. Luciferase is an oxidoreductase that forms an enzyme-substrate complex with luciferin, correctly orienting the luciferin to maximize the quantum yield of the reaction. Overall, luciferase facilitates the oxidation reaction and light emission by catalyzing the chemical reaction between luciferin and oxygen.
Oxidation Reaction
Luciferin undergoes an oxidation reaction, meaning it loses electrons and is oxidized. The enzyme luciferase catalyzes this reaction, making it occur much faster than it would on its own. In the oxidation reaction, luciferin is broken down and converted to oxyluciferin. This chemical reaction releases energy, similar to how the breakdown of sugars releases energy during cellular respiration.
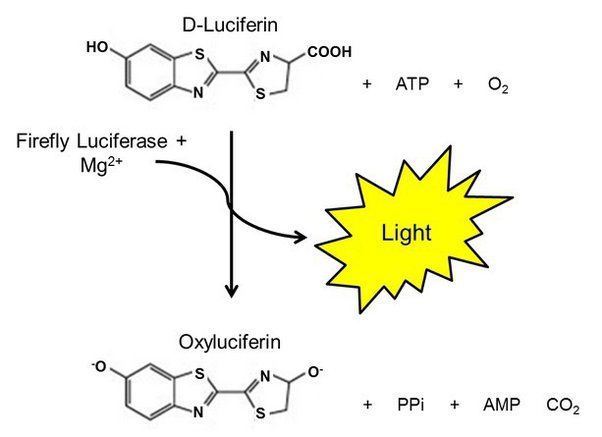
The oxidation of luciferin is unique compared to more common redox reactions. Most redox reactions involve the transfer of electrons from a reducing agent to an oxidizing agent. But in bioluminescence, luciferin acts as both the reducing agent and the oxidizing agent – it donates electrons but also accepts them at a different part of the molecule. This intramolecular electron transfer allows luciferin to oxidize without an external oxidizing agent.
Additionally, the oxidation reaction in bioluminescence occurs very efficiently. Luciferase enhances the reaction so that nearly all the energy released is in the form of light. In contrast, most oxidation reactions convert the released energy to heat rather than light.
Excited State
When the energy is released from the oxidation reaction, it excites the electrons in the luciferin molecule to higher energy levels. Normally, the electrons exist in a ground state, which is the lowest possible energy level for the electrons. However, the influx of energy from the reaction causes the electrons to jump to excited electronic states at higher energy levels.
In the excited state, the electrons occupy orbitals that are farther from the nucleus. The excited electronic state is unstable and the electrons quickly return to lower energy levels. As they return to the ground state, the electrons release energy in the form of photons of light. The color or wavelength of light released depends on the energy difference between the excited state and the ground state when the electrons return.
Relaxation
Once the electrons reach an excited state, they are unstable and seek to return to their ground state. This process is called relaxation. When the electrons fall back down to their ground state, they release energy in the form of photons of light.
The wavelength of the released photons depends on the energy gap between the excited state and ground state. Larger energy gaps result in shorter wavelength photons, while smaller energy gaps result in longer wavelength photons.
The relaxation process occurs extremely rapidly, on the order of nanoseconds. This quick release of photons is what generates the flash of light in bioluminescent organisms. The color of the light depends on the structure and properties of the particular luciferase enzyme that facilitated the initial excitation.
Wavelength
The wavelength and color of the light emitted during bioluminescence depends on the structure of the luciferin molecule. Slight changes in the luciferin structure can result in different colors of light. For example, the firefly luciferin emits a yellow-green light at around 560 nm. Whereas, the luciferin found in railroadworms emits a blue-green light at around 490 nm. The railroadworm luciferin has a thiazoline ring instead of a thiazole ring in firefly luciferin. Some marine organisms contain a cyclopentenone ring in their luciferin, resulting in a bluer light around 465 nm.
So by modifying the luciferin structure, bioluminescent organisms can emit different colors of light. Red and near infrared emitting luciferins have also been synthesized in laboratories by changing the structure. This demonstrates how the wavelength and color is directly related to the luciferin molecule.
Quantum Yield
The quantum yield is a measure of the efficiency of the chemiluminescent process. It refers to the ratio of the number of photons emitted to the number of reactant molecules that underwent the chemical reaction. A quantum yield of 1 would mean every reactant molecule produced a photon of light. In reality, the quantum yield is often less than 1.
There are several factors that can affect the quantum yield and light emission efficiency:
- Reaction kinetics – How fast the chemical reaction proceeds will impact how many molecules can emit a photon before the reaction completes.
- Excited state reactions – Side reactions or alternative relaxation pathways from the excited state can decrease the photon emission.
- Molecule structure – Some molecular structures promote more efficient light emission than others.
- Environmental conditions – Temperature, pH, solvent etc can influence the quantum yield.
Understanding what factors maximize the quantum yield allows chemists to optimize light production for different chemiluminescent applications.
Applications
Bioluminescence has many useful applications in science and medicine due to its high sensitivity and lack of background noise. Here are some examples:
-
Imaging – Bioluminescent imaging is used to non-invasively track biological processes like tumor growth, bacterial infections, and gene expression in small animal models. This is done by linking the biological process to a luciferase reporter gene.
-
Biosensors – Bioluminescent bacteria can be engineered to detect environmental pollutants or toxins. The light output indicates the presence and concentration of the target substance.
-
Tracking gene expression – Scientists insert luciferase genes under the control of a target gene promoter. The light produced indicates when and where the target gene is expressed in cell cultures or animal models.
-
Food and water testing – ATP bioluminescence assays rapidly detect microbial contamination in food and water samples. The amount of light produced is proportional to the amount of ATP and microorganisms present.
In summary, bioluminescence provides a sensitive, non-invasive way to visualize and quantitate biological processes for research and analytical purposes.
Conclusion
The process of converting chemical energy into light energy is a complex series of reactions catalyzed by the luciferase enzyme. It starts with chemical energy being stored in a molecule like ATP or luciferin. When luciferase acts on this molecule, it catalyzes an oxidation reaction, converting the chemical energy into an excited electronic state. The excited molecule then relaxes, releasing photons of light energy in the process.
The key points in this process are:
– Chemical energy is stored in molecules like ATP or luciferin
– The luciferase enzyme catalyzes the oxidation reaction
– Oxidation converts the chemical energy into an excited electronic state
– The excited molecule relaxes to ground state, releasing a photon
– The wavelength of light depends on the energy gap between ground and excited states
– The quantum yield determines the efficiency of converting chemical energy to light
By harnessing this natural process, scientists have created many applications that employ bioluminescence, from detectors to labels to models helping visualize biological processes and interactions. Overall, the conversion of chemical energy into light energy by luciferase provides a unique and useful tool across many fields.