What Is The Flow Of Energy In Physics?
Energy is the quantitative property that must be transferred to an object in order to perform work on, or to heat, the object. In physics, energy is defined as the ability to do work. Work involves applying a force to move an object across a distance. Force times distance equals work. The amount of work reflects the amount of energy transferred.
The concept of the flow of energy refers to how energy is transferred between objects or systems. Energy flows from one place to another and between objects through work and heat. Energy cannot be created or destroyed, but it can be transformed from one form into another. For example, chemical energy in gasoline can be converted into kinetic energy to move a car. The flow of energy drives all physical and chemical changes, biological processes, and ecosystems.
Forms of Energy
There are several basic forms or types of energy that are observed and studied in physics. These include:
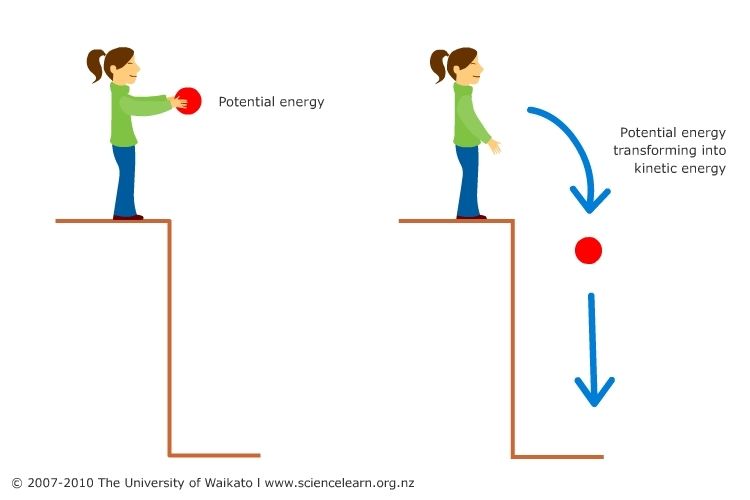
- Potential Energy – This is the stored energy an object has due to its position or state. For example, a ball held at an elevated position has potential energy due to gravity. Other examples include the energy stored in springs, charged batteries, and chemical bonds.
- Kinetic Energy – The energy of motion. Moving objects and particles have kinetic energy proportional to their mass and velocity squared.
- Thermal Energy – The internal energy of a system associated with the random motion of its atoms and molecules. Heat is the transfer of thermal energy between systems.
- Chemical Energy – The potential energy stored in the bonds between atoms and molecules. Chemical energy can be released during chemical reactions.
- Nuclear Energy – The energy stored within an atom’s nucleus and released in nuclear reactions through fission, fusion, or radioactive decay.
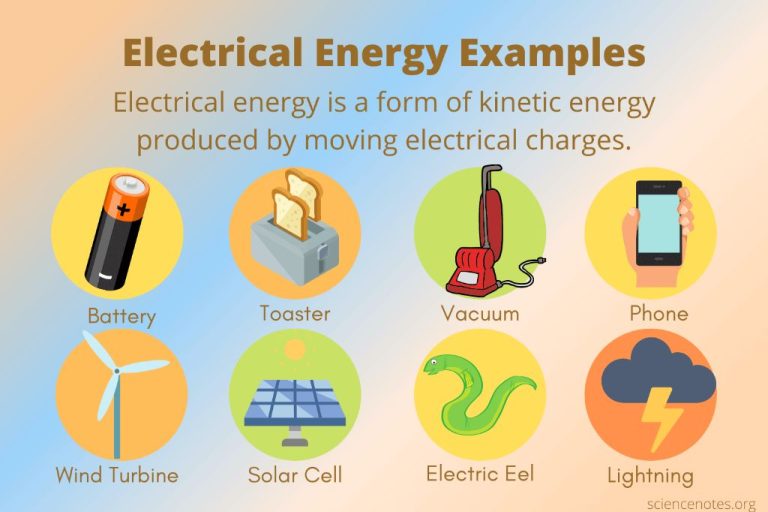
- Electromagnetic Radiation – Energy associated with electric and magnetic fields propagating through space. Examples include radio waves, microwaves, infrared radiation, visible light, ultraviolet rays, and gamma rays.
These different forms of energy can be converted from one to another through various processes. Understanding the transfers and transformations of energy is a key focus in physics.
Energy Transfer
Energy can be transferred between objects through work, heat, and radiation. Work involves applying a force to move an object, while heat is the transfer of thermal energy between objects at different temperatures. Radiation is the emission and absorption of electromagnetic waves, which can also transfer energy.
Work
When a force displaces an object, work is done. More precisely, work is equal to the magnitude of the displacement times the component of the force in the direction of the displacement. For example, lifting a box against gravity performs work equal to the weight of the box times the height it is lifted. In physics, work represents one way of transferring energy between objects through the application of force.
Heat
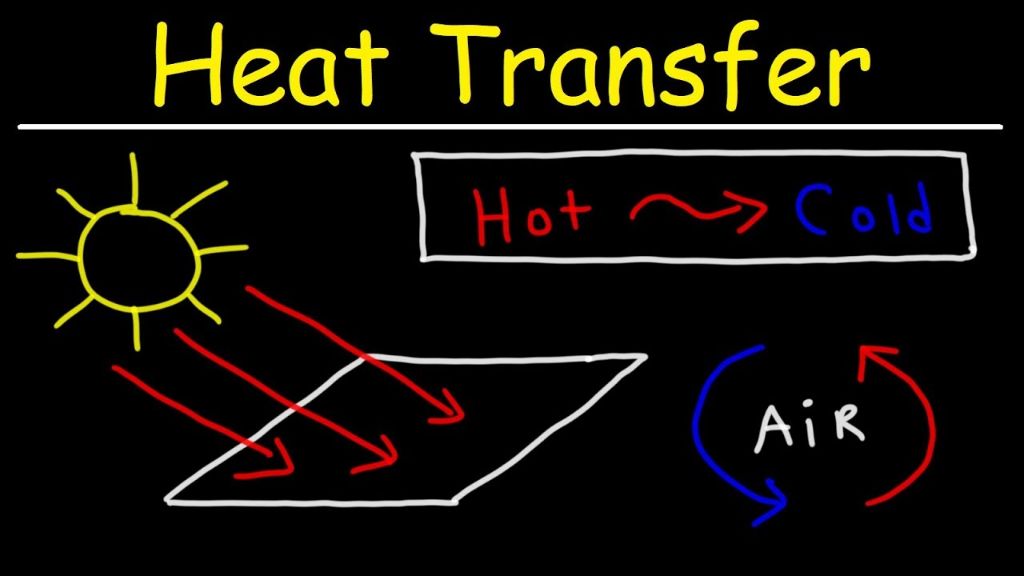
Heat is the transfer of thermal energy driven by temperature differences. It flows spontaneously from objects at higher temperatures to objects at lower temperatures. The amount of heat transferred depends on the heat capacity, temperature change, and mass of an object. Heat can be transferred through conduction, convection, and radiation. For instance, a pot of water on a stove is heated through conduction from the metal to the water molecules.
Radiation
Radiation is the emission and transfer of energy by electromagnetic waves. It does not require a medium for energy to be transferred. All objects emit thermal radiation related to their temperature. Hotter objects tend to emit more total radiation at shorter wavelengths than colder objects. Radiation allows energy to be transferred across vacant space and is responsible for the warmth provided by the sun to the Earth.
Energy Conversion
Energy conversion refers to the process of changing energy from one form to another. Some common examples of energy conversion include:
Chemical to Kinetic
Chemical energy stored in the bonds of molecules can be converted into kinetic energy of motion. For example, the chemical energy stored in gasoline is converted into kinetic energy to power a car engine.
Potential to Kinetic
Gravitational potential energy of an object at a height can be converted into kinetic energy as the object falls. For example, the potential energy of water held behind a dam is converted into electricity as it flows through turbines.
Light to Chemical
The radiant light energy from the sun can be converted into chemical energy through photosynthesis in plants. This process converts carbon dioxide and water into glucose and oxygen.
Electrical to Light and Heat
Electrical energy can be converted into light and heat energy through electrical appliances like light bulbs, heaters, and toasters. The movement of electrons is converted into visible light and infrared radiation.
Understanding the different ways energy can convert between forms is key to designing efficient energy systems and tapping into sustainable energy sources.
Thermodynamics Laws
Thermodynamics is the study of heat, energy, and the interconversion between them. There are four fundamental laws of thermodynamics that describe the relationship between heat, work, energy, and entropy (disorder). These laws place constraints on what can occur in any thermodynamic process.
Zeroth Law of Thermodynamics: If two bodies are in thermal equilibrium with a third body, they are also in thermal equilibrium with each other. This law helps define temperature.
First Law of Thermodynamics: Energy can neither be created nor destroyed, it can only be transformed from one form to another. This is the law of conservation of energy. The total energy of an isolated system remains constant.
Second Law of Thermodynamics: In any spontaneous process, the total entropy of the universe increases. Entropy is a measure of disorder, so this law states that the entropy of an isolated system always increases over time. Useful energy is gradually dissipated into heat due to irreversible processes.
Third Law of Thermodynamics: As temperature approaches absolute zero, the entropy of a system approaches a minimum value. This lowest entropy state is used as a reference point. At absolute zero, molecular motion comes as close to zero as physically possible.
These fundamental thermodynamic laws describe how energy flows and transforms in nature. They govern all heat engines, refrigerators, and other thermal devices. The laws provide boundaries on how efficient such systems can be.
Energy Flow in Ecosystems
Energy flows through ecosystems in one direction from photosynthetic organisms to herbivores to carnivores and decomposers. This flow forms food chains and more complex food webs with multiple interlocking food chains. Food chains show who eats whom with arrows pointing from the resource (food) to the consumer. Each step in the chain is called a trophic level. Green plants and other photosynthetic organisms make up the first trophic level. Herbivores that eat the plants comprise the second trophic level, while carnivores that eat the herbivores occupy the third trophic level. Top carnivores capable of killing other carnivores sit atop the fourth trophic level. Decomposers like bacteria and fungi that break down waste and dead matter are at the end of food chains.
At each transfer between trophic levels, some energy is lost as heat. Only about 10% of the energy at one trophic level is transferred up to the next, while approximately 90% is lost. This energy loss limits most food chains to four to six trophic levels. Too many transfers result in too little usable energy left. Therefore, pyramids of energy and biomass in ecosystems are upright, with the greatest quantities of biomass and energy at the lowest trophic levels.
Energy Accounting
Energy accounting is a critical concept for understanding energy flow and transformations. There are several key principles and metrics for accounting used in physics and engineering:
Exergy refers to the maximum useful work possible during a process by a system as it comes to equilibrium with its environment. Exergy accounts for the quality of energy available to do work, not just the quantity. High-quality energy inputs like electricity have more exergy than low-quality waste heat outputs.
Anergy is the portion of energy during a process that cannot perform physical work. It represents the waste heat given off when converting energy from one form to another. The buildup of anergy is a key consideration in improving efficiency.
Emergy is the amount of available energy of one kind previously required directly and indirectly to generate a product or service. Emergy analysis accounts for all the energy used across the full supply chain. It provides a common unit to compare different energy types used over the lifetime of a product or process.
Careful energy accounting using these principles allows tracking available work potential, irreversible losses, waste heat, and total costs across complex systems. This enables designing more efficient energy flows and improving thermodynamic processes.
Energy Systems Analysis
Energy systems analysis examines the flow and transformations of energy through a system. It is used to track where energy comes from, how it moves through the system, and where it ultimately ends up. This allows for assessing the efficiency by which energy is converted and used within the system.
A key tool in energy systems analysis is the energy flow diagram. These diagrams illustrate the transfers and transformations of energy by using arrows to connect different components of the system. The widths of the arrows represent the magnitude of the energy flow. Energy flow diagrams provide a visual snapshot of the paths along which energy flows and how energy enters, leaves, or accumulates in the system.
Energy systems analysis calculates efficiencies by comparing energy outputs versus energy inputs at each step of the system. This reveals inefficiencies and energy losses. For example, when coal is burned to generate electricity, a large amount of the chemical energy in the coal is lost as waste heat rather than being converted into electrical energy. By tracking these efficiency rates, energy systems analysis identifies targets for improving the system.
Overall, energy systems analysis is crucial for optimizing the use of energy resources and developing more sustainable energy solutions. By elucidating how energy flows and transforms within a system, it enables enhancing efficiencies, reducing waste, and improving processes.
Energy Storage
Energy storage plays a critical role in managing the flow of energy. It provides a buffer between energy generation and energy use, allowing energy to be captured and stored for later use when needed. There are several ways to store energy, each with its own advantages and limitations.
Batteries
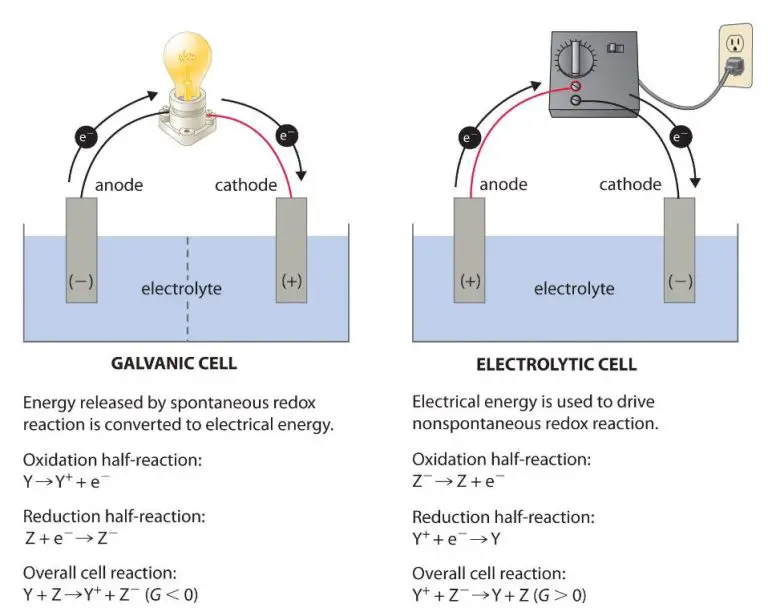
Batteries are electrochemical devices that convert chemical energy into electrical energy. They store energy in chemical form and release it as electricity on demand. Common battery types include lead-acid, lithium-ion, and nickel-cadmium. Batteries are portable, rechargeable, and can store energy for anything from a few hours to several months.
Fuel Cells
Fuel cells generate electricity from an electrochemical reaction involving hydrogen fuel and oxygen from air. The only byproducts are electricity, water and heat. Fuel cells continue generating electricity as long as fuel and oxygen are supplied. They can provide power for anything from small devices to vehicles to buildings.
Mechanical Storage
Mechanical systems store energy in the form of kinetic or potential energy that can be released on demand. Examples include pumped hydroelectric storage, compressed air energy storage, and flywheels. These methods provide large-scale energy storage capabilities for the electrical grid.
Thermal Storage
Thermal energy can be stored in materials and then recovered for heating and cooling applications. This includes storing hot water in insulated tanks for later use, using molten salt to retain heat, and making ice for air conditioning. Thermal storage allows energy to be stored until needed.
Overall, energy storage enables better management of energy flows by balancing supply and demand. It will play an increasingly vital role as more renewable energy is incorporated into energy systems.
Applications and Impacts
Energy use has broad impacts on economic development, sustainability, and the environment. The applications of energy in modern society are vast, from transportation to manufacturing to electricity generation. However, the sourcing and consumption of energy resources can also have unintended consequences.
The burning of fossil fuels like coal and oil contributes significantly to air pollution and global climate change through the emission of carbon dioxide and other greenhouse gases. This has spurred efforts to transition to cleaner renewable energy sources like solar and wind power. Energy efficiency measures are also being implemented to reduce waste and optimize energy use across sectors.
Government energy policies aim to balance priorities like energy security, affordability, and environmental protection. Regulations on fuel economy and power plant emissions shape energy use. Building codes and appliance standards determine energy efficiency. Tax credits and rebates incentivize the adoption of renewable energy and efficiency upgrades.
Sustainability in energy systems will require a multifaceted approach. Expanding renewable energy, electrifying transport, retrofitting buildings, and encouraging conservation by individuals and businesses can promote sustainable energy use. Ongoing research, innovation, and policy reform will be needed to meet rising energy demand in a carbon-conscious manner.