What Is The Cheapest Way To Produce Hydrogen?
Hydrogen is increasingly seen as a key part of the world’s transition to a clean, low carbon energy system. As a versatile energy carrier, it can be used as a fuel for transportation, power generation, and industrial applications (IEA, 2021). With its high energy density and ability to be produced from diverse domestic resources, hydrogen has the potential to reduce reliance on fossil fuels and lower greenhouse gas emissions across multiple sectors. However, most hydrogen today is produced from natural gas, which results in carbon emissions. As demand grows, there is a need to scale up affordable production of low-carbon hydrogen from renewable electricity or fossil fuels with carbon capture.
Global hydrogen demand is currently around 90 million metric tons per year and production emits approximately 830 million tons of CO2 annually (IEA, 2023). But as countries pursue decarbonization goals, demand for clean hydrogen could grow substantially. The International Renewable Energy Agency projects hydrogen could meet 12% of global energy demand by 2050. Realizing this potential in a sustainable way requires bringing down costs through technology innovation and economies of scale for electrolysis, renewable electricity, biomass gasification, and carbon capture methods.
With multiple possible production pathways, determining the cheapest option depends on factors like local energy resources, government policy incentives, and technological progress. This overview examines low-carbon hydrogen production costs to assess the most affordable routes today and in the future.
Electrolysis of Water
Electrolysis is the process of using electricity to split water into hydrogen and oxygen. This is done by passing an electric current through the water, which causes the water molecules (H2O) to separate into hydrogen (H2) and oxygen (O2) gas. The reaction takes place in a unit called an electrolyzer, which contains electrodes and an electrolyte solution to allow current to pass through.
There are several types of electrolyzers, but proton exchange membrane (PEM) electrolyzers are currently the most efficient and cost-effective for hydrogen production. PEM electrolyzers can achieve efficiency of 60-80% and have lower capital costs than other options [1]. However, the electricity costs required for the electrolysis process account for about 80% of the total hydrogen production cost [2]. So to lower the overall costs, access to low-cost renewable electricity sources is essential.
Research is ongoing to improve electrolyzer efficiency and bring down their capital costs through new materials and manufacturing methods. But electrolysis remains one of the more expensive hydrogen production methods today.
Steam Methane Reforming
Steam methane reforming (SMR) is currently the most common and cost-effective method for producing high purity hydrogen. It involves reacting methane from natural gas with steam at high temperatures (700–1100°C) and pressures (3–25 bar) in the presence of a catalyst to produce hydrogen and carbon monoxide. The overall reaction is:
CH4 + H2O → CO + 3H2
The process consists of two main steps – steam reforming followed by water-gas shift reaction. The steam reforming reaction is endothermic and requires external heating, which accounts for around 85% of the production costs. The conversion of natural gas is around 80% per pass. Then the water-gas shift reaction converts the CO into CO2 and more H2. After further purification, 99.999% pure hydrogen can be achieved.[1]
SMR has a typical efficiency of around 65-75%. The estimated cost of production is around $1.10 – $1.60 per kg of hydrogen.[2] However, this process results in significant CO2 emissions of around 9-12 kg per kg of hydrogen produced. The cost can be reduced by capturing and storing the CO2, but this adds an additional cost of $0.10-0.15 per kg.[3]
Coal Gasification
Coal gasification is a process that converts coal into syngas, which is a mixture of hydrogen, carbon monoxide, and carbon dioxide. This syngas can then be further processed to separate out the hydrogen.
The basic steps in coal gasification are:
- Coal is milled into a fine powder.
- The coal powder is fed into a gasifier along with steam and oxygen under high temperatures (1300–1500°C) and pressures (2.5–7 MPa).
- Inside the gasifier, the coal reacts with the steam and oxygen to produce syngas.
- The raw syngas is cooled and cleaned to remove impurities.
- Additional chemical processing separates the hydrogen from the other gases.
Some key factors affecting efficiency and cost of hydrogen production via coal gasification include:
- Capital costs for building a coal gasification and syngas processing plant are estimated at around $1,000 per kW of syngas capacity (source).
- Operating costs depend heavily on coal feedstock prices.
- The process generates carbon dioxide as a byproduct that must be captured and stored to avoid emissions.
- Overall energy efficiency is around 50-60% for converting the heating value of coal into hydrogen chemical energy.
According to analysis by the Department of Energy, the estimated cost of hydrogen production via coal gasification ranges from $1.40 to $2.80 per kg when capturing and storing the carbon emissions underground (source).
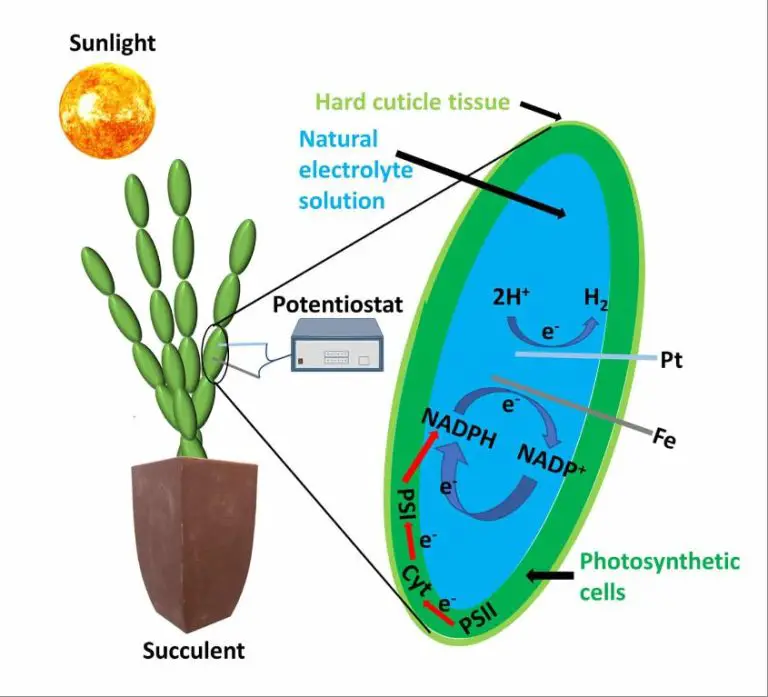
Photoelectrochemical Water Splitting
Photoelectrochemical (PEC) water splitting uses sunlight to directly split water into hydrogen and oxygen. This process utilizes specialized semiconductors called photoelectrodes that can absorb light and use the energy to power electrochemical reactions. When the photoelectrode is immersed in water and illuminated, it generates an electric current that splits water molecules into H2 and O2.
PEC water splitting is an emerging technology that offers the potential for sustainable hydrogen production using only sunlight and water as inputs. However, currently PEC hydrogen production costs are relatively high, around $10/kg H2, which is nearly 7 times more expensive than steam methane reforming at $1.5/kg H2 [1]. The U.S. Department of Energy has set cost targets of $2/kg H2 for PEC by 2025 and $1/kg H2 by 2030 [2].
Bringing down the costs of PEC hydrogen production will require improvements in photoelectrode efficiency, stability, and fabrication methods. Key areas of research are developing new semiconductor materials, catalysts, and device architectures to increase solar-to-hydrogen efficiencies above 20%. There is also a need to improve manufacturing techniques and scale up systems to produce low-cost, large-area photoelectrodes. If R&D efforts are successful in surpassing these technical targets, PEC could emerge as a viable renewable option for solar hydrogen production in the coming decades.
Microbial Biomass Conversion
Microbial biomass conversion utilizes microbes like algae and bacteria to produce hydrogen through biological processes like biophotolysis, photo-fermentation, and dark-fermentation [1]. In biophotolysis, microalgae use sunlight to split water molecules into hydrogen and oxygen. In photo-fermentation, photosynthetic bacteria produce hydrogen from organic acids. Dark-fermentation uses anaerobic bacteria to produce hydrogen from organic matter in the absence of light.
Biotech research has focused on engineering microbes and biochemical pathways to increase hydrogen yields and make the process more efficient and cost-effective. However, microbial biomass conversion currently has high production costs ranging from $2.13 to $7.24 per kg of hydrogen produced [1]. Key factors influencing cost include bioreactor design, bacterial strain selection, biomass feedstock prices, and efficient hydrogen separation and purification.
Further advances in genetic engineering, systems biology, and process integration can potentially reduce costs by improving microbial hydrogen production rates and yields. More research is needed to develop scaled-up systems that can achieve economies of scale.
Cost Comparison

The cost of hydrogen production varies widely depending on the method used. According to one study published in ScienceDirect, biohydrogen production costs range from 1.2–4.3 €/kg H2, steam methane reforming costs around 1-3 €/kg H2, and water electrolysis can cost anywhere from 2.5-10 €/kg H2.
A 2021 comparison of net-zero hydrogen production pathways found that electricity-based methods like water electrolysis cost $3-8/kg H2, while fossil-based methods like steam methane reforming and coal gasification cost around $1-3/kg H2 according to the International Energy Agency.
In summary, the least expensive options are currently fossil fuel-based methods like steam methane reforming and coal gasification. The most expensive is water electrolysis using renewable electricity. However, as renewable energy costs continue to fall, electrolytic hydrogen is expected to become cost-competitive with fossil-based production in the coming years.
Challenges
There are significant technical and economic challenges facing affordable hydrogen production today. The key hurdles include:
High capital costs – Building electrolyzers, reformers, gasifiers, and other production facilities requires major upfront investments, limiting widespread adoption (https://100re-map.net/en/green-hydrogen-market-potentials-and-challenges/).
Efficiency limits – Most production methods operate at 50-80% efficiency, with significant energy losses during hydrogen generation. Improving efficiency could dramatically lower costs (https://www.ncbi.nlm.nih.gov/pmc/articles/PMC10608639/).
Carbon emissions – Steam reforming produces significant CO2 emissions, while gasification and electrolysis can also have carbon footprints depending on the energy source. Methods are needed to cut emissions and boost sustainability.
Intermittency challenges – Renewable energy supplies used for production fluctuate based on weather and time of day. Better storage and load balancing solutions can help address intermittency.
To tackle these barriers, increased R&D funding is needed in areas like advanced electrolyzer and fuel cell materials, renewable integration, and modular production systems. Policy incentives for clean hydrogen, such as investment tax credits, are also critical to scale up production and drive down costs (https://www.jstor.org/stable/resrep26335.5?seq=8).
Future Outlook
Projections show that the costs of green hydrogen production will continue to decrease over the coming decades. According to a report by PwC, hydrogen production costs are estimated to drop by around 50% by 2030 and will continue falling steadily until 2050. They predict that by 2050, green hydrogen costs could be as low as $1-2 per kg.[1]
Many experts predict that electrolysis of water using renewable electricity will become the dominant method for green hydrogen production in the future. This is due to the expected decreases in renewable electricity costs as well as advancements in electrolyzer technology that will improve efficiency and lower capital costs. Some projections estimate that electrolysis could achieve costs below $2/kg by 2030.[2]
However, other methods like biomass gasification may still play an important role, especially in certain geographic regions where biomass feedstocks are abundant and low-cost. The optimal production methods will likely vary based on local conditions and energy resources.
Conclusion
In summary, the cheapest methods for producing hydrogen today are steam methane reforming and coal gasification. These methods benefit from economies of scale and utilize widely available feedstocks, though they produce carbon emissions. Emerging methods like photoelectrochemical water splitting have potential to produce low-cost, clean hydrogen through renewable solar energy, but more research and development is still needed.
No matter which production method is used, it is crucial that hydrogen can be made affordable at scale for mass adoption of hydrogen vehicles, energy storage, and other applications. With continued innovation and declining renewable electricity prices, there is promise for very low cost, green hydrogen in the future.