What Is Chemical To Thermal To Electrical Energy?
Energy is the ability to do work or produce heat. There are many different forms of energy such as chemical, thermal, electrical, nuclear, and more. This article will focus on chemical, thermal and electrical energy specifically.
Chemical, thermal and electrical energies can be converted between each other through various processes. For example, chemical energy stored in the bonds of molecules like gasoline or food can be released as thermal energy through combustion or cellular respiration. Thermal energy can then be used to produce electrical energy through turbines and generators. The ability to convert between these different energy types allows us to harness and utilize them in many practical ways.
This article will provide an in-depth look at chemical, thermal and electrical energies, and the processes by which they can be interconverted.
Chemical Energy
Chemical energy is the potential energy stored in the bonds between atoms and molecules. It is the energy released when a chemical compound reacts or changes form. There are several examples of chemical energy:
– Food: The nutrients in food contain chemical energy that is released when food is digested by the body. Foods like fats, carbohydrates and proteins all contain chemical bonds that store energy.
– Fuel: Gasoline, diesel, natural gas and other fuels are rich in chemical energy from the hydrocarbons they contain. When combusted in an engine, the chemical bonds are broken and chemical energy is released as heat and kinetic energy.
– Batteries: The chemicals stored in batteries contain high chemical energy that is released as electrical energy when they undergo reactions. Common battery chemistries like lithium-ion, lead-acid and alkaline all convert chemical energy to electricity.
Thermal Energy
Thermal energy refers to the total kinetic energy and potential energy of molecules within a substance. It is directly related to the temperature and heat of an object or system.
Temperature measures the average kinetic energy of molecules, while heat refers to the transfer of thermal energy between objects at different temperatures. The higher the temperature of a substance, the greater the thermal energy since the molecules have more kinetic energy.
Some examples of thermal energy include:
- Heat generated from combustion reactions like burning wood or fossil fuels.
- Heat radiating from hot objects like stovetops, engines, or the sun.
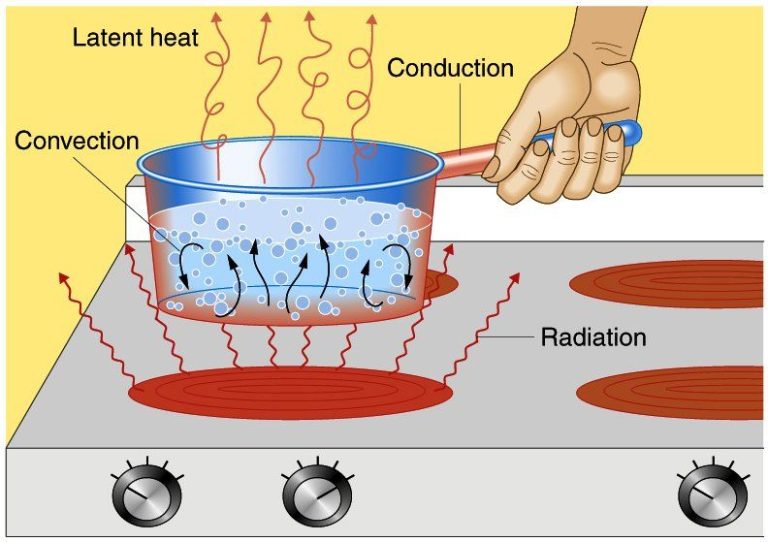
- Heat conducted through materials in contact, like a pot heating up on a stove.
In all these examples, there is a transfer of thermal energy from a high-temperature object to a lower-temperature object until they reach thermal equilibrium.
Electrical Energy
Electrical energy is the energy carried by moving electrons in an electric current. It is closely related to electricity, which flows due to the movement of electrons. Electrical energy is often generated at power stations by electromechanical generators, which convert mechanical energy into electrical energy.
Electrons are tiny particles found in atoms that carry a negative charge. When electrons move from one place to another, such as through a wire, electrical energy is transferred. The flow of electrons is called an electric current. The force driving the movement of electrons is measured in volts.
Some common examples of electrical energy include batteries, which store chemical energy and convert it into electrical energy, generators at power plants that convert mechanical energy into electrical energy, and solar cells that convert light energy into electrical energy. Electrical energy powers many devices and appliances in homes and workplaces, from lights and motors to computers and cell phones. It is an extremely useful and versatile form of energy.
Chemical to Thermal
Chemical energy can be converted into thermal energy through exothermic chemical reactions like combustion, digestion, and cellular respiration.
During combustion, a fuel source reacts with oxygen to produce carbon dioxide, water, and heat. For example, when natural gas burns, the chemical energy in the natural gas bonds is converted into thermal energy in the form of heat and light. This allows us to cook food or heat our homes with gas stoves and furnaces.
In digestion, the chemical energy stored in the bonds of large food molecules like carbohydrates, fats, and proteins is released through catabolic reactions. The energy released from breaking these bonds is converted into thermal energy that helps maintain body temperature.
Cellular respiration also converts chemical energy from glucose into thermal energy. Within cells, the glucose molecule is broken down through a series of reactions like glycolysis and the Krebs cycle. The energy released allows cells to perform work and produces heat as a byproduct.
In all these examples, breaking down larger molecules through chemical reactions releases energy that is then converted into thermal energy in the surrounding environment.
Thermal to Electrical Energy
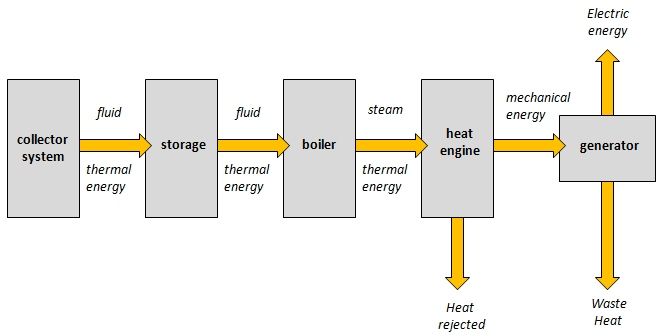
The conversion of thermal energy into electrical energy commonly takes place in power plants that use heat to generate electricity. There are several ways this can occur:
Power plants – Many power plants, such as coal, natural gas, and nuclear plants, use heat to boil water and produce steam that spins a turbine connected to a generator that produces electricity. The thermal energy from burning fuel or nuclear reactions is converted into mechanical energy by the turbine, then into electrical energy by the generator.
Thermoelectric effect – When two metals or semiconductors joined together have a temperature difference, electrons flow from the hotter side to the cooler side, generating an electrical voltage. This phenomenon is called the thermoelectric effect or Seebeck effect. Thermoelectric generators can convert heat directly into electricity using this effect.
Seebeck effect – Discovered in 1821 by Thomas Seebeck, the Seebeck effect describes how an electromotive force is produced when the junctions of two different conductors or semiconductors are kept at different temperatures. This phenomenon is used in thermocouples to measure temperature differences and in thermoelectric generators to produce electricity from heat differentials.
Electrical to Chemical
Electrical energy can be converted into chemical energy through a process called electrolysis. Electrolysis uses an electric current to drive a non-spontaneous chemical reaction, like the splitting of water into hydrogen and oxygen. The electric current provides the energy needed to break the bonds between the hydrogen and oxygen atoms in water molecules.
The amount of electrical energy put into the electrolysis process determines the amount of chemical energy that is stored in the resulting hydrogen and oxygen gases. The gases can then be burned later to release energy, converting the chemical energy back into other forms like heat and light.
Another example of converting electrical to chemical energy is charging batteries. Batteries store chemical energy and convert it into electrical energy. When a battery is charged, an external electrical power source provides energy to reverse the chemical reaction inside the battery that produces electricity. This regenerates the chemicals so the battery can continue producing electricity when needed. The electrical energy during charging gets stored in the battery as chemical potential energy.
Chemical to Electrical
There are two main ways to convert chemical energy into electrical energy: batteries and fuel cells.
Batteries contain two electrodes separated by an electrolyte. Chemical reactions at the electrodes produce an electric current that can be used to power electrical devices. Common battery types include lead-acid batteries used in vehicles and lithium-ion batteries used in consumer electronics.
Fuel cells convert the chemical energy in a fuel into electricity through a chemical reaction. They require a continuous source of fuel and oxygen to run. Fuel cells are very efficient at extracting energy from fuel and have potential applications in electric vehicles and power generation. The most common fuel cell type uses hydrogen as a fuel and oxygen from the air to generate electricity.
Both batteries and fuel cells convert the energy stored in chemical bonds into electrical current that can power devices and machines. Batteries have the advantage of being self-contained energy storage devices while fuel cells can continuously generate electricity as long as they are supplied with fuel and oxygen.
Thermal to Chemical
Thermal energy can be converted into chemical energy through endothermic chemical reactions. Endothermic reactions require an input of heat energy to proceed. This heat energy gets absorbed by the reactants, raising their internal energy. The increased internal energy allows the reactants to overcome activation barriers and transform into products.
A common example of an endothermic reaction is photosynthesis. Plants use the sun’s thermal radiation to drive photosynthesis. The heat energy gets absorbed by chlorophyll molecules, providing the energy needed to convert carbon dioxide and water into glucose and oxygen. The overall chemical reaction requires 289 kJ of energy input per mole of glucose produced.
Another example is the industrial production of ammonia through the Haber process. Nitrogen and hydrogen gases combine to form ammonia, but only with the input of significant amounts of heat energy, typically around 200-400 kJ per mole of NH3. This endothermic reaction would not occur without the external supply of thermal power.
In both cases, thermal energy gets converted into the chemical energy contained in the newly formed bonds of the reaction products. The heat provides the necessary work to reconfigure chemical bonds into higher energy arrangements. This demonstrates the conversion of thermal energy to chemical energy through endothermic chemical reactions.
Summary
In summary, we explored the processes of converting energy from one form to another. Chemical energy stored in molecules can be converted into thermal energy through combustion or metabolic reactions. Thermal energy results in increased molecular motion and can be used to generate mechanical power. Mechanical energy can then be converted into electrical energy through generators and dynamos. Electrical energy is highly useful for powering devices and appliances. The ability to convert energy between chemical, thermal, and electrical forms is essential for modern society. Energy conversions enable us to harness stored chemical energy and convert it into usable electricity that powers our homes, appliances, and devices. Understanding these fundamental energy conversions provides key insights into how we utilize different energy sources.