What Energy Is Absorbed During Light Reactions?
Photosynthesis is the process plants and some microorganisms use to convert sunlight into chemical energy. This chemical energy is stored in the bonds of glucose molecules, which are made from carbon dioxide and water. The conversion of sunlight into chemical energy takes place in two stages of photosynthesis: the light reactions and the Calvin cycle.
The light reactions begin the process of photosynthesis by harnessing energy from sunlight. When light hits the leaves of a plant, it is absorbed by pigments such as chlorophyll in the chloroplasts of the plant cells. This excites electrons in the pigments and drives the transfer of electrons, creating chemical energy molecules like ATP and NADPH. The light reactions transform solar energy into the chemical energy that will power the rest of photosynthesis.
Sunlight
Sunlight contains a wide spectrum of electromagnetic radiation, including ultraviolet (UV), visible light, and infrared wavelengths. The most important component for photosynthesis is visible light, which ranges in wavelength from approximately 400 to 700 nanometers. Visible light is the part of sunlight that our eyes can detect and perceive as color. However, plants don’t have complex visual systems like humans and cannot actually “see” sunlight. Instead, they have specialized molecules called pigments that can absorb visible light of certain wavelengths.
Ultraviolet radiation, with wavelengths from 10 to 400 nanometers, is also present in sunlight. A small portion of UV light is absorbed and utilized during photosynthesis. However, too much UV exposure can damage plants, so many have evolved mechanisms to protect themselves by absorbing and blocking some amount of UV.
Infrared radiation, with wavelengths from 700 nanometers to 1 millimeter, makes up over half of the sunlight spectrum. While plants don’t use infrared light directly for photosynthesis, the warmth it provides plays important roles in temperature regulation, water absorption, and enzyme activity.
Photosynthetic Pigments
Plants contain specialized pigment molecules that can absorb light energy. The main photosynthetic pigments in plant cells are chlorophylls. There are several forms of chlorophyll, but chlorophyll a and chlorophyll b are the most important for light absorption. Chlorophyll gives plants their characteristic green color and absorbs light mostly in the blue and red regions of the visible light spectrum.
In addition to chlorophyll, plants also contain accessory pigments such as carotenoids. Carotenoids absorb light in the 400-500 nm wavelength range, which chlorophylls cannot absorb. By absorbing different wavelengths of light, the combination of chlorophylls and carotenoids allows plants to utilize a larger portion of the visible light spectrum. Some of the energy absorbed by carotenoids can also be transferred to chlorophyll to be used in photosynthesis.
Light Absorption
Photosynthetic organisms like plants contain light-absorbing molecules called pigments that are key to absorbing light energy. The main pigments involved in photosynthesis are chlorophyll a, chlorophyll b, carotenoids, phycobilins and phycoerythrin.
Each type of pigment can only absorb light at specific wavelengths. For example, chlorophyll a absorbs violet-blue and red light, while carotenoids absorb blue-green light. When a pigment absorbs light, the energy from the photon of light excites an electron in the pigment molecule to a higher energy state.
Because different pigments absorb light at different wavelengths, plants contain a mixture of pigments like chlorophyll, carotenoids and phycobilins. This allows them to efficiently absorb light energy across the visible light spectrum.
The amount and type of pigments present allow plants to adapt to their light environments. Plants grown in the shade maximize light absorption by increasing levels of chlorophyll b and carotenoids.
Photons
Sunlight is composed of packets of energy called photons. Photons are particles that exhibit properties of both waves and particles. The energy of a photon is proportional to its wavelength. Shorter wavelengths have higher energy and longer wavelengths have lower energy.
When a photon of sunlight hits the pigments in the chloroplast, the photon may be absorbed. If the photon has enough energy, it raises an electron in the pigment molecule to a higher energy level. This excitation of the electron is the first step in converting light energy into chemical energy that can be used by plants.
The number of photons absorbed and their energy determines how much light energy is available for photosynthesis. Different wavelengths of light contain photons with different energies. The pigments are optimized to absorb photons in the visible light spectrum, which is the type of light that is most abundant in sunlight.
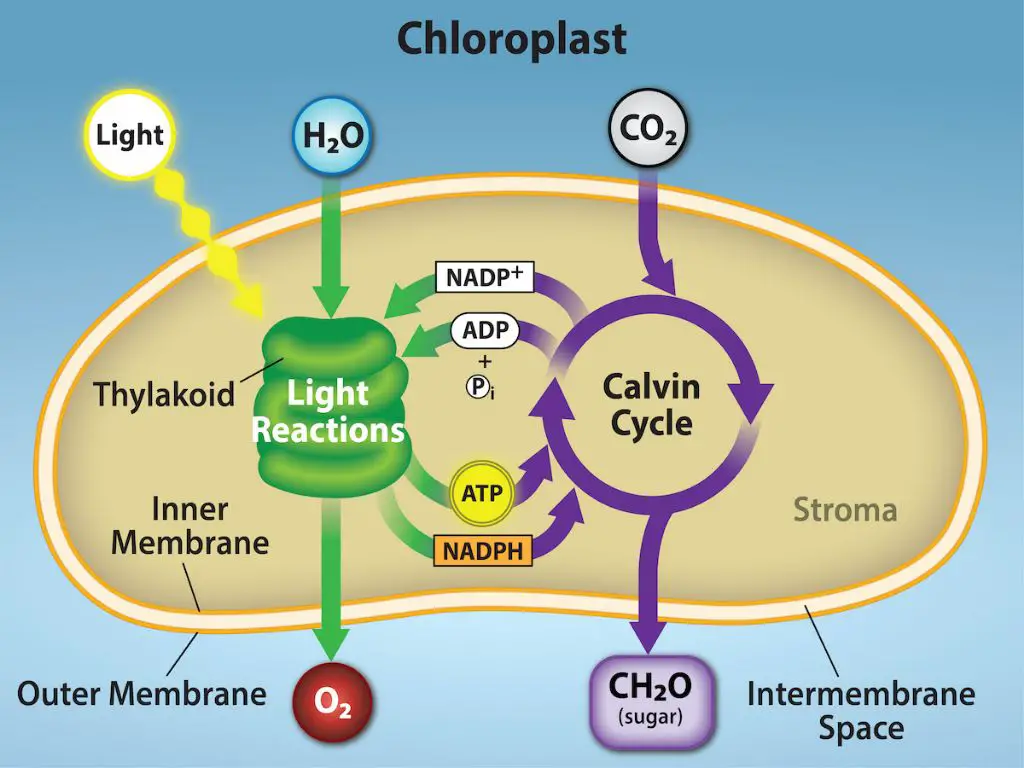
Excitation of Electrons
When photons are absorbed by chlorophyll in the photosystems, the energy from the photons causes electrons in the chlorophyll molecules to become excited to a higher energy state. The absorbed energy excites an electron enough to be transferred and move freely to other molecules. Specifically, chlorophyll absorbs photons most strongly in the red and blue portions of the visible spectrum. The different types of chlorophyll, including chlorophyll a and chlorophyll b, work together to absorb light across a wide range of wavelengths. When a photon is absorbed by chlorophyll, the energy is funneled to a specialized pair of chlorophyll a molecules at the reaction center of the photosystem. This transfer of energy causes the excited electron to be boosted to an even higher energy level, allowing it to be available for the electron transport chain.
Electron Transport Chain
The electron transport chain (ETC) is a series of proteins and organic molecules within the thylakoid membrane that shuttle electrons. When electrons are excited by photons during the light reactions, they are picked up by the electron carrier NADP+. NADP+ passes these high-energy electrons through the ETC.
As the electrons move through the ETC, they go through a series of redox reactions, with each component of the chain becoming reduced as it accepts electrons. The energy released during these redox reactions is used to pump hydrogen ions (H+) across the thylakoid membrane into the lumen.
This creates a proton gradient and membrane potential across the thylakoid membrane. The ETC ends with the electrons combining with hydrogen and oxygen to form water. The energy stored in the proton gradient is used to power ATP synthase, which produces ATP from ADP + Pi.
ATP Synthesis
The excited electrons from the electron transport chain contain energy that is used to fuel ATP synthesis. ATP, or adenosine triphosphate, is the main energy currency of cells. As the high-energy electrons flow down the electron transport chain, they pass through protein complexes embedded in the thylakoid membrane. These protein complexes use the energy from the electron flow to pump hydrogen ions (H+) across the thylakoid membrane, generating an electrochemical gradient.
This gradient builds up a high concentration of hydrogen ions in the thylakoid space. The protein complex ATP synthase acts as a channel that allows the hydrogen ions to diffuse back across the membrane down their concentration gradient. As they flow through ATP synthase, their energy is used to drive the phosphorylation of ADP, generating ATP. So in summary, the energy from the flowing electrons is used to pump hydrogen ions across a membrane. Then, the flow of hydrogen ions back down their gradient provides the power to make ATP.
NADPH
In addition to ATP, the light reactions also produce the energy carrier molecule NADPH. NADPH is produced when electrons get energized by photons and transported down the electron transport chain. The energized electrons are used to convert NADP+ to NADPH.
NADPH provides the chemical energy that is later used in the Calvin cycle to fix carbon dioxide and build carbohydrates. Just like ATP, NADPH provides the fuel for steps that require energy during photosynthesis. The production of NADPH along with ATP during the light reactions provides the chemical energy that drives the rest of photosynthesis.
Summary
In summary, during the light reactions of photosynthesis, light energy from the sun is absorbed by photosynthetic pigments like chlorophyll in plant cells. This absorbed light energy causes the excitation of electrons in the pigments. The excited electrons get transferred through an electron transport chain, which drives the synthesis of ATP. The electron transport chain also results in the reduction of NADP+ to NADPH. Therefore, through the light reactions, light energy gets converted into chemical energy in the form of ATP and NADPH. These energy carriers are then used in the Calvin cycle reactions to fix carbon from CO2 into glucose. To recap, the light reactions involve the absorption of light energy and its conversion into chemical energy that can be used to power the rest of photosynthesis.