What Do You Mean About Light?
Light is fundamental to our understanding of the universe. At its most basic, light is the part of the electromagnetic spectrum that is visible to the human eye. This narrow band of wavelengths provides the illumination that makes sight possible. Beyond enabling vision, light exhibits properties of both waves and particles, which leads to unique interactions with matter. Harnessing these properties has enabled revolutionary technologies like lasers, fiber optics, imaging systems, and more.
Light comes from various natural and artificial sources. The sun is the primary source of natural light on Earth, while common artificial light sources include incandescent bulbs, fluorescent lamps, LEDs, and more. Regardless of origin, visible light possesses qualities essential to life, discovery, and innovation. This includes the capacity to propagate through space, reflect off surfaces, refract through media, and convey color sensations. By studying light from stars, galaxies, and other cosmic objects, scientists gain insight into the origin and evolution of the universe. On the human scale, light is vital for agriculture, energy generation, communications, medicine, and innumerable other applications across industries. For these reasons and more, light remains indispensable to society today.
Properties of Light
Light exhibits properties as both waves and particles. As a wave, light has a speed, wavelength, frequency, amplitude, and polarization. As particles called photons, light has discrete energy levels based on its frequency or wavelength.
The speed of light in a vacuum is a fundamental constant of nature, approximately 300,000,000 meters per second. This speed is thought to be the maximum speed possible in the universe based on Einstein’s theory of special relativity.
Wavelength refers to the distance between successive wave peaks and is typically measured in nanometers (billionths of a meter) or micrometers (millionths of a meter). The visible light spectrum that humans can see ranges in wavelength from about 400 to 700 nanometers.
Frequency refers to how many wave cycles pass a point per second and is measured in hertz (cycles per second). Frequency and wavelength are related in that higher frequency light has a shorter wavelength.
Amplitude corresponds to the intensity or brightness of light. Higher amplitude waves are more intense.
Polarization describes the orientation of light waves. Polarized lenses filter out light waves oriented in one direction to reduce glare.
When atoms emit or absorb light, they do so at specific frequencies corresponding to discrete energy levels. This emission spectrum is unique to each element and can be used to identify elements present in light sources.
Electromagnetic Spectrum
The electromagnetic spectrum encompasses all forms of electromagnetic radiation, ranging from radio waves on the low-energy end to gamma rays on the high-energy end. Electromagnetic radiation is characterized by its wavelength or frequency.
Some key types of electromagnetic radiation include:
- Radio waves – Long wavelength, low frequency waves used for radio communication, TV broadcasting, GPS, and microwave ovens.
- Microwaves – Shorter wavelength than radio, used for radar, satellite communication, and microwave ovens.
- Infrared – Invisible to human eyes, used for thermal imaging and remote controls.
- Visible light – The narrow slice of EM spectrum visible to human eyes. Ranges in wavelength from violet to red.
- Ultraviolet – Shorter wavelength than visible light, carcinogenic in high doses but used beneficially for disinfection.
- X-rays – High frequency waves that can penetrate soft tissue and are used for medical imaging and security scanning.
- Gamma rays – Highest frequency EM waves, extremely dangerous but used for cancer treatment.
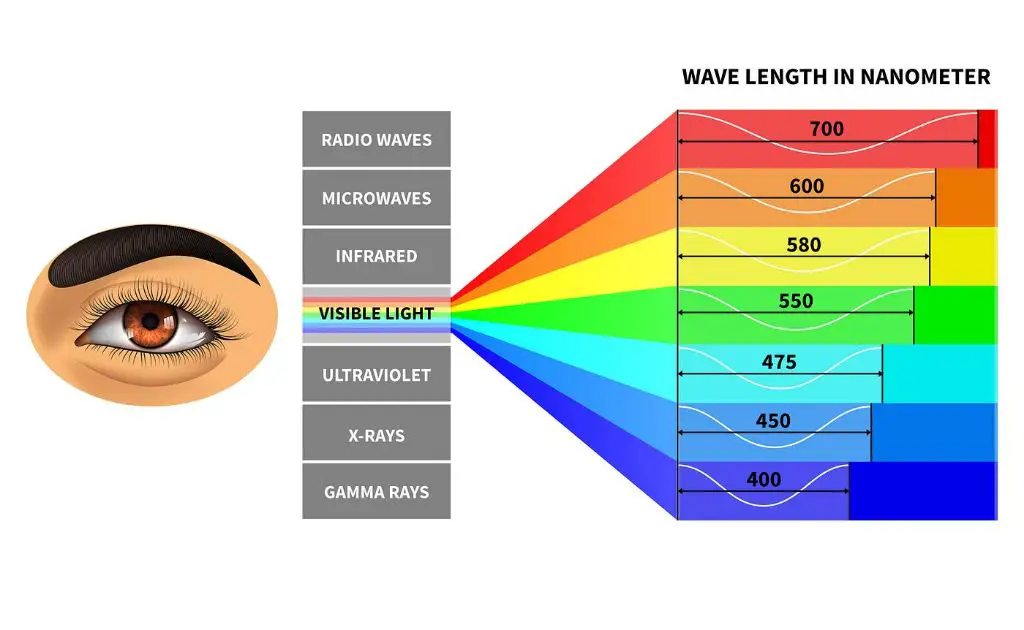
Understanding the electromagnetic spectrum allows us to harness different types of radiation for various beneficial technologies, from radio broadcasting to medical imaging. The visible light portion also enables human vision.
Reflection and Refraction
When light hits a surface, some of it is reflected and some of it is refracted. The reflection and refraction of light can be described by the law of reflection and Snell’s law.
Law of Reflection
The law of reflection states that the angle of incidence equals the angle of reflection. When a beam of light hits a reflective surface like a mirror, it bounces off at the same angle as it arrived.
For example, if a light beam hits a mirror at a 30° angle relative to the normal (perpendicular line), it will reflect off the mirror at a 30° angle on the other side of the normal. The incident and reflected angle are always equal on opposite sides of the normal.
Refraction
When light travels from one medium to another, such as from air to water, it changes direction slightly. This bending of light as it passes between media is called refraction.
The amount of bending depends on the refractive indices of the two media. The higher the difference in refractive indices, the more the light will bend.
As light enters a more optically dense medium like water or glass, it slows down and bends towards the normal line. As it leaves a dense medium, it speeds up and bends away from the normal line.
Snell’s Law
The relationship between the angles of incidence and refraction at the boundary between two media is described by Snell’s law:
n1 * sin(θ1) = n2 * sin(θ2)
Where n1 and n2 are the refractive indices of the two media, and θ1 and θ2 are the angles measured from the normal of the incident and refracted light beams respectively.
Snell’s law shows that the angles of incidence and refraction depend on the change in refractive index when light crosses a boundary between media.
Total Internal Reflection
When light travels from a medium with a higher refractive index to one with a lower refractive index, there is a critical angle at which the refracted light will be bent at 90°. Beyond this critical angle, all the light is reflected back into the denser medium rather than transmitted. This phenomenon is called total internal reflection.
Total internal reflection allows light to be trapped and guided within materials like glass and fiber optics, enabling technologies like endoscopes and telecommunications.
Color and Vision
Color is a phenomenon of light perception that arises from different wavelengths striking the retina of the eye. Visible light is part of the electromagnetic spectrum and ranges from violet light with short wavelengths around 380-450 nanometers to red light with longer wavelengths of 620-750 nanometers.
The human eye contains two types of photoreceptor cells that are responsible for vision – rods and cones. Cones allow us to perceive color through specialized photopigments that absorb light at different wavelengths. There are three main types of cones: S-cones, M-cones, and L-cones that are most sensitive to short, medium and long wavelengths of light respectively.
When light of different spectral compositions hits the cones, the relative level of stimulation of the different cone types allows the brain to perceive all the colors of the visible spectrum through a phenomenon known as trichromatic color vision. The perception and identification of thousands of possible colors arises through the combination and ratio of stimulation of the three cone types.
The distribution of cones across the retina is not uniform and color perception can vary based on factors like aging and genetics. Color perception is a complex neuro-biological process that allows humans to make visual distinctions in the world.
Optics
Optics is the branch of physics that studies the behavior and properties of light. Some of the key concepts in optics include:
Lenses
Lenses use refraction to focus or disperse light beams. The most common types of lenses are converging lenses, which focus parallel rays of light to a single point, and diverging lenses, which spread out parallel rays of light. The focal length of a lens determines its focusing power.
Mirrors
Mirrors use reflection to redirect light. Plane mirrors reflect light at the same angle as the incoming light beam. Curved mirrors can focus or disperse light depending on their shape. Concave mirrors converge parallel light rays while convex mirrors cause divergence of light.
Diffraction
Diffraction occurs when light passes through a narrow opening or past a sharp edge, causing bending and spreading out of the light waves. Diffraction gratings and patterns demonstrate this wave effect of light.
Interference
The principle of superposition of waves causes interference patterns to emerge when two or more light waves overlap. Constructive interference occurs at points where crests align, increasing amplitude. Destructive interference happens when a crest aligns with a trough, cancelling out the waves.
Polarization
Polarizing filters only allow light waves with a specific orientation to pass through, blocking waves at other angles. This can reduce glare, enhance contrast, or reveal properties of materials. Light can also be polarized by reflection and scattering.
Light and Matter
Light and matter interact in several key ways that illuminate the quantum nature of light. When light shines on some materials, electrons can be ejected through the photoelectric effect. The energy of these emitted electrons depends on the frequency, not the intensity, of the light. This demonstrates that light comes in quantized packets of energy called photons.
The photon energy equals the frequency of the light multiplied by Planck’s constant. By absorbing photons, electrons in atoms and molecules can transition between discrete energy levels. The exact frequencies of light absorbed or emitted in these transitions correspond to the differences in energy levels. This gives every atom and molecule a unique emission spectrum and absorption spectrum.
The photoelectric effect, quantized energy levels, and spectral lines all provide direct evidence that light behaves as both particles and waves. Matter and light interact by exchanging energy in discrete amounts, with the quantum of light being the photon. Analyzing the interactions between light and matter has unlocked many of the mysteries of quantum physics.
Quantum Properties
One of the most fascinating aspects of light is its quantum nature. At very small scales, light exhibits properties of both waves and particles. This phenomenon is known as wave-particle duality.
Light’s energy is quantized into discrete packets called photons. The energy of a photon is proportional to its frequency. Photons demonstrate particulate properties by colliding with electrons and imparting quantized amounts of energy. Yet photons also interfere with each other like waves.
This quantum behavior allows many unique phenomena and applications. For example, the photoelectric effect demonstrates light transferring energy in photon packets to eject electrons from metals. Individual photons can be manipulated using quantum optics to carry information for quantum computing and cryptography. On the microscopic scale, light’s quantum properties are crucial to understanding its interactions with matter.
The discovery of light’s quantum nature in the early 20th century revolutionized physics and our understanding of reality. It remains an active area of research today, with many mysteries still to unravel regarding the wave-particle duality of light and other quantum particles.
Relativistic Effects
According to Einstein’s theory of special relativity, the speed of light in a vacuum is the same for all observers, regardless of their motion relative to the light source. However, this principle leads to some counter-intuitive phenomena when considering observers moving at very high speeds relative to one another.
One of these effects is time dilation. This means that time passes slower for an observer who is moving relative to a stationary observer. For example, a clock moving along with a spaceship traveling at 99% the speed of light would tick much slower than an identical clock at rest on Earth. The faster the relative velocity, the greater the difference in the passing of time.
Length contraction is another relativistic effect. When an object moves at very high speeds, its length appears to contract or shorten along the direction of motion to an outside observer. So if a rocket flies past Earth at nearly the speed of light, it would look slightly scrunched or flattened to those watching it whiz by.
In addition, as an object approaches the speed of light its mass increases from the point of view of a stationary observer. This is called relativistic mass increase. The faster the object moves, the more its mass increases compared to its rest mass. This effect has to be taken into account when accelerating particles to great speeds in particle accelerators.
Applications of Light
Light has numerous practical applications in fields like imaging, manufacturing, medicine, and communications. Understanding the properties of light has enabled scientists and engineers to develop innovative technologies and tools.
In imaging, light is used for photography, microscopy, endoscopy, and other visualization techniques. High-speed and low-light imaging have advanced significantly with developments like image intensifiers and CMOS/CCD sensors. Specialized imaging like infrared, X-ray, and gamma imaging provide views beyond visible light.
The highly focused, intense beams of lasers have led to widespread uses in manufacturing, healthcare, entertainment, research, and defense. Lasers can precisely cut, weld, drill, mark, and heat materials. They enable high-precision surgery, makeup removal, lithography, holography, barcode scanning, and equipment alignment. Fiber lasers allow flexible delivery of laser light.
Fiber optic communication uses light pulses in optical fibers to transmit information. This allows for high bandwidth, low loss, and electromagnetic immunity compared to electrical transmission. Optical fibers also enable medical imaging techniques like endoscopy.
For illumination, properties of light like color temperature, CRI, and luminance are important design factors. LED and OLED lighting have revolutionized efficiency, dimming, form factors, and smart lighting capabilities.
Spectroscopy analyzes the interaction between light and matter to reveal material properties. It enables chemical sensing, material identification, and spectral signature matching. Common examples include IR spectroscopy, Raman spectroscopy, and spectral color analysis.
Overall, our deep understanding of light has enabled remarkable advances in science, healthcare, manufacturing, communications, and many other fields.