What Chemical Produces Energy?
Energy is essential for life. It powers everything from the movement of our muscles to the firing of our neurons. On a chemical level, energy is the ability to do work and bring about change. Organisms require chemical energy to carry out their daily activities and maintain the processes needed to sustain life. This chemical energy comes primarily from the breakdown of food molecules like fats, carbohydrates, and proteins.
The energy in food is stored in the chemical bonds that hold the molecules together. When these bonds are broken through metabolism, the stored energy is released and can be used to power biological reactions and processes. The main energy currency of the cell is a molecule called adenosine triphosphate, or ATP. ATP contains high-energy chemical bonds that can be split to release energy on demand. The breakdown of ATP provides the power that cells need to accomplish the reactions necessary for life.
ATP
ATP (adenosine triphosphate) is the main molecule that cells use for energy. It consists of an adenine nucleotide bound to three phosphate groups. The chemical structure of ATP contains the nitrogenous base adenine, the five-carbon sugar ribose, and three phosphate groups bound together.
The adenine and ribose make up the adenosine component. The three phosphate groups are linked together in series by high-energy phosphoanhydride bonds. It is these bonds between the phosphate groups that store energy. When the bonds are broken through hydrolysis, energy is released which can be used to power cellular reactions and biological processes.
ATP Production
ATP is produced through a series of chemical reactions known as cellular respiration. This process takes place in the cytoplasm and mitochondria of cells and converts energy from nutrients like glucose into ATP. There are three main stages of cellular respiration:
- Glycolysis – This initial stage takes place in the cytoplasm and breaks down glucose into pyruvate molecules.
- Krebs Cycle – Also called the citric acid cycle, this stage occurs in the mitochondrial matrix and further breaks down pyruvate into carbon dioxide while also generating energy carriers like NADH and FADH2.
- Oxidative Phosphorylation – This final stage takes place along the inner membrane of the mitochondria and harnesses energy from NADH and FADH2 to generate ATP via the electron transport chain and chemiosmosis.
Together, these three stages of cellular respiration convert the energy in fuel molecules like glucose into ATP through a series of redox reactions. The majority of ATP is generated in the final oxidative phosphorylation stage.
Glycolysis
Glycolysis is a metabolic pathway that breaks down glucose molecules into pyruvate. This process occurs in the cytoplasm of the cell and does not require oxygen. Glycolysis is the first step in cellular respiration and is common to both aerobic and anaerobic respiration.
In glycolysis, each molecule of glucose is broken down into two molecules of pyruvate. This process yields two ATP molecules and two NADH molecules per glucose molecule. There are two phases of glycolysis:
- The energy investment phase – ATP is used to phosphorylate glucose, converting it to glucose-6-phosphate.
- The energy payoff phase – glucose-6-phosphate is converted to pyruvate, producing ATP and NADH.
The enzyme hexokinase phosphorylates glucose in the first step of glycolysis. Additional enzymes like phosphofructokinase and pyruvate kinase catalyze intermediate steps. Glycolysis sets up the cell for further ATP production in the Krebs cycle and oxidative phosphorylation.
Krebs Cycle
The Krebs cycle, also known as the citric acid cycle or tricarboxylic acid (TCA) cycle, is the second stage of cellular respiration. This cycle occurs in the mitochondrial matrix and oxidizes pyruvate that was generated during glycolysis.
Pyruvate generated from glycolysis first gets converted to acetyl CoA. Acetyl CoA then enters the Krebs cycle by combining with oxaloacetate to form citrate. Through a series of oxidation reactions, citrate gets converted back to oxaloacetate, producing NADH, FADH2, and ATP molecules in the process.
Some key steps in the Krebs cycle oxidation of pyruvate include:
- Citrate is converted to isocitrate by the enzyme aconitase
- Isocitrate is oxidized and decarboxylated to form α-ketoglutarate
- α-ketoglutarate is oxidized to succinyl CoA, producing NADH
- Succinyl CoA is converted to succinate, making GTP
- Succinate is oxidized to fumarate, forming FADH2
- Fumarate is hydrated to malate
- Malate is oxidized to oxaloacetate, generating NADH
After each turn of the Krebs cycle, additional NADH, FADH2, and ATP are generated from pyruvate oxidation. These molecules then provide energy for later stages of cellular respiration.
Electron Transport Chain
The electron transport chain is a series of protein complexes and electron carrier molecules within the inner membrane of the mitochondria that shuttles electrons from donors to acceptors via redox reactions. This process couples the transfer of electrons with the pumping of protons (H+ ions) across the mitochondrial membrane, creating an electrochemical proton gradient that drives ATP production.
In the electron transport chain, electrons from NADH and FADH2 are passed along a series of acceptor proteins via oxidation-reduction reactions. The electron acceptors are organized in three main protein complexes: NADH dehydrogenase complex (Complex I), cytochrome bc1 complex (Complex III), and cytochrome c oxidase complex (Complex IV).
As electrons are transported along the complexes, protons are pumped from the mitochondrial matrix across the inner membrane into the intermembrane space, creating an electrochemical gradient. The gradient builds up a large amount of potential energy that will be used to produce ATP.
Chemiosmosis
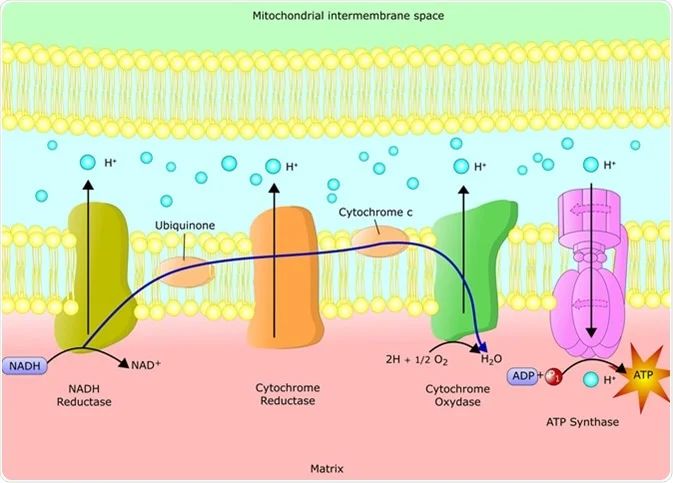
Chemiosmosis is the process that uses the flow of hydrogen ions (protons) across the mitochondrial membrane to generate ATP. As electrons move through the electron transport chain, energy is used to pump protons from the mitochondrial matrix into the intermembrane space. This creates an electrochemical proton gradient and a pH difference across the inner mitochondrial membrane. The accumulation of protons in the intermembrane space creates a concentration gradient and a charge separation.
The protons want to flow down this gradient back into the matrix, but they can only pass through special channels provided by ATP synthase. As the protons flow through ATP synthase, the enzyme harnesses the proton motive force to phosphorylate ADP, producing ATP. So in summary, the electron transport chain pumps protons and generates the proton gradient, while ATP synthase lets protons flow down their gradient and uses that energy to synthesize ATP. The generation and dissipation of the mitochondrial proton gradient is the essential final step of cellular respiration that allows us to utilize the energy from nutrients to produce ATP.
ATP Synthase
ATP synthase is an enzyme that plays a crucial role in the synthesis of ATP. It is located within the inner mitochondrial membrane and functions to couple the proton gradient formed by the electron transport chain to the phosphorylation of ADP to form ATP.
As protons flow through ATP synthase down their electrochemical gradient, the enzyme undergoes a conformational change that mechanically rotates its central stalk subunit. This rotation causes conformational changes in the catalytic sites, driving the phosphorylation of ADP. Specifically, ATP synthase binds ADP and phosphate, then uses the energy from the proton gradient to phosphorylate the bound ADP, forming ATP.
The mechanical energy from proton flow through the enzyme provides the energy needed to form the high-energy phosphate bonds in ATP. Without ATP synthase, the energy stored in the proton gradient created by the electron transport chain could not be harnessed into a usable form for the cell. Overall, ATP synthase serves as a critical link between the processes of cellular respiration and the production of ATP needed to power cellular processes.
Aerobic vs Anaerobic Respiration
Aerobic and anaerobic respiration differ in the processes they use to produce ATP. Aerobic respiration requires oxygen and occurs in three main stages – glycolysis, the Krebs cycle, and the electron transport chain. The electron transport chain, which takes place in the mitochondria, is the stage that requires oxygen. Anaerobic respiration does not require oxygen and only involves the glycolysis stage.
Aerobic respiration produces much more ATP than anaerobic respiration. Aerobic respiration produces around 36 ATP molecules per glucose molecule during cellular respiration. Anaerobic respiration produces only 2 ATP molecules per glucose molecule during glycolysis. This is because the Krebs cycle and electron transport chain, which account for the majority of ATP production, cannot occur without oxygen.
Aerobic respiration is more efficient and provides more energy to cells. Anaerobic respiration provides less energy but allows cells to continue producing some ATP without oxygen. Some cells and organisms can switch between aerobic and anaerobic respiration depending on oxygen availability. The end products of respiration also differ – aerobic respiration produces carbon dioxide and water, while anaerobic respiration produces lactic acid in animals and ethanol or lactate in plants and microbes.
Conclusion
In summary, the key points are:
- ATP is the main chemical that provides energy to power cellular reactions.
- ATP is produced through cellular respiration, which has three main pathways: glycolysis, the Krebs cycle, and the electron transport chain.
- Glycolysis breaks down glucose into pyruvate and produces a small amount of ATP without oxygen.
- The pyruvate enters the Krebs cycle which further breaks it down and produces more ATP.
- The electron transport chain, powered by the products of the Krebs cycle, creates a proton gradient that drives ATP synthase to produce the majority of ATP.
- Chemiosmosis couples the electron transport chain with ATP synthase via the proton gradient.
- Aerobic respiration that utilizes oxygen is far more efficient at producing ATP than anaerobic respiration.
In conclusion, ATP is the key chemical that cells use to power their metabolic activities, and it is predominantly produced through the interlinking pathways of cellular respiration.