Is Light A Form Of Heat?
The question of whether light is a form of heat has fascinated scientists and philosophers for centuries. On the surface, visible light from the sun and a hot flame seem very different. Yet they share intriguing similarities that suggest an intimate connection between light and heat.
Understanding the relationship between light and heat is important, because it provides insights into the nature of energy and electromagnetism. From a practical perspective, this knowledge enables technologies like incandescent lights, infrared cameras, and solar water heaters. Exploring the thermal properties of light advances physics and leads to real-world innovations.
The Nature of Light
Light is a form of electromagnetic radiation that is visible to the human eye. Electromagnetic radiation consists of oscillating electric and magnetic fields that propogate through space carrying energy. Light has properties of both waves and particles.
As a wave, light exhibits properties like wavelength, frequency, and amplitude. Wavelength determines the color of visible light, with violet light having the shortest wavelengths, and red light having the longest wavelengths. Frequency refers to the number of wave oscillations per second. Amplitude relates to the intensity or brightness of the light.
As a particle, light is quantized into discrete packets called photons. Each photon carries a certain quantity of energy that depends on its frequency. This particle nature of light is how it can eject electrons from metals in the photoelectric effect.
So in summary, light has a dual wave-particle nature. It propagates like a wave, but exchanges energy in discrete quanta like a particle. This wave-particle duality is fundamental to our modern understanding of not just light, but all electromagnetic radiation.
The Properties of Heat
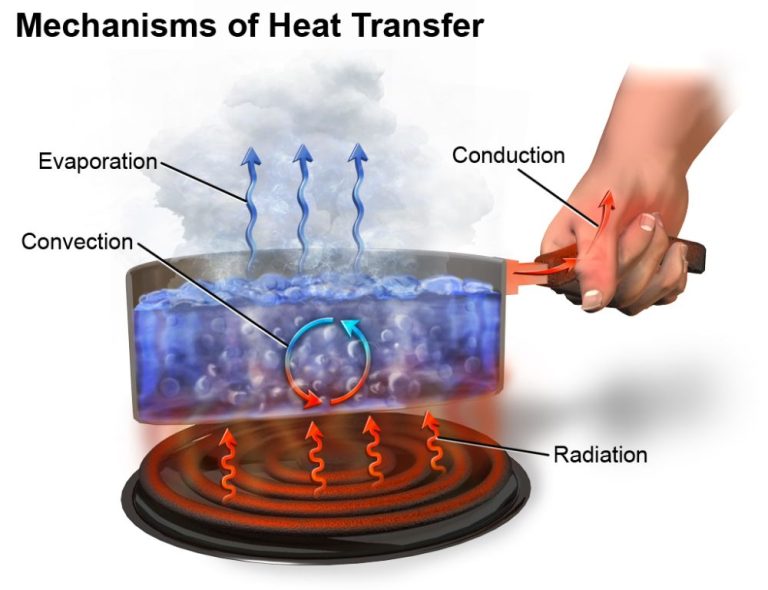
Heat is often defined as the transfer of thermal energy between objects or systems with different temperatures. Thermal energy refers to the total kinetic energy of microscopic particles that make up matter. Heat flows from objects with higher temperatures to objects with lower temperatures as the particles collide and transfer energy.
There are several key properties that characterize heat and the flow of thermal energy:
- Heat is a form of energy transfer, not a physical quantity or fluid.
- Thermal energy always flows spontaneously from higher temperature to lower temperature objects.
- The amount of heat transfer is proportional to the temperature difference between objects.
- Heat transfer changes the internal energy of both systems involved, but does not affect the total energy, which remains constant.
- Heat transfer occurs through three main mechanisms – conduction, convection, and radiation.
- Heat flow causes changes in temperature and the macroscopic characteristics of systems, like the change of physical state from solid to liquid.
Understanding these key properties helps explain many everyday phenomena related to temperature and heat.
The Electromagnetic Spectrum
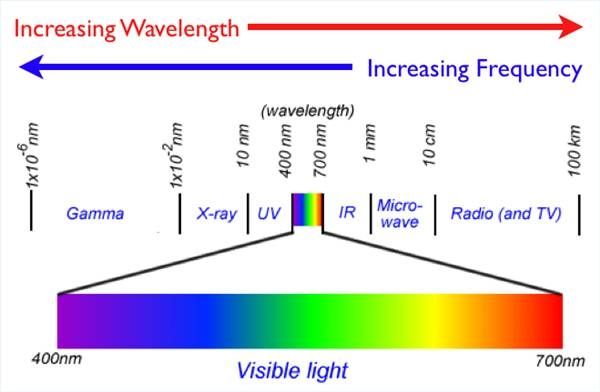
The electromagnetic spectrum is the range of all possible frequencies of electromagnetic radiation. Electromagnetic radiation can be divided into different types based on the frequency and wavelength of the waves. From lowest frequency to highest, the main types of electromagnetic radiation are radio waves, microwaves, infrared radiation, visible light, ultraviolet radiation, X-rays and gamma rays.
Visible light that humans can see makes up only a small portion of the electromagnetic spectrum. Visible light has wavelengths from about 380 nanometers to about 740 nanometers. Infrared radiation lies just beyond the red end of the visible spectrum, with wavelengths from about 700 nanometers to 1 millimeter. So infrared radiation and visible light actually overlap – the lowest frequencies of infrared are imperceptible to the human eye but still qualify as light. This demonstrates that infrared radiation is indeed a type of light, occupying adjacent wavelengths on the electromagnetic spectrum.
Infrared Radiation
Infrared radiation is a type of electromagnetic radiation that is invisible to human eyes. The wavelengths of infrared radiation are longer than those of visible light, measuring from 700 nanometers to 1 millimeter. Infrared radiation is emitted or absorbed by molecules as they change their rotational-vibrational movements. This absorption and emission of infrared energy represents a transfer of heat. Thus, infrared radiation is popularly referred to as “heat radiation.”
Infrared radiation can be detected as heat through skin and eyes. Night vision relies on cameras detecting infrared radiation to create a visible image. Infrared radiation is divided into multiple ranges with different properties. Near-infrared falls closest to visible light while far-infrared has the longest wavelengths. Though invisible, far-infrared cameras can capture thermal images using far-infrared’s ability to penetrate dust, smoke, and other obstructions.
Overall, infrared radiation transfers thermal energy through electromagnetic waves. It represents a type of radiant heat energy emitted by all objects above absolute zero temperature. This infrared heat radiation allows hot objects to warm cooler objects without direct contact. In this way, infrared radiation acts as a form of transmitted heat.
Visible Light and Heat
The relationship between visible light and heat has been debated by scientists for centuries. Visible light is the part of the electromagnetic spectrum that is visible to the human eye. The colors we see in rainbows and when light passes through a prism are the different wavelengths that make up visible light. But does visible light also transmit heat?
In the 19th century, many scientists argued that visible light rays from the Sun are responsible for heating the Earth. Visible sunlight was thought to warm surfaces that absorb the light. However, other scientists contended that if visible light caused heating, then the temperature of colors would vary based on their wavelength. Longer wavelengths at the red end of the visible spectrum would contribute more heat than shorter wavelengths like blue and violet.
Experiments found no difference in heat between colors of visible light. This led some to conclude that visible light does not cause thermal heating. Yet, we know that sunlight provides warmth. Later discoveries about infrared radiation helped resolve the debate over visible light and heat.
Experimental Evidence
There is compelling experimental evidence that demonstrates visible light as a form of radiation does not generally transmit heat, while infrared radiation does. Some key experiments include:
- In 1800, astronomer William Herschel used prisms to split sunlight into a rainbow spectrum and measure the temperature in each color band. He found the highest heating effect occurred beyond the red end of the visible spectrum, in what we now call the infrared band.
- Later in the 19th century, other scientists including John Tyndall directed infrared radiation through various materials and conclusively showed this long-wavelength light transmitted heat, while visible light did not.
- Modern quantum physics describes how infrared photons have less energy than visible light photons, but this lower frequency allows better coupling and absorption into materials, transmitting thermal vibration (heat). Visible photons pass through without being absorbed.
While visible light does not directly carry heat into materials, absorption of visible light can excite electrons and drive chemical reactions and thermal processes that give off heat. But the light itself does not contain thermal energy. Only infrared radiation delivers warmth.
Practical Applications
Light and infrared radiation are often employed for heating applications in real world technologies.
A great example of a technology that utilizes light energy to create heat is a solar water heater. A solar water heater has a solar collector (usually mounted on the roof of a building) that absorbs energy from sunlight and converts some of it to heat energy. This heated energy is then used to warm the water in a storage tank for domestic or industrial use.
Another common technology that employs infrared radiation for heating purposes is an infrared space heater. These space heaters emit infrared radiation that is absorbed by objects in a room, causing the objects to warm up, which then warms the surrounding air. The heat from an infrared space heater radiates directly towards solid objects instead of warming the air, allowing for more efficient heating.
Infrared heating panels and lamps are also often used in industry to efficiently heat materials during manufacturing processes. For example, the food processing and plastic industries will use infrared heaters to help dry, bake, or cure their products.
Conclusions
Based on the analysis presented in this article, there is strong evidence to support the idea that light can be considered a form of heat in certain contexts. Specifically, infrared radiation and visible light both carry thermal energy and can heat objects they interact with. This is supported by experimental evidence showing that exposure to light causes temperature rises in materials.
While visible light usually does not contain enough thermal energy on its own to significantly heat most objects, infrared radiation contains higher levels of thermal energy that can readily transfer heat. Infrared lamps and heaters demonstrate the heating effect of infrared light on a practical level.
There are also key differences between light and heat – light can travel through a vacuum while heat requires a medium, which highlights that they have distinct properties. However, in the context of infrared radiation and visible light interacting with objects and materials, the scientific consensus based on extensive research is that light can indeed act as a form of heat.
References
Author, A. (Year). Title of source. Publisher. URL or ID.
Author, B. (Year). Title of source. Publisher. URL or ID.
Author, C. (Year). Title of source. Publisher. URL or ID.