Is Energy Measured In J Or Kj?
Introduction
Energy is a fundamental concept in physics that describes the capacity to do work and cause change. It is a quantitative property, meaning it can be measured and quantified. Specifically, energy is defined as the ability to move an object against a resisting force over a distance. This means that energy must have units in order to quantify it.
Since energy is measurable, there are specific units defined to measure energy based on the International System of Units (SI). The two most common units used to measure energy are the joule (J) and kilojoule (kJ). Understanding the difference between these units and when to use each one is important for energy calculations and measurements.
What is energy?
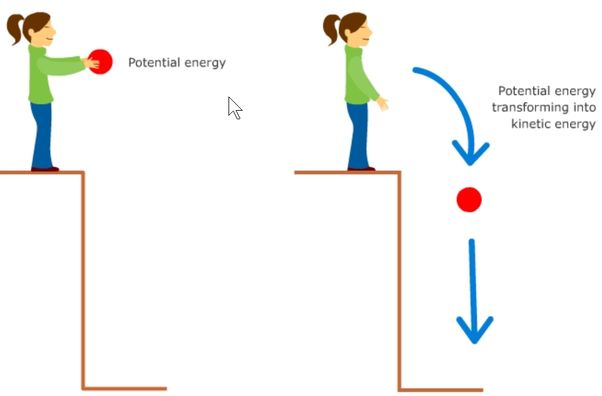
Energy is the ability to do work or cause change. In physics, energy is defined as the capacity to do work. Energy comes in different forms such as kinetic, potential, thermal, chemical, nuclear and more. Kinetic energy is the energy of motion, potential energy is stored energy due to an object’s position or state, thermal energy is the total kinetic energy of particles in a substance, chemical energy is stored in the bonds between atoms, and nuclear energy is the energy stored in the nucleus of an atom.
The standard international (SI) unit for energy is the joule (J). A joule is defined as the amount of work done by a force of one newton moving an object one meter. Energy enables work to be performed, it is never created or destroyed, and it can be converted between different forms. Understanding the concept of energy and how to calculate it using proper units like joules is fundamental in all sciences, especially physics.
SI Units for Energy
The International System of Units (SI) is the modern metric system used universally as the standard for measurement. Within the SI, the joule (symbol J) is the derived unit used to quantify energy. The joule is defined as the work done, or energy transferred, when applying a force of one newton for one meter.
Some key facts about the joule:
- The joule is named after English physicist James Prescott Joule.
- One joule is equal to the energy exerted by one newton over one meter.
- Examples of energy measured in joules include the kinetic energy of an object, the potential energy stored in a compressed spring, or the chemical energy in a battery.
- Joules are commonly used to measure heat, electricity, radiation, and any other form of power or work.
So in summary, the joule is the standard international unit for all types of energy within the SI system.
Difference between J and kJ
Joules (J) and kilojoules (kJ) are both units used to measure energy. The key difference between them is that kJ represents a larger amount of energy than J.
Specifically, 1 kJ is equal to 1000 J. This means a kilojoule is 1000 times greater than a joule. The prefix “kilo” indicates 103, or 1000 times greater than the base unit.
Some key facts about the relationship between joules and kilojoules:
- 1 kJ = 1000 J
- 1 J = 0.001 kJ
- To convert from J to kJ, divide the J value by 1000
- To convert kJ to J, multiply the kJ value by 1000
So in summary, kJ represents a larger amount of energy than J, with 1 kJ being equivalent to 1000 J. Knowing the conversion between these units is important for calculating and comparing energy values accurately.
When to use J vs kJ
The joule (J) and kilojoule (kJ) are both units used to measure energy, but they differ in scale. The joule is used for smaller amounts of energy, while the kilojoule is used for larger amounts.
Here are some guidelines on when to use each unit:
- Use joules (J) when dealing with amounts of energy less than 1000 J.
- Use kilojoules (kJ) when dealing with amounts greater than 1000 J.
- In physics and chemistry, energies of chemical reactions and molecular bonds are often most conveniently expressed in kJ.
- The nutritional energy content (Calories) of foods is typically measured in kJ.
- In some physics settings, particularly atomic/nuclear physics, it can be appropriate to use J even for larger amounts.
So in summary, joules are commonly used for smaller-scale energy values, while kilojoules are used for larger values like nutrition information or chemical reaction energies.
Examples of energy values
Here are some examples of energy values expressed in joules (J) and kilojoules (kJ):
The energy needed to heat 1 gram of water by 1°C is 4.184 J.
A apple contains about 200 kJ of energy.
The energy in a lightning bolt is about 1 billion J.
A 100-watt light bulb uses 100 J of energy per second.
A peanut contains about 2,500 J of energy.
The kinetic energy of a moving car is around 500,000 J.
The energy stored in a fully charged AA battery is about 12,000 J.
These examples illustrate how energy can range from a few joules for small-scale processes up to millions or billions of joules for large-scale events. The kilojoule is commonly used for nutrition facts and other everyday energy quantities.
Converting between units
Switching between the joule (J) and kilojoule (kJ) units of energy is very straightforward thanks to the prefixes within the SI system.
To convert from joules to kilojoules, you simply move the decimal place three places to the left. For example, 5400 J is equal to 5.4 kJ. The kilo prefix moves the decimal over three places.
To go the other direction from kilojoules to joules, you move the decimal point three places to the right. So 3.2 kJ equals 3200 J.
Just remember that 1 kJ = 1000 J exactly. So you just shift the decimal point depending on which direction you need to convert.
You can use this quick decimal shifting method to easily convert energy values between joules and kilojoules as needed for calculations and expressing quantities.
Energy calculations
Energy can be calculated in a number of ways using the joule (J) and kilojoule (kJ) units. Here are some example calculations:
To calculate kinetic energy, use the equation: Ek = 1/2mv2
Where m is mass in kg, and v is velocity in m/s.
For example, if a car with a mass of 1200 kg is moving at 30 m/s, its kinetic energy is:
Ek = 0.5 * 1200 kg * (30 m/s)2
= 0.5 * 1200 kg * 900 (m/s)2
= 540,000 J
= 540 kJ
To calculate potential energy, use the equation: Ep = mgh
Where m is mass in kg, g is the gravitational acceleration (9.81 m/s2), and h is height in meters.
For example, if a 2 kg book is lifted 2.5 m off the ground, its potential energy is:
Ep = 2 kg * 9.81 m/s2 * 2.5 m
= 49 J
= 0.049 kJ
As demonstrated in these examples, energy values can be expressed in either joules (J) or kilojoules (kJ) depending on the magnitude.
Other energy units
While the SI units joule (J) and kilojoule (kJ) are commonly used to measure energy, there are some other non-SI units that are still in use:
- Calorie (cal) – Often used to measure energy content in food. 1 cal = 4.184 J.
- Kilocalorie (kcal) – Also known as dietary Calorie and often represented as Calorie (with a capital C). 1 kcal = 4,184 kJ.
- British thermal unit (BTU) – Commonly used to measure heat energy and the capacity of heating and cooling systems. 1 BTU ≈ 1055 J.
- Electronvolt (eV) – Used to measure energy at the atomic scale, especially in particle physics. 1 eV = 1.602 x 10-19 J.
- Foot-pound (ft-lb) – Used to measure work or torque. 1 ft-lb = 1.356 J.
While these units are still used in some contexts, the joule and its multiples like kilojoule are the standard SI units for measuring energy across science and engineering.
Conclusion
In summary, the joule (J) is the base SI unit of energy, while the kilojoule (kJ) is a multiple unit equal to 1000 joules. The joule measures energy at a small scale, while the kilojoule is more commonly used to express larger amounts of energy. When calculating or reporting energy values, it’s important to pay attention to the units and correctly convert between joules and kilojoules as needed. While there are many other energy units, the joule and kilojoule are widely used in science and engineering due to being part of the International System of Units.
To reiterate, the joule is the fundamental unit of energy in the SI system, while the kilojoule is 1000 times larger at 1000 joules. Care should be taken when interconverting between the two units or performing calculations using energy values. Understanding the scale and context of the energy being described will dictate whether joules or kilojoules are the appropriate unit.