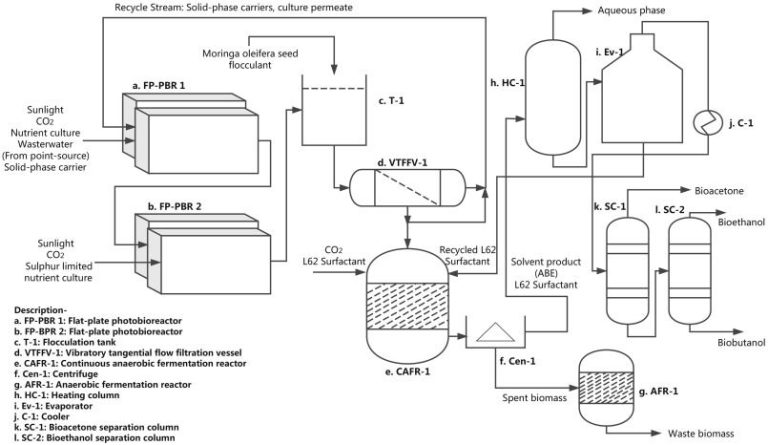
How Is The Heat Defined?
Heat is a form of energy that flows from one body to another due to their temperature difference. The study of heat is crucial as it plays an important role in fields like thermodynamics, heat transfer, and more. Understanding the principles of heat allows us to build more efficient engines, improve industrial processes, and stay comfortable in our homes and workplaces.
Heat is also a fundamental concept in physics that is related to the internal kinetic energy of molecules and atoms that make up matter. The hotter an object is, the faster its molecules vibrate and move. When two objects at different temperatures come in contact, heat flows spontaneously from the hotter to the colder body until they reach thermal equilibrium.
This article will provide a comprehensive overview of the scientific meaning of heat, how it is measured and transferred, the difference between heat and temperature, its real-world effects and applications, and more. By the end, you will have a deeper understanding of this ubiquitous form of energy in our universe.
Scientific Definition
Heat is defined scientifically as the transfer of thermal energy between objects or systems that are at different temperatures. Thermal energy refers to the total kinetic and potential energy of atoms and molecules, while temperature measures the average kinetic energy of particles. Heat flows spontaneously from objects at higher temperatures to objects at lower temperatures until they reach thermal equilibrium.
On a microscopic scale, heat arises from the random motions and vibrations of atoms and molecules. Higher temperature means the particles have greater average kinetic energy, allowing them to collide into neighboring particles and transfer energy. Heating increases molecular motion, while cooling decreases it. Heat is essentially the macroscopic measure of this microscopic particle energy transfer.
Heat always flows from hotter to colder objects when they are placed in contact, as described by the second law of thermodynamics. This occurs because higher energy particles will collide with and transfer energy to lower energy particles. Energy is not destroyed during this process, but simply dispersed more evenly between both objects.
While related, heat and temperature are distinct concepts. Temperature measures the average kinetic energy of particles, whereas heat refers to the total energy transferred between objects due to temperature differences. An object may contain thermal energy due to its molecular activity but not lose any heat if it is thermally isolated.
Measuring Heat
Heat energy is commonly measured in a few standard units:
– Calories (cal): This is the amount of heat energy needed to raise the temperature of 1 gram of water by 1°C.
– Joules (J): The joule is the SI unit of energy and work. 1 calorie is equal to 4.184 joules.
– British thermal units (BTU): This is a traditional unit of heat still used today, especially for heating systems. 1 BTU is the amount of heat required to raise 1 pound of water by 1°F.
– There are also larger units like the kilocalorie (Calorie with a capital C), which equals 1000 calories. Food energy is often measured in kilocalories.
To measure heat, instruments like thermometers, calorimeters, and thermistors are used. Knowing the specific heat capacity of a material allows you to calculate the energy transferred as heat using the measured temperature change.
Heat Transfer
Heat is transferred between objects or systems through three main mechanisms – conduction, convection, and radiation.
Conduction
Conduction is the transfer of heat between objects that are in direct contact with each other. Heat flows from the hotter object to the cooler one until they reach thermal equilibrium. Metals are good conductors of heat because their atoms vibrate easily and can transfer energy to neighboring atoms.
Convection
Convection is the transfer of heat by the motion of fluids. When a liquid or gas is heated, it expands, becomes less dense, and rises. As it rises, the cooler surrounding fluid circulates to take its place, causing the continuous motion that transfers heat. Convection occurs in the Earth’s mantle, atmosphere, and oceans.
Radiation
Radiation is the transfer of heat energy by electromagnetic waves. All objects emit infrared radiation based on their temperature. Hotter objects radiate more intensely than cooler objects. Radiation does not require direct contact between objects and can travel long distances through any medium, even through vacuums, which conduction and convection cannot.
Thermal Energy vs Temperature
Thermal energy and temperature are often used interchangeably, but they are distinct scientific concepts. Thermal energy refers to the total internal kinetic energy of molecules within a substance, while temperature is a measure of the average kinetic energy of those molecules.
Thermal energy depends on the mass, temperature, and chemical composition of an object. Increasing the amount of matter increases the total thermal energy, even if the temperature remains constant. Temperature measures the intensity of that internal kinetic energy — how rapidly the molecules are vibrating and moving. The same material can have high thermal energy but low temperature if the mass is large, or low thermal energy and high temperature if the mass is small.
For example, a bathtub filled with hot water has high thermal energy due to the large mass of water. Yet the temperature may not be very hot. Conversely, the burner on an electric stove may reach 600°F, so the metal has a very high average molecular kinetic energy. But the temperature drops rapidly if you touch it because the mass of the burner is low, so it contains much less total thermal energy.
In summary, thermal energy depends on both temperature and mass, while temperature measures the average molecular kinetic energy independent of mass. This distinction is important in the study of thermodynamics and heat transfer.
Effects of Heat
Heat causes several important effects that we can observe in everyday life. Two of the main effects are phase changes and thermal expansion.
Phase Changes
When heat is added or removed from a substance, it can cause that substance to change between solid, liquid, and gaseous states. For example, adding heat to solid ice turns it into liquid water, while removing heat from liquid water turns it into solid ice. These changes between states are called phase changes.
Heat must be added to a substance for it to melt (solid to liquid) or evaporate (liquid to gas). On the other hand, heat must be removed for a substance to freeze (liquid to solid) or condense (gas to liquid). The amount of heat required depends on the substance.
Thermal Expansion

Most substances expand when heated and contract when cooled. This is known as thermal expansion. The molecules in a substance gain kinetic energy as heat is added, causing them to vibrate and move farther apart, increasing the substance’s volume. Metals typically expand the most, while substances like water expand less.
Thermal expansion explains many common phenomena. For example, why gaps need to be left in railroad tracks and bridges to allow for expansion in hot weather. It also explains why hot air rises – as air is heated, it decreases in density and becomes more buoyant than the surrounding cooler air.
The effects of heat like phase changes and thermal expansion have widespread impacts on materials, devices, and systems at both the macro and microscopic levels.
Heat Capacity
Heat capacity is a material’s ability to store heat energy. It refers to the amount of heat required to raise a material’s temperature by 1 degree. Materials with higher heat capacities require more heat to increase their temperature.
The heat capacity of a material depends on its mass, specific heat, and temperature change:
Q = mcΔT
Where:
- Q = heat energy (J or calories)
- m = mass of material (kg or g)
- c = specific heat of material (J/g°C or cal/g°C)
- ΔT = change in temperature (°C or K)
Substances like water have a high specific heat, so they can absorb significant amounts of heat with little temperature change. Materials like metal have a low specific heat, so their temperature changes rapidly with heat addition or loss.
Engineers use heat capacity knowledge when designing systems that involve heating/cooling materials for manufacturing, cooking, construction, and more. For example, the high heat capacity of water enables its use as a coolant in nuclear reactors.
Heat Engines
A heat engine is a device that converts heat into mechanical energy or work. It operates on the basis of the laws of thermodynamics, specifically the second law which states that heat can spontaneously flow from a hotter body to a colder one. The most common heat engines are internal combustion engines and steam turbines.
In a heat engine, there is a hot reservoir, which could be something like a furnace or combustion chamber, and a cold reservoir, which could be the outside air or a body of water. The engine takes heat from the hot reservoir, converts some of it into work, and rejects the rest to the cold reservoir.
Some examples of heat engines are:
- Internal combustion engines – The hot reservoir is the combustion chamber where fuel burns, the cold reservoir is the engine block or cooling system. The expanding gases from combustion apply pressure to move the pistons.
- Steam engines – The hot reservoir is the boiler where water is heated to high pressure steam. The steam applies pressure to move the pistons or turn the turbine blades. The condensed steam is the cold reservoir.
- Gas turbines – Hot gases from combustion turn the blades of the turbine. The turbine spins a shaft for power. The exhaust gases are expelled as the cold reservoir.
- Thermoelectric generators – A temperature difference across a thermoelectric material causes electrons to move and generate current. The hot side is heated by a fuel source.
Heat engines require a continual input of heat from the hot reservoir to keep operating. Their efficiency is limited by thermodynamic principles to how much of the heat input can be converted into useful work. However, they allow the extraction of mechanical work from the flow of heat, which makes them very useful in many applications.
Real-World Applications
Heat has many practical applications in everyday life. Here are some of the most significant real-world uses of heat:
Cooking
Heating food is essential for cooking. Applying heat breaks down proteins and plant cell walls, changing the chemistry, texture, flavor, and digestibility of ingredients. Common cooking methods like baking, frying, grilling, etc. all rely on heat to transform raw ingredients into delicious meals.
Heating
Heat provides warmth and comfort indoors. Buildings are heated using furnaces, boilers, electric baseboard heaters, and other systems that raise indoor air temperature during cold weather. Controlling ambient heat prevents hypothermia and creates habitable interior spaces.
Thermodynamics
The branch of physics called thermodynamics studies heat and its relation to energy flows in systems. This field underlies modern power generation, refrigeration, air conditioning, combustion engines, and more. Practical applications of thermodynamics principles enable large-scale control over heat transfer processes.
Conclusion
In summary, heat is defined scientifically as the transfer of thermal energy between objects or systems due to a temperature difference. While related, heat and temperature are distinct concepts. Heat describes the total kinetic energy of molecules while temperature measures the average energy in a substance. Understanding heat transfer through conduction, convection and radiation is key to many technologies and real-world applications from engines to weather patterns. By grasping the nuances around defining and measuring heat, we gain a deeper appreciation for an everyday phenomenon that powers our lives.