How Can You Prove That Light Is A Form Of Energy?
Light has properties of both waves and particles (Smith 2022). This phenomenon is known as “wave-particle duality” and means that light behaves like both an electromagnetic wave and as particles called photons. As a form of electromagnetic radiation, light radiates energy in the form of oscillating electric and magnetic fields. But light also has particle properties, since it transfers energy in discrete packets called photons. The fact that light exhibits wavelike and particle-like properties means that it fundamentally behaves as a form of energy.
Definition of Energy
Energy is defined as the ability to do work or cause change (https://www.britannica.com/science/energy). In physics, energy is considered a quantitative property that is transferred to an object or physical system and can be recognized by the performance of work. Energy exists in various forms such as potential, kinetic, thermal, electrical, chemical, nuclear and more (https://ingeniumcanada.org/scitech/education/tell-me-about/physics-of-energy). The common theme across all these forms is the capacity to do work and cause movement or change.
Energy can be converted from one form to another, but it cannot be created or destroyed according to the law of conservation of energy. For example, chemical energy in gasoline can be converted into kinetic energy to move a vehicle. The total energy before and after remains the same.
Light Radiates Energy
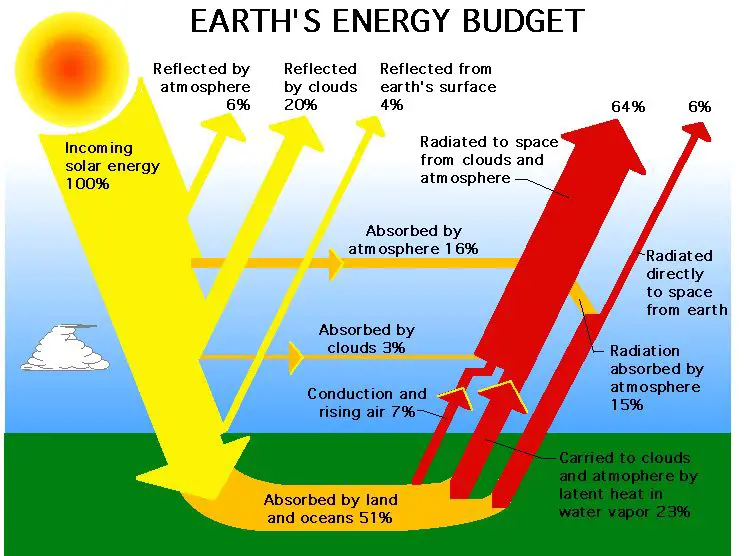
Light is a form of electromagnetic radiation that carries energy as it travels through space (https://www.esa.int/Science_Exploration/Space_Science/Energy_light_radiation_temperature). Light radiates energy in the form of electromagnetic waves, which are self-propagating oscillations of electric and magnetic fields. The energy in electromagnetic waves is directly proportional to their frequency and inversely proportional to their wavelength. When light interacts with matter, such as atoms and molecules, the energy carried by the electromagnetic waves can be absorbed and transformed into other forms of energy.
For example, visible light from the sun radiates energy that is absorbed by the chlorophyll molecules in plant leaves. The plants then use this light energy to power photosynthesis and produce chemical energy that fuels growth and other biological processes. So in summary, light is able to radiate energy across vast distances through space because its electromagnetic waves act as energy carriers.
Light Transfers Energy
Light can transfer energy from one object to another in several ways. For example, when light shines on a solar panel, it transfers energy to the solar panel which can then be used to generate electricity (https://www.cbsd.org/cms/lib/PA01916442/Centricity/Domain/3232/generationgenius_energy-transfer-lesson-for-kids-sound-light-heat-grades-3-5.pdf). The light energy is converted into electrical energy. Another example is photosynthesis in plants. When sunlight shines on the leaves of a plant, the light energy is absorbed by the plant and converted into chemical energy that the plant uses to grow and function.
Light can also transfer heat energy. Darker colored objects absorb light more than lighter colored objects. As light is absorbed, it turns into heat energy, causing the object to warm up. This is why wearing dark clothes on a sunny day makes you feel warmer than if you wore lighter colored clothes (https://www.varsitytutors.com/4th_grade_science-help/observe-how-energy-is-transferred-by-light). The light transfers heat energy to the dark object.
Overall, light transfers energy by being absorbed and converted into other forms of energy like electricity, chemical energy, and heat.
Light Exerts Radiation Pressure
Light exerts radiation pressure because it carries momentum. When light hits an object, it transfers some of its momentum to the object through radiation pressure. This transfer of momentum requires energy, demonstrating that light is a form of energy.
Radiation pressure arises from the exchange of momentum between light and the surface it interacts with, according to the law of conservation of momentum in physics. The photons that make up light have momentum [1]. When they collide with a surface, the surface absorbs some of the photons’ momentum, exerting a small force on the surface. This radiation pressure force requires the light to give up some of its energy.
The radiation pressure exerted by light can be calculated using the equation P = I/c, where P is the pressure in pascals, I is the intensity of the light in watts per square meter, and c is the speed of light in meters per second [2]. This equation demonstrates that light with greater intensity exerts a larger radiation pressure force, requiring more energy.
Radiation pressure from light is very small, but can have noticeable effects over large scales, such as causing the tails of comets to point away from the Sun. The ability of light to exert pressure and do mechanical work proves it carries energy.
Light Can Do Mechanical Work
Light exerts force and can do mechanical work through radiation pressure. When light shines on an object, it exerts a small force called radiation pressure. This happens because light has momentum, so when it hits an object and gets absorbed or reflected, it transfers some of that momentum to the object.
A good example of light doing mechanical work is a Crookes radiometer. This device consists of a partial vacuum glass bulb with a rotor inside that has four vanes. The vanes are black on one side and silver on the other. When light shines on the radiometer, the black sides of the vanes absorb more light than the silver sides. This creates more radiation pressure on the black sides, which pushes the rotor and causes it to spin.
Another example is using lasers to physically move or trap tiny objects through radiation pressure. Scientists have used this technique, called optical tweezers, to manipulate cells and microscopic particles. The force from the laser beam can hold and move the objects similar to tweezers.
Researchers have also demonstrated light exerting enough force to bend micron-thin silicon beams inside a chip (Popular Mechanics, 2023[1]). This effect could potentially be harnessed to build opto-mechanical devices powered entirely by light.
These examples show that light, although intangible, has momentum and can exert pressure and force to physically move objects when absorbed or reflected. This allows light to do measurable mechanical work.
Light Induces Chemical Reactions
Light can provide the activation energy needed to initiate chemical reactions. The energy that is associated with light, known as photons, can be absorbed by molecules. When a molecule absorbs light energy, it becomes “electronically excited,” which means it enters a higher energetic state. This excitation provides molecules with enough energy to overcome reaction barriers and take part in chemical reactions that would not otherwise occur.
The field of study related to light-induced chemical reactions is called photochemistry. Some common examples of photochemical reactions include photosynthesis, photodynamic therapy, and photopolymerization. In photosynthesis, plants use the light energy from the sun to convert carbon dioxide and water into carbohydrates and oxygen. This process stores the light energy in the chemical bonds of the newly synthesized carbohydrate molecules. In photodynamic therapy, light activates drug compounds which then interact with oxygen in the body to produce cell-damaging reactants, thereby destroying cancer cells. Photopolymerization utilizes light to convert liquid monomers into solid polymers and is used widely in curing processes for coatings, inks, and adhesives.
Overall, light irradiation supplies the needed activation energy to enable or accelerate many different chemical reactions. Quantum mechanical selection rules govern what specific chemical reactions can occur using light excitation. By understanding photochemistry, scientists can better control light-based chemical synthesis processes across disciplines including materials science, medicine, and energy research.
Cite url: https://pubs.acs.org/doi/10.1021/acs.jpclett.2c01119
Light Causes Heat
When light is absorbed by an object, the energy of the light is converted into heat energy, causing the object to increase in temperature. This occurs due to the interaction between the light photons and the atoms of the material.
Specifically, when a photon is absorbed by an atom, it excites the atom’s electrons to higher energy states. As the electrons then relax back down to lower energy states, they release energy in the form of heat. This causes the atoms and molecules in the material to vibrate more rapidly, increasing the thermal energy and temperature [1].
Different materials absorb different wavelengths of light. Darker colored objects tend to absorb more visible light energy than lighter colored objects. This explains why dark surfaces heat up more than light surfaces under the same illumination [2]. The light energy is efficiently converted into heating of the dark object.
In summary, light causes heating of objects because the photons’ energy is deposited into the material as heat when the light is absorbed. This excitation of atoms and increased molecular vibration manifests as a temperature increase.
Light Can Generate Electricity
When light interacts with matter, it can produce an electric current. This phenomenon is utilized in solar cells to convert sunlight into electrical energy.
In solar cells, photons from sunlight strike a semiconductor material like silicon. This transfers the photon’s energy to electrons in the semiconductor, causing them to break free of their atomic bonds. The freed electrons then flow through an external circuit as electricity (McGrath, 2019).
For example, photovoltaic (PV) solar cells contain a p-n junction made of two differently doped silicon layers. When sunlight hits the PV cell, photons excite electrons in the p-type layer. The built-in electric field at the junction separates positive and negative charges, driving electrons to the n-type layer and holes to the p-type layer. Metal contacts on the PV cell collect the electrons and holes, generating a DC electric current (Olympus Corporation, 2022).
Thus, PV solar cells and other light-sensitive materials like photodiodes directly convert absorbed light energy into electrical energy through the photovoltaic effect. This demonstrates that light does indeed carry energy that can generate electricity.
McGrath, M. (2019, October 29). A new way to convert light to electricity. Chemistry World. https://www.chemistryworld.com/news/a-new-way-to-convert-light-to-electricity/7915.article
Olympus Corporation. (2022). Introduction to Light and Energy. Olympus Life Science. https://www.olympus-lifescience.com/en/microscope-resource/primer/lightandcolor/lightandenergyintro/
Conclusion
As observed throughout this article, there is compelling evidence from multiple scientific fields demonstrating light behaves as a form of energy. Light radiates energy in the form of electromagnetic waves and photons. It can transfer heat energy by absorbing and emitting radiation. Light exerts radiation pressure from carrying momentum. Through photoelectric and photochemical effects, light induces chemical changes and generates electricity. Light also does mechanical work by exerting force during radiation pressure. All of these examples showcase light transferring or transforming energy in different forms. While exhibiting wave-particle duality, the particle aspect of light as photons shows it carries discrete quanta of energy. From photosynthesis in plants to solar panels generating electricity, light’s ability to produce chemical and electrical energy through biological and technological processes further confirms its nature as a fundamental form of energy in our universe.