Do Hydrogen Fuel Cells Have A Higher Energy Efficiency?
Hydrogen fuel cells are electrochemical devices that convert the chemical energy of hydrogen into electrical energy. They work by passing hydrogen through the anode of a fuel cell and oxygen through the cathode, separated by an electrolyte. This electrochemical reaction generates electricity, with water and heat as the only byproducts.
Energy efficiency is defined as the ratio of useful energy output from a system to the total energy input into the system. In the case of fuel cells, it measures how much of the fuel’s chemical energy is converted into electrical energy, rather than being lost as heat or other forms of waste energy. Improving energy efficiency is a key potential benefit of hydrogen fuel cells.
This article will examine the energy efficiency of hydrogen fuel cells, factors that affect it, how it compares to other energy technologies, and the challenges to improving efficiency further.
How Hydrogen Fuel Cells Work
Hydrogen fuel cells convert the chemical energy from hydrogen into electrical energy through an electrochemical reaction. This reaction takes place between hydrogen and oxygen across an electrolyte membrane. Hydrogen is fed into the anode side of the fuel cell and oxygen is fed into the cathode side. At the anode side, a catalyst causes the hydrogen atoms to split into protons and electrons. The protons pass through the electrolyte membrane to the cathode side while the electrons create an electric current that can be utilized before recombining with the protons and oxygen to form water. This flow of electrons is harnessed as electrical energy (1).
In essence, hydrogen fuel cells use hydrogen and oxygen to create electricity through a clean and efficient chemical reaction. The only byproducts are electricity, water and heat. Unlike batteries or combustion engines, fuel cells can run indefinitely as long as they are supplied with hydrogen and oxygen. This makes them well suited for providing continuous electrical power.
(1) https://www.thezebra.com/resources/driving/how-do-hydrogen-cars-work/
Efficiency of Hydrogen Fuel Cells
The theoretical maximum efficiency of a hydrogen fuel cell is approximately 83%, according to the U.S. Department of Energy (https://www.energy.gov/eere/fuelcells/hydrogen-fuel-cells). This is significantly higher than the maximum efficiency of internal combustion engines, which is around 35-45% (https://slideplayer.com/slide/4218543/).
However, most real-world hydrogen fuel cells operate at 40-60% efficiency range. This is still higher than combustion engines. According to Flux Power, hydrogen fuel cells can achieve up to 2 times the efficiency of comparable lead-acid batteries in material handling applications like forklifts (https://www.fluxpower.com/choosing-the-right-forklift-power-source).
The key factors affecting real-world hydrogen fuel cell efficiency include the fuel cell system design, operating conditions like temperature and pressure, and power load. While hydrogen fuel cells may not achieve maximum theoretical efficiency in practice, they still offer significantly higher efficiency than traditional internal combustion engines.
Factors Affecting Efficiency
Hydrogen fuel cell efficiency is impacted by several factors that affect the chemical reactions occurring within the fuel cell. Some key factors include:
Temperature – Higher operating temperatures allow for faster reaction kinetics within the fuel cell, improving efficiency. However, too high of a temperature can damage cell components. Typical PEM fuel cells operate in the range of 60-100°C (1).
Pressure – Increased reactant pressure results in higher partial pressures within the fuel cell, which improves electrode kinetics and increases voltage and efficiency. However, higher pressures require more robust components (2).
Fuel purity – Impurities in the hydrogen fuel can poison the electrodes, reducing efficiency over time. High purity levels of 99.999% are required for optimum performance (3).
Water management – Keeping the membrane hydrated while avoiding flooding improves ion conductivity and efficiency. Careful water management is needed, often requiring humidifiers and condensers (4).
By optimizing these factors, hydrogen fuel cell efficiency can be maximized. However, tradeoffs exist between efficiency, durability, and system complexity. Finding the right balance is an ongoing research challenge.
(2) https://www.energy.gov/eere/fuelcells/articles/fuel-cells-fact-sheet
(3) https://www.californiahydrogen.org/wp-content/uploads/files/doe_fuelcell_factsheet.pdf
Improving Efficiency
There are several promising research developments aimed at improving the efficiency of hydrogen fuel cells. Scientists at Los Alamos National Laboratory have developed a new fuel cell design that uses grooved electrodes, which enables higher power density and improved durability. Their design achieves up to 50% higher performance compared to existing technologies.
Additionally, researchers have discovered new ionic liquid materials that can boost efficiency. As described in a ScienceDaily article, these ionic liquids enable faster charging and discharging of protons in the fuel cell. Early testing indicates efficiency increases of 5-10%, which is a major advancement for commercial viability.
Further improvements in catalysts, membrane materials, and manufacturing processes will also incrementally improve efficiency over time. With continued research and development, experts are optimistic that fuel cell efficiency can reach levels comparable to internal combustion engines.
Challenges to Widespread Adoption
Despite their potential benefits, there are several key challenges that have hindered the widespread adoption of hydrogen fuel cell vehicles:
High upfront costs – Fuel cell vehicles currently cost much more than conventional gasoline vehicles. According to [1], the Honda Clarity Fuel Cell has an MSRP of around $58,490, compared to around $26,000 for the standard Honda Civic. The complex manufacturing and materials for fuel cell stacks contribute to the high costs.
Lack of fueling infrastructure – There are only around 48 public hydrogen fueling stations in the entire U.S. as of 2019 [2]. The limited fueling infrastructure makes it difficult for people to actually fuel and drive their vehicles outside of certain regions. More stations will need to be built to enable long distance travel.
Hydrogen production and delivery – Producing pure hydrogen sustainably and delivering it to fueling stations also presents logistical challenges. 95% of hydrogen today comes from natural gas, but greener production from water electrolysis is still expensive [3]. Distribution and delivery networks for hydrogen will need further development.
Storage limitations – Hydrogen has very low volumetric energy density compared to gasoline, and it needs to be stored at high pressures. This limits the driving range per tank, and presents some safety issues related to the high pressure tanks.
Safety concerns – While pure hydrogen is not toxic or radioactive, some education is still needed around the safety of high pressure storage compared to gasoline. Ensuring robust protocols and hardware for safe operation and crash integrity will help alleviate public concerns.
Hydrogen Production Efficiency
The efficiency of hydrogen production depends greatly on the source and method used. Some of the main ways to produce hydrogen include:
Steam Methane Reforming: This process uses high-temperature steam to produce hydrogen from natural gas. It has an efficiency of around 70-85% (1). However, as it relies on fossil fuels, it produces carbon emissions.
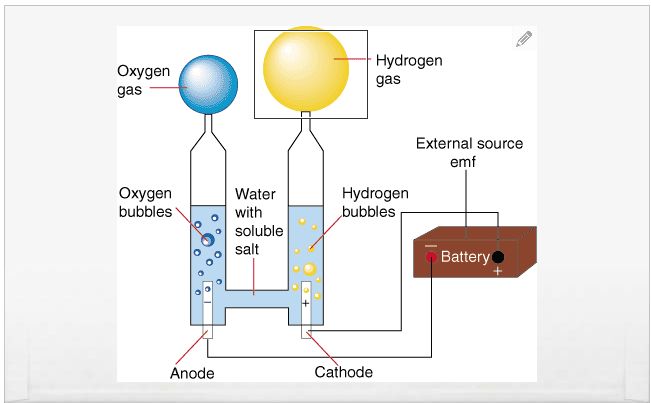
Electrolysis: Passing electric current through water splits it into hydrogen and oxygen. Electrolyzer efficiency ranges from 60-82%, depending on the technology used (2). While electricity can come from renewable sources, generating that electricity has its own efficiency losses.
Thermochemical Water Splitting: Heat and chemical reactions break water into hydrogen and oxygen. Efficiency can reach 50-60%. Significant heat is required, often 700-1000°C (3).
Photoelectrochemical Water Splitting: Combines electrolysis with photocatalysts driven by sunlight. Efficiency is currently 2-20%, with targets of 30% (4). Does not need electricity input but relies on sunlight.
In summary, hydrogen production efficiency depends greatly on the method. Processes powered by electricity or sunlight offer lower carbon emissions but have efficiency challenges. Fossil fuel-based methods are more efficient but produce greenhouse gases. Research is ongoing to improve efficiency across production methods.
(1) https://www.researchgate.net/topic/Hydrogen-Production
(2) https://www.researchgate.net/post/Is_it_time_to_think_other_catalyst_than_TiO2_for_water_splitting
Overall Energy Efficiency
When evaluating the energy efficiency of hydrogen fuel cell vehicles, it’s important to consider the full well-to-wheel efficiency rather than just the efficiency of the fuel cell itself. This accounts for the energy used to produce and transport the hydrogen fuel to the vehicle. Well-to-wheel analysis provides a complete picture of the total energy consumed.
According to a 2021 study by Zemo Partnership, the well-to-wheel efficiency of hydrogen fuel cell electric vehicles ranges from 22-27% depending on if the hydrogen is produced from natural gas reforming or water electrolysis (Zemo, 2021). This is lower than battery electric vehicles which have a well-to-wheel efficiency of 37-46%.
The lower well-to-wheel efficiency of hydrogen vehicles can be attributed to the energy intensive process of producing hydrogen fuel, especially if produced by water electrolysis. Natural gas reforming is more efficient but has higher emissions. Energy is also lost when converting hydrogen back to electricity in the fuel cell. Despite lower efficiency, hydrogen fuel cells offer advantages like rapid refueling and longer range compared to batteries.
Overall, hydrogen fuel cell vehicles have a moderate well-to-wheel energy efficiency but there are active research efforts to improve the efficiency of hydrogen production and fuel cells to increase total energy utilization.
Comparisons to Other Technologies
When it comes to efficiency, hydrogen fuel cells compare favorably to some alternative technologies, while lagging behind others.
Compared to traditional internal combustion engines, fuel cells can achieve much higher efficiencies. Gasoline engines typically have efficiency rates of around 20-30%, while diesel engines may reach 35-45% efficiency. Hydrogen fuel cells commonly operate in the 45-60% efficiency range, giving them a clear advantage over traditional combustion engines 1.
However, battery electric vehicles currently surpass fuel cell efficiency. Lithium-ion batteries have efficiency rates from 80-90%, higher than the 25-35% rates achieved by most hydrogen fuel cells today 2. The roundtrip efficiency of batteries, from grid to wheel and back, also exceeds that of producing and using hydrogen in a fuel cell vehicle.
Biofuels like ethanol and biodiesel represent another comparison point. The efficiency of biofuels varies greatly depending on the feedstock and production method used. More advanced biofuels can reach 40-50% efficiency or more. However, most commercially available biofuels today have lower efficiency rates than hydrogen fuel cells.
Overall, hydrogen fuel cells stand out for their high efficiency versus combustion engines. But batteries currently hold the efficiency advantage for electric propulsion. Continued research and development may help close the efficiency gap between fuel cells and batteries in the future.
Conclusion
To summarize, hydrogen fuel cells can have high energy efficiencies relative to combustion engines, but there are some important caveats. The fuel cell itself can convert hydrogen to electricity at efficiencies over 60%, but producing and delivering hydrogen fuel requires energy and lowers the overall system efficiency. Factors like fuel cell type, operating temperature, scale, and drive cycle all impact efficiency as well. Improvements in materials, system integration and hydrogen production methods continue to push efficiencies higher. But hydrogen fuel cells still face challenges around high costs and lack of fueling infrastructure before they can be adopted at scale. While hydrogen vehicles offer benefits like zero tailpipe emissions, the overall energy efficiency depends greatly on how the hydrogen itself is produced – using renewable electricity yields higher efficiencies than producing hydrogen from natural gas. In many cases today, battery electric vehicles offer higher well-to-wheel energy efficiency than hydrogen fuel cell vehicles. But there remains great potential to leverage hydrogen as an emissions-free fuel, especially for larger vehicles and long-range transportation needs. Continued development and adoption of hydrogen infrastructure could enable fuel cell vehicles to play a greater role in sustainable transportation in the future.